Scalable Synthesis of Aza-bicyclo[3.3.0]octane Derivatives for Advanced Pharmaceutical Applications
Scalable Synthesis of Aza-bicyclo[3.3.0]octane Derivatives for Advanced Pharmaceutical Applications
The pharmaceutical industry continuously demands robust, scalable, and cost-effective synthetic routes for complex heterocyclic scaffolds, particularly those serving as core structures for potent therapeutic agents. Patent CN102718695B introduces a groundbreaking methodology for the synthesis of aza-bicyclo[3.3.0]octane derivatives, a critical structural motif found in emerging classes of serine protease dipeptidyl peptidase-4 (DDP-4) inhibitors used for diabetes treatment. This technology represents a significant leap forward in process chemistry, transitioning from hazardous, high-energy protocols to a mild, high-yield sequence that begins with the inexpensive and commercially abundant 1,2,3,6-tetrahydrophthalimide. By systematically addressing the limitations of previous art through innovative protective group strategies and optimized oxidation-cyclization sequences, this patent provides a reliable foundation for the commercial scale-up of complex pharmaceutical intermediates.
For R&D directors and process chemists, the value of this innovation lies in its operational simplicity and exceptional purity profiles. The route avoids the use of exotic catalysts or extreme conditions, instead relying on standard unit operations such as liquid-liquid extraction and crystallization which are easily validated under GMP guidelines. The strategic implementation of nitrogen protection not only enhances the yield of the critical reduction step but also ensures that the final cyclodecarboxylation proceeds with minimal byproduct formation. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for delivering high-purity aza-bicyclo[3.3.0]octane derivatives that meet the stringent specifications required by global drug developers.
![General reaction route from 1,2,3,6-tetrahydrophthalimide to aza-bicyclo[3.3.0]octane derivatives](/insights/img/aza-bicyclo-octane-synthesis-pharma-supplier-20260305052103-02.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the construction of the aza-bicyclo[3.3.0]octane core was fraught with significant technical and safety challenges that hindered industrial adoption. One established method, depicted in equation (a) of the prior art, involved the reaction of dimethyl esters with secondary amines in sealed tubes at temperatures as high as 190°C for extended periods of 16 hours. Such harsh thermal conditions are not only energy-intensive but also pose severe safety risks regarding pressure containment and potential decomposition of sensitive functional groups. Furthermore, alternative approaches utilizing the Pauson-Khand reaction, as shown in equation (b), relied heavily on dicobalt octacarbonyl Co2(CO)8 as a catalyst. This metal carbonyl complex is highly toxic, volatile, and unstable, creating substantial occupational health hazards and complicating waste disposal protocols in a manufacturing environment.
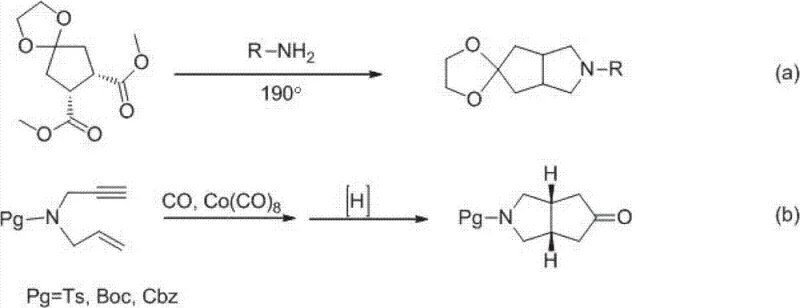
The reliance on these legacy methods resulted in processes that were difficult to control, exhibited variable yields, and generated complex impurity profiles that were costly to remove. For procurement managers, the implications of these technical deficits were clear: higher raw material costs due to low efficiency, increased expenditure on safety infrastructure, and unpredictable lead times caused by batch failures. The inability to safely scale these reactions meant that supply chains for DDP-4 inhibitor intermediates remained fragile and dependent on small-batch specialty synthesis rather than efficient bulk manufacturing. Consequently, there was an urgent industry need for a synthetic pathway that could deliver the target bicyclic structure under mild conditions without compromising on yield or safety.
