Advanced Synthesis of Chiral Spiro Pyrrolidine Oxindole Intermediates for Pharmaceutical Manufacturing Scale-Up
The recently granted Chinese patent CN111171037B introduces a groundbreaking synthetic methodology for chiral spiro [3,2'] pyrrolidine oxindole skeleton compounds, representing a significant advancement in the field of pharmaceutical intermediates with demonstrated anti-inflammatory, antibacterial, and anti-tumor biological activities. This innovative approach addresses longstanding challenges in the construction of complex chiral architectures that have previously hindered the commercial viability of these pharmacologically valuable molecules through strategic substrate design and optimized catalytic conditions enabling production of high-purity intermediates with exceptional enantioselectivity exceeding 97% ee in optimized cases. Unlike traditional approaches requiring multiple protection/deprotection steps with low yields around 50%, this novel process achieves remarkable efficiency while maintaining strict stereochemical control essential for pharmaceutical applications through a streamlined three-step synthesis route compatible with industrial-scale manufacturing parameters. The methodology's ability to utilize readily available starting materials under mild reaction conditions positions it as a transformative solution for pharmaceutical companies seeking reliable sources of complex chiral intermediates without compromising on quality or consistency requirements mandated by regulatory authorities worldwide.
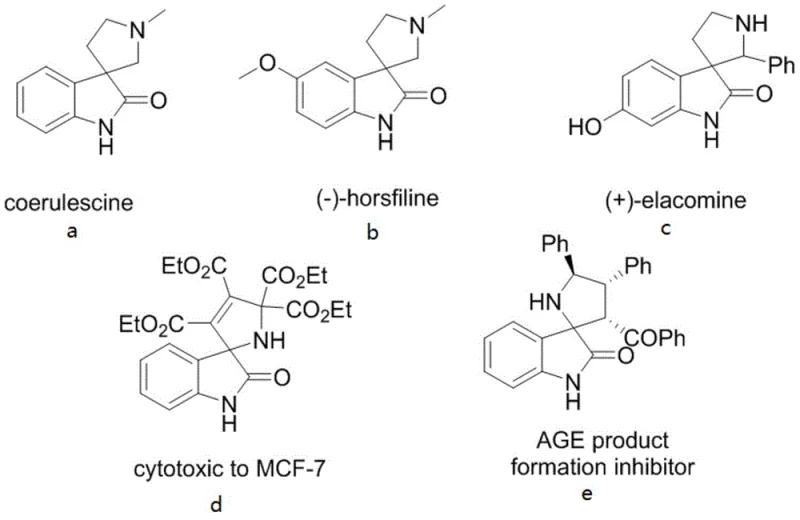
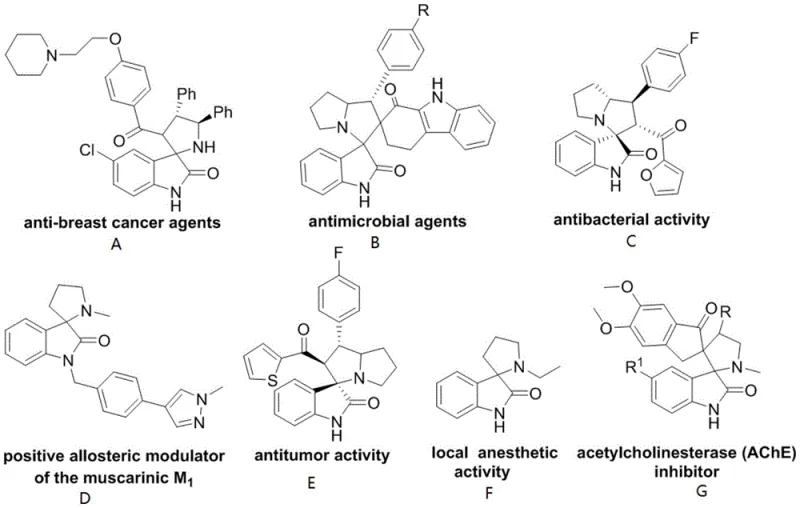
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing spiro [3,2'] pyrrolidine oxindole skeletons have been plagued by significant limitations that impede their commercial adoption in pharmaceutical manufacturing operations requiring consistent quality at scale. Method A (the 1,3-dipolar cycloaddition reaction) imposes severe constraints on both dipole components requiring specific substituents like aryl or trifluoromethyl groups that introduce unnecessary complexity into final product structures while limiting structural diversity needed for drug discovery programs. Method B (nucleophilic addition of isatin imines) suffers from extended synthetic routes with lower overall yields typically below 60% due to multiple protection/deprotection steps required when using bulky trityl groups that negatively impact physiological activity profiles essential for therapeutic applications. Method C (reactions between 3-aminooxindoles and amphiphilic electrophiles) faces challenges with selective substitution control due to high reactivity leading to side products requiring extensive purification processes that reduce overall yield below acceptable commercial thresholds while complicating waste management procedures required by environmental regulations.
