Advanced Manufacturing of 3,4-Dibenzoyl-1,2,5-Oxadiazole via Efficient Deoxygenation
The chemical landscape for heterocyclic intermediates is constantly evolving, driven by the need for greener processes and higher efficiency. A pivotal advancement in this domain is documented in Chinese Patent CN114105900B, which details a novel preparation method for 3,4-dibenzoyl-1,2,5-oxadiazole. This compound serves as a critical building block in the synthesis of fluorescent labeling dyes and specialized esters, necessitating a robust and scalable supply chain. The patent introduces a transformative approach that replaces conventional heavy metal reductants with triethyl phosphite, achieving a remarkable yield enhancement while drastically simplifying the post-reaction workup. This technological leap addresses long-standing pain points in fine chemical manufacturing, specifically regarding waste generation and energy consumption.
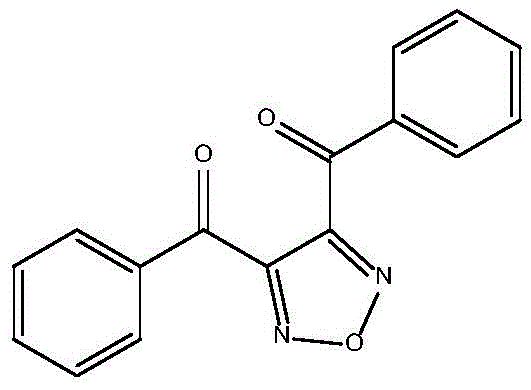
For R&D directors and process chemists, understanding the structural integrity and synthetic accessibility of such intermediates is paramount. The molecule features a stable 1,2,5-oxadiazole core substituted with two benzoyl groups, a configuration that imparts specific physical properties suitable for high-performance applications. The ability to synthesize this scaffold efficiently without compromising purity is a key differentiator for suppliers aiming to serve the pharmaceutical and specialty chemical sectors. The following analysis dissects the technical merits of this patented route, highlighting its potential to redefine cost structures and supply reliability for global buyers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dibenzoyl-1,2,5-oxadiazole relied heavily on reduction protocols utilizing zinc powder as the primary reductant. As illustrated in the reaction scheme below, this traditional pathway typically employed acetic anhydride and acetic acid in an acetonitrile solvent system, requiring extended reaction times of up to 24 hours at relatively low temperatures of 20-30°C. Despite the widespread use of this method, it suffered from inherent inefficiencies, including incomplete reduction and the generation of substantial quantities of zinc-containing waste streams. The reliance on stoichiometric amounts of zinc not only increased raw material costs but also introduced significant challenges in wastewater treatment and environmental compliance.
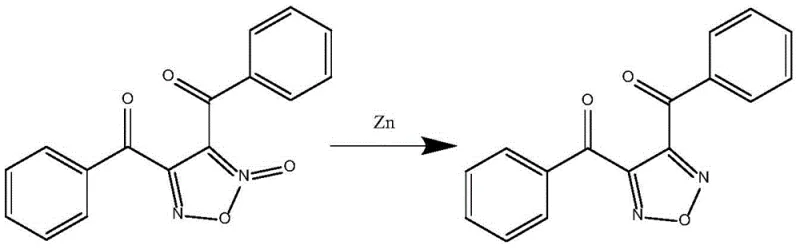
Furthermore, the product quality obtained from the zinc-mediated process was often suboptimal, with reported yields capping at approximately 65%. The presence of residual metal impurities and side products necessitated rigorous and costly purification steps, which eroded profit margins and extended lead times. For procurement managers, these factors translated into higher unit costs and potential supply disruptions due to the complexity of waste disposal regulations. The inability of the conventional method to consistently deliver high-purity material at scale made it a bottleneck for downstream applications requiring stringent quality specifications, such as electronic chemicals or advanced optical materials.
