Scalable Synthesis of Dabigatran Etexilate Intermediates for Global Pharma Supply Chains
Scalable Synthesis of Dabigatran Etexilate Intermediates for Global Pharma Supply Chains
The pharmaceutical landscape for anticoagulant therapies has been significantly shaped by the introduction of direct thrombin inhibitors, with Dabigatran Etexilate standing as a cornerstone molecule in this class. As global demand for this critical medication continues to rise, the reliability of its supply chain becomes paramount, hinging heavily on the efficient production of its key precursors. Patent CN103539730A, filed in early 2014, addresses a critical bottleneck in this supply chain by disclosing robust preparation methods for the essential intermediate 3-[N-(2-pyridyl)-3-amino-4-methylamino benzamido]-ethyl propionate. This technical disclosure is not merely an academic exercise but a strategic response to the industrial limitations of prior art, offering pathways that enhance both safety and scalability for commercial manufacturing. By leveraging specific chemical innovations, this patent provides a blueprint for overcoming the stagnation often encountered when scaling nitro-reduction reactions, ensuring that procurement teams can secure high-quality intermediates without the risk of production delays.
![Chemical structure of the key Dabigatran intermediate 3-[N-(2-pyridyl)-3-amino-4-methylamino benzamido]-ethyl propionate](/insights/img/dabigatran-intermediate-synthesis-supplier-20260302221813-02.png)
The structural complexity of this intermediate necessitates precise control over functional group transformations, particularly the reduction of the nitro group in the presence of other sensitive moieties. The molecule serves as a pivotal building block, linking the benzamide core with the pyridyl and ester functionalities required for the final active pharmaceutical ingredient. Understanding the nuances of its synthesis is crucial for R&D directors who must validate the purity and impurity profiles of their supply sources. The patent highlights that while the final structure is well-defined, the journey to create it efficiently has historically been fraught with challenges related to reaction kinetics and catalyst deactivation. Therefore, the methods described herein represent a significant evolution in process chemistry, moving away from fragile protocols toward rugged, industrial-grade solutions that can withstand the rigors of multi-kilogram production runs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in this patent, the standard industrial approach for synthesizing this intermediate relied heavily on palladium-on-carbon (Pd/C) catalyzed hydrogenation. While effective on a small laboratory scale, this conventional method exhibited severe deficiencies when attempted at larger scales, specifically when substrate loads exceeded 100 grams. In these larger batches, the reaction would frequently proceed abnormally slowly or stop entirely, necessitating the filtration and replacement of the palladium catalyst multiple times to drive the conversion to completion. This iterative process of catalyst replacement is not only labor-intensive and time-consuming but also introduces significant safety hazards due to the handling of pyrophoric materials and the potential for exothermic events during repeated filtrations. Furthermore, the prolonged reaction times required to force conversion often led to the formation of complex impurity profiles, making the subsequent purification of the target compound difficult and reducing overall yield. These operational bottlenecks rendered the traditional method unsuitable for the continuous, high-volume manufacturing required by the modern pharmaceutical market.
The Novel Approach
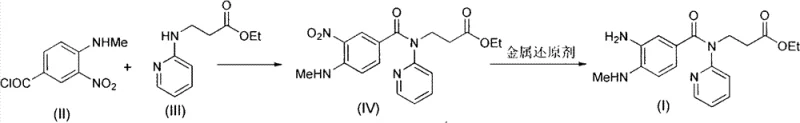
The patent introduces two distinct yet complementary strategies to circumvent these historical limitations, fundamentally altering the process landscape for this intermediate. The first method employs metallic reducing agents such as iron, zinc, or tin in an acetic acid and water system, bypassing the need for expensive noble metal catalysts entirely. The second, and perhaps more elegant solution, retains the use of Pd/C but introduces a catalytic amount of a primary or secondary amine promoter. This amine additive dramatically accelerates the hydrogenation rate, allowing the reaction to proceed rapidly at mild temperatures between 0 and 40 degrees Celsius without the need for catalyst replacement. Both methods share a critical process intensification feature: the ability to use the crude nitro-intermediate directly from the previous acylation step without purification. This telescoping of steps eliminates unit operations, reduces solvent consumption, and significantly lowers the cost of goods sold. The reaction scheme below illustrates the streamlined transformation from the starting materials to the final amino-intermediate.
