Scalable Asymmetric Synthesis of Triptonide and Triptolide via Metal-Catalyzed Hydrogen Atom Transfer
Scalable Asymmetric Synthesis of Triptonide and Triptolide via Metal-Catalyzed Hydrogen Atom Transfer
The pharmaceutical industry continuously seeks robust and scalable pathways for complex bioactive molecules, particularly those derived from traditional medicinal plants with potent therapeutic profiles. Patent CN113292630A discloses a groundbreaking method for the asymmetric synthesis of Triptonide and Triptolide, two critical compounds known for their anti-inflammatory, immunosuppressive, and anticancer activities. This technology leverages a sophisticated strategy starting from (-)-Taniguchi lactone, employing alkylation reactions, transition metal-catalyzed hydrogen atom transfer (HAT), and aldol-type ring closures to efficiently construct the intricate pentacyclic diterpenoid skeleton. By shifting away from reliance on low-content natural extraction, this synthetic methodology offers a reliable pharmaceutical intermediates supplier pathway that ensures high purity and structural diversity for drug development programs.
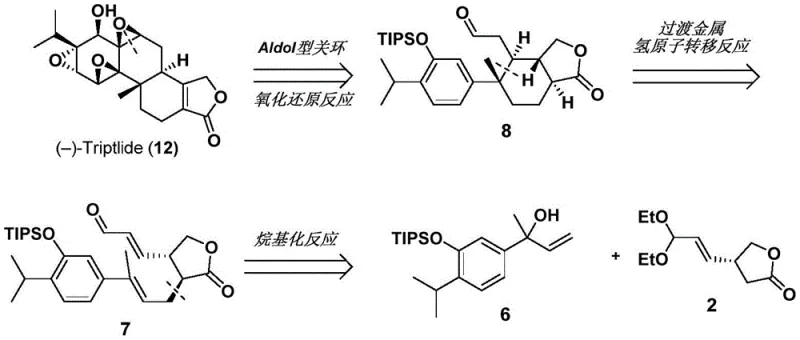
The strategic value of this patent lies in its ability to streamline the total synthesis of these high-value targets. Traditional isolation from Tripterygium wilfordii is plagued by low yields and significant environmental impact, while previous semi-synthetic routes often suffered from excessive step counts and poor stereocontrol. The disclosed invention addresses these bottlenecks by introducing a convergent synthesis where the core carbon framework is established early through a key alkylation step, followed by a biomimetic cyclization. This approach not only enhances the overall yield but also provides a versatile platform for generating analogues, thereby facilitating structure-activity relationship (SAR) studies essential for optimizing therapeutic efficacy and reducing toxicity in next-generation drug candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of Triptonide and Triptolide has been constrained by the limitations of natural extraction and early semi-synthetic efforts. Extracting these compounds directly from plant sources results in extremely low recovery rates due to their trace presence in the biomass, leading to volatile market pricing and inconsistent supply chains that cannot meet the demands of clinical trials or commercial launch. Furthermore, earlier synthetic attempts often involved lengthy linear sequences with numerous protection and deprotection steps, which drastically increased the cost of goods sold (COGS) and generated substantial chemical waste. The lack of efficient stereocontrol in older methodologies frequently resulted in complex mixtures of diastereomers, necessitating expensive and time-consuming purification processes that hindered the rapid scale-up required for global pharmaceutical distribution.
The Novel Approach
In stark contrast, the novel approach detailed in CN113292630A utilizes a highly efficient cascade of reactions to construct the target molecules with precision. The synthesis begins with the preparation of a key unsaturated aldehyde intermediate through a controlled alkylation reaction, setting the stage for the subsequent ring-forming events. The centerpiece of this innovation is the application of a transition metal-catalyzed hydrogen atom transfer reaction, which enables the rapid assembly of the cyclic core under mild conditions. This method significantly reduces the number of synthetic steps compared to traditional routes, thereby minimizing material loss and operational complexity. By establishing the complete carbon skeleton early in the sequence, chemists can focus subsequent steps on fine-tuning the oxidation states, resulting in a streamlined process that is both economically viable and environmentally superior for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Transition Metal Catalyzed Hydrogen Atom Transfer
The mechanistic elegance of this synthesis is best exemplified by the conversion of the unsaturated aldehyde precursor into the cyclic aldehyde framework. This transformation relies on a visible-light-promoted hydrogen atom transfer (HAT) process mediated by a cobalt porphyrin catalyst, such as Co(TPP), in the presence of a silane reductant like phenylsilane. Upon irradiation with 450 nm LED light, the cobalt catalyst facilitates the generation of a radical species that undergoes intramolecular cyclization, effectively closing the ring with high regioselectivity and stereoselectivity. This radical mechanism bypasses the high energy barriers associated with thermal ionic cyclizations, allowing the reaction to proceed at room temperature and preserving sensitive functional groups that might otherwise degrade under harsh acidic or basic conditions typically found in classical terpene synthesis.