The Novel Approach
In stark contrast, the methodology disclosed in CN114105900B leverages triethyl phosphite as a highly efficient oxygen acceptor to drive the deoxygenation of the oxadiazole-N-oxide precursor. This innovative route operates in toluene, a solvent favored for its ease of recovery and industrial availability, at elevated temperatures ranging from 40-80°C. The reaction kinetics are significantly improved, allowing the transformation to reach completion within 9-14 hours, effectively halving the processing time compared to the zinc-based legacy method. The mechanistic elegance of using a phosphorus-based reductant ensures a cleaner reaction profile, minimizing the formation of intractable byproducts and facilitating a straightforward isolation procedure.
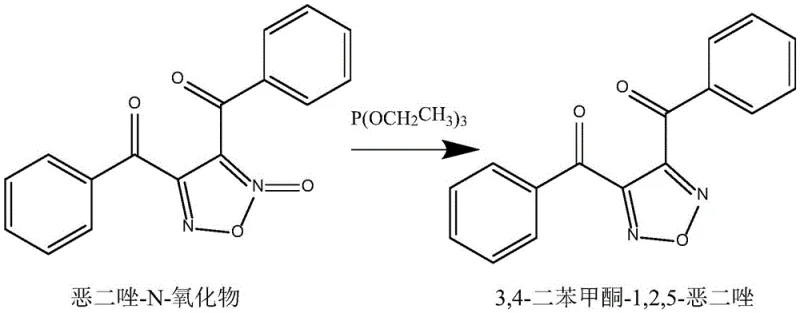
The impact of this novel approach on overall process efficiency is profound, with experimental data demonstrating yields soaring to 82.3%. By eliminating the need for heavy metals, the process inherently reduces the environmental footprint, aligning with modern green chemistry principles. For supply chain stakeholders, this translates to a more resilient manufacturing process that is less susceptible to regulatory hurdles associated with heavy metal discharge. The switch to triethyl phosphite also optimizes the atom economy of the reaction, ensuring that a greater proportion of input materials are converted into valuable product, thereby driving down the effective cost of goods sold (COGS) without sacrificing quality.
Mechanistic Insights into Triethyl Phosphite-Mediated Deoxygenation
The core of this technological advancement lies in the specific interaction between the N-oxide functionality and the phosphorus center of the triethyl phosphite. Unlike zinc, which acts as a heterogeneous electron donor often leading to surface passivation and inconsistent reactivity, triethyl phosphite functions as a homogeneous nucleophile that attacks the oxygen atom on the nitrogen. This results in the formation of a transient phosphine oxide species and the simultaneous restoration of the aromatic oxadiazole system. The homogeneity of the reaction mixture ensures uniform heat and mass transfer, which is critical for maintaining consistent product quality across large batch sizes. This mechanistic clarity allows process engineers to precisely control reaction parameters, minimizing the risk of thermal runaways or incomplete conversions.
From an impurity control perspective, the absence of zinc ions eliminates the formation of coordination complexes that often trap organic impurities, making them difficult to remove during crystallization. The primary byproduct, triethyl phosphate, is generally more soluble in aqueous washes or can be easily separated during the solvent recovery phase, especially when treated with dilute hydrochloric acid as described in the patent examples. This simplification of the impurity profile is a major advantage for R&D teams tasked with validating analytical methods and establishing stability-indicating assays. The high purity levels achieved (up to 98.4% in optimized examples) reduce the burden on downstream purification units, allowing for a more streamlined production flow that enhances overall throughput.
How to Synthesize 3,4-Dibenzoyl-1,2,5-Oxadiazole Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize the benefits observed in the patent literature. The process begins with the dissolution of the oxadiazole-N-oxide starting material in toluene, followed by the addition of triethyl phosphite in a molar ratio optimized between 1:1 and 1:1.6. Maintaining the reaction temperature within the 40-80°C window is crucial; too low a temperature slows the kinetics, while excessive heat may promote degradation. The subsequent workup involves a strategic acid wash to hydrolyze any remaining phosphite esters, ensuring the final product meets rigorous purity standards. For detailed operational parameters and safety considerations, refer to the standardized guide below.