![Reaction scheme illustrating Method A showing limitations in constructing spiro [3,2'] pyrrole epoxidized indole skeleton including Ma's work demonstrating yield up to 99%, dr up to 20:1, ee up to >99% but requiring specific substituent constraints](/insights/img/chiral-spiro-pyrrolidine-oxindole-cinchona-catalyst-pharma-cost-reduction-20260228164540-03.webp)
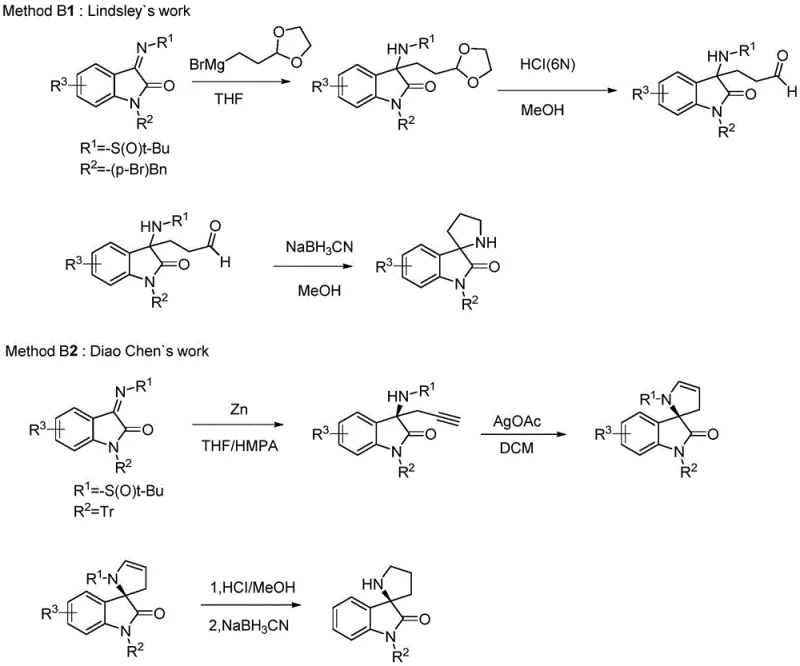
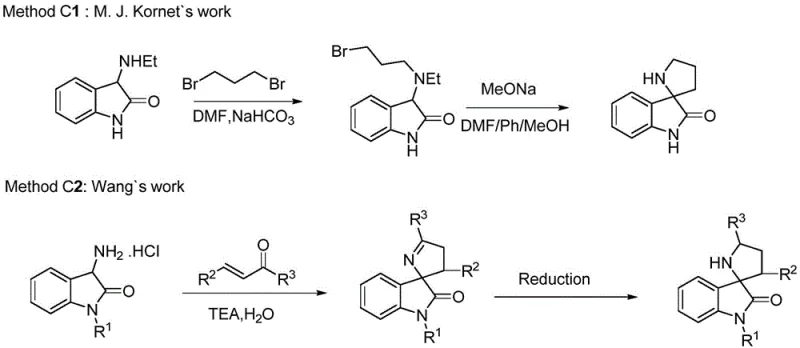
The Novel Approach
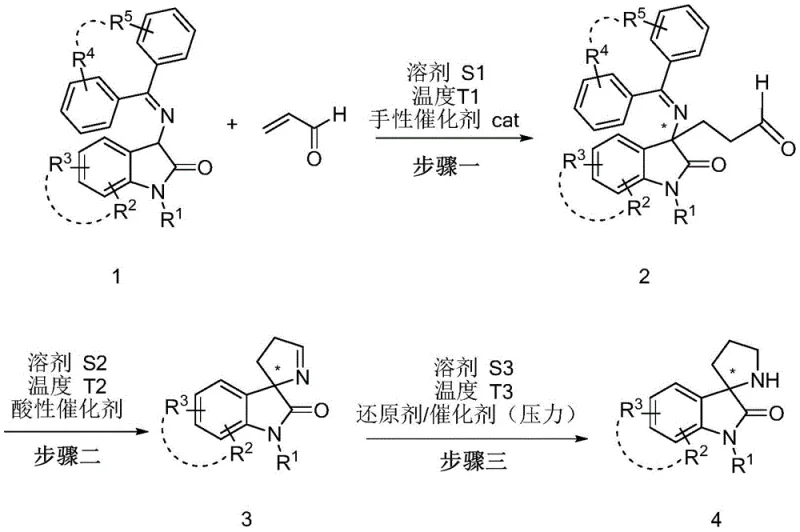
The patented methodology presented in CN111171037B introduces a revolutionary three-step synthesis that overcomes fundamental limitations through strategic innovation in substrate design using diaryl ketone protected 3-ketiminooxindole derivatives as starting materials which provide optimal balance between reactivity and steric hindrance necessary for high enantioselectivity exceeding 97% ee when using cat5 catalyst under mild conditions at room temperature. This critical substrate design choice enables utilization of readily available acrolein under optimized catalytic conditions achieving yields above 70% across multiple substrate variants while maintaining exceptional stereochemical control essential for pharmaceutical applications requiring strict enantiomeric purity specifications. The second step cleverly combines deprotection and ring closure in a single operation under mild acidic conditions (pH 1-6), eliminating intermediate isolation requirements while allowing efficient recycling of protecting groups which significantly reduces raw material costs without requiring complex recovery systems or specialized equipment modifications.
Mechanistic Insights into Cinchona Alkaloid-Catalyzed Asymmetric Michael Addition
The core innovation lies in the sophisticated mechanism where cinchona alkaloid derivatives operate through dual activation - their tertiary amine moiety deprotonates the acidic α-position of oxindole substrates while simultaneously coordinating with acrolein via hydrogen bonding interactions creating a well-defined chiral environment directing acrolein approach with exceptional stereocontrol up to 97.8% ee as demonstrated in Example 9 using cat5 catalyst under optimized conditions at room temperature. This bifunctional activation mechanism is enhanced by steric bulk from diaryl ketone protecting groups which create additional spatial constraints favoring one enantiomeric pathway over others while preventing unwanted side reactions common in traditional approaches requiring harsher reaction parameters that compromise product quality consistency at scale.