The Novel Approach
The methodology disclosed in CN102718695B fundamentally reimagines the synthetic trajectory by employing a linear, five-step sequence that prioritizes safety and scalability. Starting from 1,2,3,6-tetrahydrophthalimide, the process utilizes a clever protection-reduction-deprotection strategy that circumvents the need for high-pressure or high-temperature reactors. By initially installing a robust protecting group on the nitrogen atom, the subsequent reduction of the imide carbonyls to methylene groups using lithium aluminum hydride proceeds with exceptional efficiency and selectivity. This approach effectively decouples the ring-forming events from the harsh conditions previously deemed necessary, allowing each transformation to be optimized independently for maximum yield.
Moreover, the novel route replaces toxic cobalt catalysts with benign oxidants like potassium permanganate for the oxidative cleavage of the double bond, followed by a thermal cyclodecarboxylation in acetic anhydride. This shift not only eliminates the handling of hazardous heavy metals but also simplifies the downstream purification process, as the byproducts are water-soluble or easily separable organic acids. For supply chain heads, this translates to cost reduction in pharmaceutical intermediate manufacturing through reduced waste treatment costs and streamlined logistics. The ability to perform these reactions at or near ambient pressure and moderate temperatures significantly lowers the barrier to entry for contract manufacturing organizations, ensuring a more resilient and continuous supply of this vital scaffold.
Mechanistic Insights into Protective Group Mediated Cyclization
The core innovation of this synthesis lies in the meticulous management of the nitrogen atom's reactivity throughout the multi-step sequence. The process begins with the N-alkylation or N-silylation of 1,2,3,6-tetrahydrophthalimide (Compound V) to form Compound IV. This protection step is crucial because it prevents the nitrogen from interfering with the subsequent strong reducing agent, LiAlH4. Without this protection, the free imide might undergo incomplete reduction or polymerization. The patent details various protecting groups (PG) such as trityl, tert-butyl, MOM, and benzyl, demonstrating the versatility of the method. Following reduction, the resulting amine (Compound III) is highly nucleophilic; however, direct oxidation of the alkene in this state could lead to N-oxide formation or other side reactions. Therefore, the protocol involves a deprotection step followed immediately by re-protection with a carbamate or amide group (Compound II).
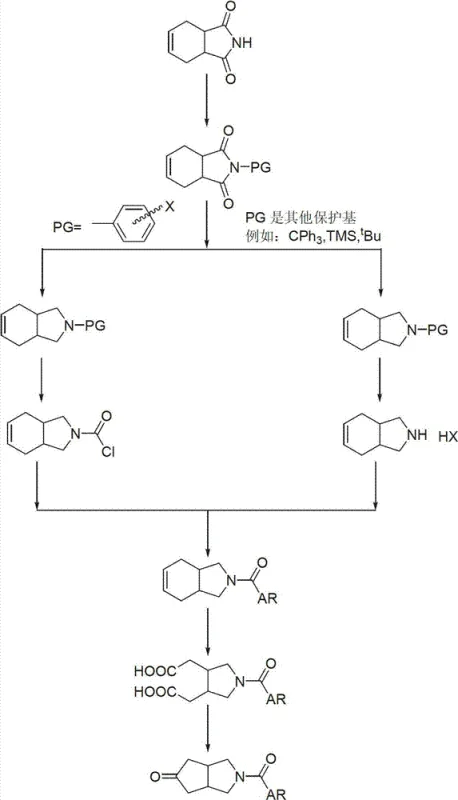
This second protecting group serves a dual purpose: it stabilizes the amine during the vigorous oxidative conditions required to cleave the cyclohexene double bond into a dicarboxylic acid, and it acts as an internal nucleophile trap during the final cyclization. When the dicarboxylic acid intermediate is heated in acetic anhydride, it undergoes dehydration to form a cyclic anhydride, which is then attacked by the adjacent nitrogen lone pair. This intramolecular nucleophilic attack, followed by decarboxylation, closes the second five-membered ring to form the target aza-bicyclo[3.3.0]octane system (Compound I). The choice of the A-R group (where A is O or N) on the nitrogen allows for the direct installation of diverse functional handles, enabling rapid diversification of the scaffold for SAR studies. This mechanistic elegance ensures that impurities are minimized at every stage, resulting in a final product that requires minimal chromatographic purification, a key factor for commercial viability.
How to Synthesize Aza-bicyclo[3.3.0]octane Derivatives Efficiently
The execution of this synthetic route requires precise control over stoichiometry and reaction parameters to maximize the benefits outlined in the patent. While the general pathway is robust, specific attention must be paid to the quenching of the lithium aluminum hydride reduction and the pH control during the oxidation phase to prevent hydrolysis of the carbamate protecting group. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring reproducibility and safety in a pilot or production plant setting.