Mechanistic Insights into Amine-Promoted Catalytic Hydrogenation
The introduction of primary or secondary amines as promoters in the catalytic hydrogenation step represents a sophisticated manipulation of surface chemistry on the palladium catalyst. In conventional hydrogenation, the adsorption of the nitro-aromatic substrate onto the catalyst surface can be inhibited by the presence of other functional groups or by the accumulation of reaction byproducts, leading to the observed stalling at larger scales. The amine promoter likely acts by modifying the electronic environment of the catalyst surface or by facilitating the desorption of the reduced amino-product, thereby preventing catalyst poisoning. Specifically, the patent notes that amines such as diisopropylamine or methyl ethanolamine, when used in molar ratios between 0.05:1 and 0.2:1 relative to the substrate, enable complete conversion within reasonable timeframes even at room temperature. This mechanistic enhancement ensures that the reaction kinetics remain favorable regardless of the batch size, effectively decoupling the reaction rate from the scale of operation. For R&D teams, this implies a much more predictable impurity profile, as the shorter reaction times and lower temperatures minimize thermal degradation and side reactions that typically plague prolonged hydrogenation processes.
Furthermore, the alternative metal reduction pathway offers a different mechanistic advantage rooted in classical organic synthesis. By utilizing iron or zinc powder in an acidic medium, the reduction proceeds via electron transfer mechanisms that are inherently robust and less sensitive to the trace impurities that might deactivate a heterogeneous catalyst. The use of acetic acid and water as the solvent system provides a green chemistry benefit, replacing volatile organic solvents often used in hydrogenation. This method ensures that the nitro group is selectively reduced to the amine while preserving the integrity of the ester and amide linkages within the molecule. The ability to perform this reduction on the crude reaction mixture from the acylation step suggests that the metallic reagents are tolerant of the residual acyl chloride or amine salts present in the pot. This tolerance is a critical quality attribute, as it simplifies the workflow and reduces the risk of introducing new contaminants during intermediate workups, ultimately leading to a cleaner final product that meets stringent pharmaceutical specifications.
How to Synthesize 3-[N-(2-pyridyl)-3-amino-4-methylamino benzamido]-ethyl propionate Efficiently
The synthesis of this critical anticoagulant intermediate requires careful attention to reaction conditions to maximize yield and purity while maintaining operational safety. The patent outlines a two-stage process beginning with the acylation of 3-[N-(2-pyridyl)]-ethyl propionate with 3-nitro-4-methylamino Benzoyl chloride, followed immediately by the reduction of the nitro group. The efficiency of this route lies in its telescoped nature, where the crude product of the first reaction is carried forward without isolation, significantly reducing processing time and solvent waste. Operators must ensure strict control over the stoichiometry of the base used in the acylation step, typically triethylamine, to neutralize the generated hydrochloric acid and drive the reaction to completion. Following the acylation, the choice of reduction method—whether metal-mediated or amine-promoted hydrogenation—should be dictated by the available infrastructure and specific purity requirements of the downstream API synthesis. Detailed standardized operating procedures for executing these steps safely and effectively are provided in the technical guide below.
- Condense 3-nitro-4-methylamino Benzoyl chloride with 3-[N-(2-pyridyl)]-ethyl propionate in the presence of a base like triethylamine to form the nitro-intermediate.
- Perform reduction using either metallic agents (Fe, Zn) in acetic acid/water or catalytic hydrogenation (Pd/C) with an amine promoter at low temperatures.