- Charge oxadiazole-N-oxide and triethyl phosphite into a reactor containing toluene solvent under stirring.
- Heat the reaction mixture to a temperature range of 40-80°C (optimally 60°C) and maintain stirring for 9-14 hours to ensure complete reduction.
- Upon completion, treat the reaction mixture with dilute hydrochloric acid to remove ester byproducts, separate the organic layer, wash, and recover the solvent to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from zinc-based reduction to triethyl phosphite chemistry offers compelling economic and logistical advantages that extend far beyond the laboratory bench. For procurement professionals, the elimination of zinc powder removes a volatile cost component subject to fluctuating metal markets and stringent transport regulations. Moreover, the significant reduction in reaction time directly correlates to increased asset utilization, allowing manufacturing facilities to produce more batches per annum without capital expansion. This efficiency gain is a powerful lever for negotiating better pricing structures with end-users, as the lowered operational expenditure can be passed down the value chain while maintaining healthy margins.
- Cost Reduction in Manufacturing: The adoption of triethyl phosphite eliminates the need for expensive heavy metal removal technologies, such as scavenger resins or complex filtration systems, which are mandatory when using zinc. This simplification of the downstream processing train results in substantial savings in both consumables and labor hours. Additionally, the higher yield means that less starting material is required to produce the same amount of finished goods, effectively lowering the raw material cost per kilogram. The use of toluene, a commodity solvent with a well-established recovery infrastructure, further enhances the economic viability of the process compared to acetonitrile.
- Enhanced Supply Chain Reliability: By removing heavy metals from the process, manufacturers face fewer regulatory bottlenecks related to environmental discharge permits, ensuring uninterrupted production schedules. The reagents used, specifically triethyl phosphite and toluene, are widely available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base enhances resilience against market shocks, ensuring that delivery commitments to pharmaceutical and agrochemical clients are met consistently. The robustness of the reaction conditions also reduces the likelihood of batch failures, contributing to a more predictable and reliable supply stream.
- Scalability and Environmental Compliance: The homogeneous nature of the reaction facilitates seamless scale-up from pilot plant to commercial tonnage, as heat transfer limitations are minimized compared to heterogeneous zinc reductions. The reduction in hazardous waste generation aligns with increasingly strict global environmental standards, reducing the liability and cost associated with waste disposal. This eco-friendly profile is increasingly becoming a prerequisite for inclusion in the supply chains of major multinational corporations committed to sustainability goals. Consequently, suppliers adopting this technology position themselves as preferred partners for long-term contracts focused on green chemistry initiatives.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this synthesis route, we have compiled answers to common inquiries regarding its implementation and performance. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation. Understanding these nuances is essential for assessing the feasibility of integrating this intermediate into your existing manufacturing workflows or sourcing strategies.
Q: How does the triethyl phosphite method compare to traditional zinc reduction?
A: The triethyl phosphite method significantly outperforms traditional zinc reduction by eliminating heavy metal waste, reducing reaction time from 24 hours to approximately 12 hours, and increasing yields from roughly 65% to over 82%.
Q: What are the primary impurities controlled in this new synthesis route?
A: By avoiding zinc powder and acetic anhydride, this route prevents the formation of difficult-to-remove zinc salts and complex acetylated byproducts, resulting in a cleaner crude profile that simplifies downstream purification.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes common solvents like toluene and operates at moderate temperatures (40-80°C), making it highly scalable with lower energy consumption and simplified waste treatment compared to heavy metal-based alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dibenzoyl-1,2,5-Oxadiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex intermediates like 3,4-dibenzoyl-1,2,5-oxadiazole requires more than just a patent; it demands engineering excellence and unwavering quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the triethyl phosphite reduction are fully realized in practice. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for fluorescent dye and pharmaceutical applications. Our commitment to process optimization ensures that we deliver not just a chemical, but a solution that enhances your downstream efficiency.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your strategic goals. Let us be your partner in navigating the complexities of fine chemical sourcing with confidence and precision.