- Protect 1,2,3,6-tetrahydrophthalimide (V) with a suitable group (PG) like benzyl or trityl under alkaline conditions to form compound IV.
- Reduce the imide carbonyls of compound IV to methylene groups using LiAlH4 in an aprotic solvent like THF to generate compound III.
- Remove the initial protecting group and install a carbamate or amide group to form compound II, ensuring stability for subsequent oxidation.
- Oxidize the double bond in compound II using KMnO4 to form a dicarboxylic acid intermediate.
- Perform cyclodecarboxylation in acetic anhydride at 120°C to yield the final aza-bicyclo[3.3.0]octane derivative (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this patented synthesis method offers tangible economic and operational benefits that extend far beyond simple yield improvements. By shifting away from the legacy Pauson-Khand chemistry, manufacturers can eliminate the substantial costs associated with sourcing, handling, and disposing of toxic cobalt catalysts. This transition not only reduces the direct material cost but also mitigates the regulatory burden and environmental compliance costs that are increasingly stringent in the global chemical sector. The use of commodity chemicals like potassium permanganate and acetic anhydride ensures that raw material supply is stable and不受 geopolitical fluctuations that often affect specialty metal catalysts.
- Cost Reduction in Manufacturing: The elimination of high-pressure sealed tube reactions (190°C) drastically reduces energy consumption and capital expenditure on specialized reactor equipment. Standard glass-lined or stainless steel reactors capable of atmospheric reflux are sufficient for this process, lowering the barrier for contract manufacturing partners. Furthermore, the high yields reported in the patent examples, particularly in the reduction and cyclization steps, mean that less raw material is wasted per kilogram of final product, directly improving the gross margin for API manufacturers.
- Enhanced Supply Chain Reliability: The reliance on 1,2,3,6-tetrahydrophthalimide as a starting material leverages a well-established supply chain for bulk petrochemical derivatives. Unlike specialized precursors that may have single-source suppliers, this feedstock is widely available from multiple vendors globally. This diversity in sourcing options reduces the risk of supply disruptions and provides procurement teams with greater leverage in price negotiations. Additionally, the mild reaction conditions reduce the likelihood of batch failures due to thermal runaways, ensuring consistent delivery schedules to downstream drug product manufacturers.
- Scalability and Environmental Compliance: The process generates aqueous waste streams containing manganese salts and acetates, which are significantly easier and cheaper to treat than heavy metal waste containing cobalt. This aligns with modern green chemistry principles and simplifies the permitting process for new manufacturing facilities. The scalability of the oxidation and cyclization steps has been demonstrated in the patent through gram-scale examples that translate linearly to kilogram and ton scales without the need for complex process intensification technologies, facilitating rapid commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within patent CN102718695B, providing clarity for technical teams evaluating this route for their pipeline.
Q: Why is the new synthesis method safer than prior art Pauson-Khand reactions?
A: The patented method eliminates the use of toxic cobalt carbonyl Co2(CO)8 catalysts required in traditional Pauson-Khand reactions, significantly reducing hazardous waste and safety risks in large-scale production.
Q: What are the key advantages of the protective group strategy in this route?
A: The strategic use of protecting groups (PG) allows for high-yield reduction of the imide to the amine without side reactions, and facilitates easy removal and replacement to enable the final oxidative cyclization step efficiently.
Q: Is this process suitable for industrial scale-up of DDP-4 inhibitor intermediates?
A: Yes, the process utilizes mild reaction conditions (e.g., room temperature oxidation, standard reflux reduction) and readily available raw materials like 1,2,3,6-tetrahydrophthalimide, making it highly scalable for commercial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aza-bicyclo[3.3.0]octane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in accelerating drug development timelines. Our technical team has thoroughly analyzed the methodology described in CN102718695B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. We are equipped with rigorous QC labs and stringent purity specifications to ensure that every batch of aza-bicyclo[3.3.0]octane derivative meets the exacting standards of the pharmaceutical industry. Our commitment to process excellence means we can offer this complex scaffold with the reliability and consistency that global supply chains demand.
We invite R&D and procurement leaders to collaborate with us to optimize this route for your specific API needs. By leveraging our expertise in protective group chemistry and oxidative cyclization, we can provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your next breakthrough therapy.