Impurity profile management is achieved through multiple synergistic mechanisms where carefully selected diaryl ketone protecting groups moderate substrate reactivity preventing both under-reaction and decomposition pathways while maintaining compatibility with industrial-scale operations through simple deprotection protocols allowing recycling without complex separation requirements. The one-pot deprotection/ring closure sequence eliminates potential impurities from intermediate isolation handling while mild acidic conditions prevent racemization of sensitive chiral centers essential for maintaining therapeutic efficacy requirements mandated by regulatory authorities worldwide.
How to Synthesize Chiral Spiro Pyrrolidine Oxindole Efficiently
This patented methodology represents a significant advancement by addressing critical challenges limiting commercial production through strategic innovations including optimized substrate design enabling high-yielding reactions under mild conditions compatible with standard pharmaceutical manufacturing facilities without specialized equipment requirements or hazardous reagents typically associated with traditional approaches requiring cryogenic temperatures or heavy metal catalysts requiring complex removal procedures adding both cost and environmental burden.
- Perform asymmetric Michael addition using diaryl ketone protected 3-ketiminooxindole derivative (formula 1) with acrolein under mild conditions (0-25°C) using cinchona alkaloid catalyst (15-25% loading) in dichloromethane or ethyl acetate solvent system
- Execute one-pot deprotection/ring closure under acidic conditions (pH 1-6) at room temperature using citric acid or hydrochloric acid in THF or methanol solvent system
- Complete reduction reaction via catalytic hydrogenation (Raney Ni catalyst at 3-6 KPa H₂ pressure) or chemical reduction (sodium borohydride) at room temperature in methanol solvent
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthetic route delivers substantial value by addressing critical pain points associated with traditional manufacturing methods through multiple strategic advantages translating directly into operational benefits including reduced raw material complexity while maintaining high product quality standards required by pharmaceutical clients through elimination of unnecessary protection/deprotection steps creating quality variability common in more complex synthetic routes requiring extensive purification procedures increasing both cost and lead time variability impacting supply chain reliability metrics essential for just-in-time manufacturing operations.