- Isolate the final amino-intermediate directly from the crude reaction mixture without purifying the preceding nitro-compound, followed by crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technologies described in this patent translates directly into enhanced supply security and optimized cost structures. The primary commercial advantage stems from the elimination of the catalyst replacement cycle, which was a major source of variability and delay in previous manufacturing campaigns. By ensuring that the reaction proceeds to completion in a single batch without intervention, manufacturers can guarantee shorter lead times and more reliable delivery schedules for this high-demand intermediate. Additionally, the ability to skip the purification of the nitro-intermediate represents a substantial reduction in processing costs, as it removes an entire unit operation involving filtration, drying, and quality control testing. This streamlining of the process flow not only reduces direct manufacturing expenses but also minimizes the physical footprint required for production, allowing for greater throughput within existing facilities. Consequently, suppliers utilizing this technology can offer more competitive pricing models while maintaining healthy margins, a critical factor in the cost-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The economic impact of this process innovation is driven by the significant simplification of the synthetic route. By removing the need to isolate and purify the nitro-intermediate, the process saves on solvent usage, energy consumption for drying, and labor hours associated with handling solid intermediates. Furthermore, the amine-promoted hydrogenation method extends the life of the palladium catalyst, eliminating the costly and hazardous procedure of filtering and reloading fresh catalyst multiple times per batch. These cumulative efficiencies result in a drastically simplified production cost model, allowing for substantial cost savings that can be passed down the supply chain or reinvested into quality assurance programs. The use of inexpensive metallic reducing agents in the alternative method further diversifies the cost options, providing flexibility in raw material sourcing to mitigate price volatility.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the new reaction conditions, which are far less prone to the stalling issues that plagued earlier methods. The ability to run reactions at ambient temperatures without the need for heating reduces the energy load on the facility and minimizes the risk of thermal runaway incidents, ensuring uninterrupted production cycles. Since the process is scalable beyond the 100g limit without modification, manufacturers can confidently plan for large-scale commercial production runs ranging from hundreds of kilograms to metric ton quantities without fearing a drop in yield or quality. This scalability ensures that sudden spikes in demand for Dabigatran Etexilate can be met without the long lead times typically associated with re-optimizing fragile chemical processes, thereby securing the continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the new methods offer distinct advantages in waste management and safety compliance. The metal reduction pathway utilizes aqueous acetic acid systems, which are easier to treat and dispose of compared to the complex organic waste streams generated by repeated catalyst filtrations. Moreover, the reduction in processing steps inherently lowers the total volume of hazardous waste generated per kilogram of product, aligning with increasingly strict global environmental regulations. The improved safety profile, characterized by the absence of high-temperature hydrogenation and reduced handling of pyrophoric catalysts, lowers the operational risk for manufacturing sites. This alignment with green chemistry principles and safety standards facilitates smoother regulatory audits and approvals, ensuring that the supply chain remains compliant with international pharmaceutical manufacturing guidelines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this Dabigatran intermediate, based on the specific embodiments and data provided in the patent literature. These insights are designed to clarify the operational benefits and chemical feasibility of the disclosed methods for industry stakeholders. Understanding these details is essential for making informed decisions about supplier qualification and process validation. The answers reflect the practical outcomes observed in the patent examples, highlighting the reproducibility and robustness of the technology.
Q: How does this patent solve the scalability issues of traditional Pd/C hydrogenation?
A: Traditional methods often stall at scales above 100g requiring catalyst replacement. This patent introduces amine promoters or metal reduction agents that allow the reaction to proceed to completion at ambient temperatures without changing the catalyst, ensuring consistent batch-to-batch reliability.
Q: What are the purity specifications achievable with these new methods?
A: The patent data indicates that using the amine-promoted hydrogenation method can achieve purities as high as 99%, while the metal reduction method yields purities around 95%, both suitable for subsequent coupling reactions in API synthesis.
Q: Does this process require purification of the nitro-intermediate?
A: No, a key advantage of this technology is the 'telescoped' nature of the synthesis. The crude nitro-intermediate (IV) is used directly in the reduction step, eliminating isolation, drying, and purification costs associated with that specific stage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-[N-(2-pyridyl)-3-amino-4-methylamino benzamido]-ethyl propionate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex anticoagulants relies on a foundation of high-quality, reliably sourced intermediates. Our technical team has extensively analyzed the pathways described in patent CN103539730A and possesses the expertise to implement these advanced reduction strategies at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-[N-(2-pyridyl)-3-amino-4-methylamino benzamido]-ethyl propionate meets the exacting standards required for API synthesis. By leveraging our capabilities in both metal-mediated and amine-promoted reduction chemistries, we offer a flexible and resilient supply solution tailored to the dynamic needs of the global pharmaceutical market.
We invite procurement leaders and R&D directors to engage with us to explore how our optimized manufacturing processes can support your project goals. Our team is prepared to provide a Customized Cost-Saving Analysis that demonstrates the specific economic benefits of switching to our supply chain for this critical intermediate. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your upcoming production campaigns. Partnering with us ensures not just a transaction, but a strategic alliance focused on quality, efficiency, and the successful delivery of life-saving medications to patients worldwide.