- Cost Reduction in Manufacturing: Elimination of multiple protection/deprotection steps significantly reduces raw material costs while improving overall process efficiency through reduced cycle times lowering facility utilization costs per batch without requiring capital investment in specialized equipment typically needed for traditional approaches involving cryogenic temperatures or heavy metal catalysts requiring complex removal procedures adding both cost and environmental burden through additional processing steps increasing waste generation metrics.
- Enhanced Supply Chain Reliability: Reliance on readily available starting materials with multiple potential suppliers reduces vulnerability to supply chain disruptions while ensuring consistent quality through well-defined reaction parameters minimizing batch-to-batch variability common in more complex synthetic routes; robustness across different solvent systems provides flexibility adapting to regional raw material availability without compromising product quality or yield creating resilience against localized supply chain challenges impacting production continuity essential for meeting regulatory timelines required by global pharmaceutical manufacturers.
- Scalability and Environmental Compliance: Process demonstrates excellent scalability from laboratory to commercial production volumes without significant re-engineering due to compatibility with standard manufacturing infrastructure worldwide; mild reaction conditions avoiding heavy metal catalysts reduce environmental impact simplifying waste treatment compared to conventional methods; one-pot deprotection/ring closure step minimizes solvent usage improving process mass intensity metrics increasingly important for sustainable manufacturing practices meeting growing regulatory pressure toward greener chemistry principles without compromising economic viability required by commercial operations.
Frequently Asked Questions (FAQ)
The following questions address common concerns raised by procurement teams evaluating this innovative manufacturing approach based on detailed technical analysis reflecting specific advantages documented in patent CN111171037B providing insight into how this methodology addresses critical challenges through scientifically validated mechanisms rather than theoretical assumptions focusing on practical implementation considerations directly impacting procurement decisions including quality consistency metrics essential for informed sourcing strategies in today's competitive pharmaceutical marketplace where supply chain reliability has become paramount following recent global disruptions.
Q: How does this methodology achieve higher enantioselectivity compared to conventional approaches?
A: The patented process utilizes specifically designed diaryl ketone protected substrates that provide optimal steric hindrance combined with cinchona alkaloid catalysts (cat5 achieving up to 97.8% ee) operating under mild conditions (0-25°C), creating a precise chiral environment that conventional methods lack due to their requirement for harsher reaction parameters.
Q: What are the key advantages regarding raw material availability?
A: The synthesis employs readily available starting materials including acrolein, common solvents (dichloromethane, methanol), and commercially accessible cinchona alkaloid derivatives as catalysts, eliminating dependency on specialized reagents required by traditional methods which often involve rare or expensive components.
Q: How does this process support scalable manufacturing?
A: The methodology demonstrates excellent scalability from laboratory (gram scale) to commercial production (up to 100 MT/annual) through its compatibility with standard equipment, mild reaction conditions (room temperature operations), one-pot processing steps reducing intermediate handling, and flexible reduction options adaptable to existing facility capabilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiro [3,2'] Pyrrolidine Oxindole Skeleton Compound Supplier
Our company possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation ensuring consistent product quality meeting global regulatory requirements across all major markets including FDA, EMA, PMDA, and NMPA standards; our technical team has successfully implemented similar asymmetric catalytic processes across multiple product lines demonstrating consistent ability to translate laboratory-scale innovations into reliable commercial manufacturing operations addressing both technical challenges and commercial requirements faced by global pharmaceutical companies seeking stable sources for complex chiral intermediates critical to their drug development pipelines.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team who can provide detailed COA data packages containing comprehensive analytical results including HPLC chromatograms showing purity profiles exceeding industry standards along with route feasibility assessments tailored specifically to your production needs helping you optimize both quality consistency metrics required by regulatory authorities and cost-effectiveness parameters essential for competitive advantage in today's challenging market environment where supply chain resilience has become equally important as product quality specifications.