From an impurity control perspective, this catalytic system offers distinct advantages by minimizing side reactions such as polymerization or over-reduction. The use of a specific cobalt porphyrin complex ensures that the hydrogen atom transfer occurs selectively at the desired olefinic position, directing the formation of the thermodynamically favored cis-fused ring junctions essential for the biological activity of Triptonide. Furthermore, the mild reaction conditions prevent the epimerization of chiral centers adjacent to carbonyl groups, a common pitfall in terpene chemistry that can lead to difficult-to-separate impurities. This high level of stereochemical fidelity simplifies downstream purification, ensuring that the final active pharmaceutical ingredient (API) meets stringent regulatory specifications for chirality and purity without the need for extensive recrystallization or chiral chromatography.
How to Synthesize Triptonide Intermediates Efficiently
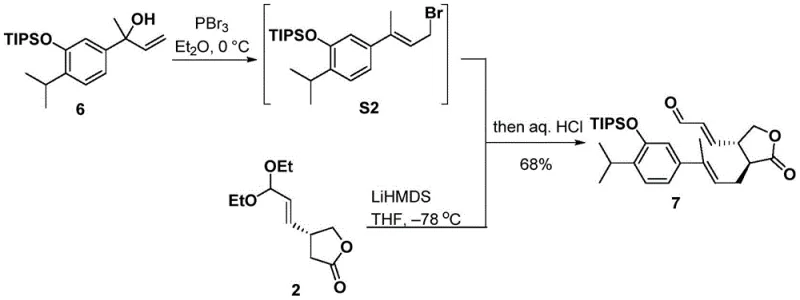
The practical execution of this synthesis involves a series of well-defined chemical transformations that begin with the coupling of two advanced fragments. The process initiates with the alkylation of a lithiated acetal species with a benzylic bromide derivative, forming the crucial carbon-carbon bond that links the aromatic and aliphatic domains of the molecule. Following this coupling, the acetal protecting group is removed to reveal the aldehyde functionality required for the subsequent cyclization. This sequence is critical for establishing the correct connectivity before the ring-closing event, and it demonstrates the robustness of the route in handling multifunctional intermediates without compromising yield or selectivity.
- Prepare unsaturated aldehyde compound 7 by reacting acetal compound 2 with alcohol compound 6 under alkylation conditions using LiHMDS.
- Convert compound 7 to aldehyde 8 using a transition metal catalyst (e.g., Co(TPP)) and phenylsilane under 450 nm LED irradiation.
- Perform deprotection, ring-closing, and oxidation sequences on compound 8 to yield the epoxy compound 9, followed by further oxidation to Triptonide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents compelling advantages centered around stability and cost efficiency. By moving away from agriculture-dependent extraction, manufacturers can decouple production volumes from seasonal harvest variations and geopolitical risks associated with raw plant material sourcing. The synthetic pathway utilizes commercially available starting materials and reagents, such as (-)-Taniguchi lactone derivatives and standard silanes, which are sourced from established chemical supply chains. This shift ensures a consistent and predictable supply of high-purity intermediates, mitigating the risk of production delays that often plague natural product-based supply networks and enabling long-term planning for clinical and commercial manufacturing campaigns.
- Cost Reduction in Manufacturing: The implementation of this technology drives significant cost optimization by eliminating the need for expensive chiral resolution steps and reducing the total number of unit operations. The use of visible-light photocatalysis replaces energy-intensive heating or cooling protocols, lowering utility costs per kilogram of product. Additionally, the high atom economy of the hydrogen atom transfer step minimizes waste generation, reducing the financial burden associated with hazardous waste disposal and solvent recovery. These cumulative efficiencies translate into a lower cost of goods, making the final API more accessible for broad therapeutic applications while maintaining healthy profit margins for manufacturers.
- Enhanced Supply Chain Reliability: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are readily transferable from laboratory glassware to industrial reactors. The reliance on robust chemical transformations rather than fragile enzymatic or fermentation processes ensures that production can be ramped up quickly to meet surging demand without compromising quality. Furthermore, the modular nature of the synthesis allows for the stocking of key intermediates, providing a buffer against supply chain disruptions. This resilience is critical for maintaining continuity of supply for life-saving medications, ensuring that patients have uninterrupted access to treatments derived from these complex molecular scaffolds.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns with green chemistry principles by reducing solvent usage and avoiding toxic heavy metal catalysts often found in traditional cross-coupling reactions. The photochemical step operates at ambient temperature and pressure, enhancing process safety by eliminating high-energy hazards. The simplified workup procedures, primarily involving standard extractions and chromatography, facilitate easier compliance with increasingly strict environmental regulations regarding effluent discharge. This sustainability profile not only reduces regulatory risk but also enhances the corporate social responsibility (CSR) standing of the manufacturing entity, appealing to eco-conscious partners and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the patented synthesis of Triptonide and Triptolide. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of this novel chemical route. Understanding these details is essential for technical teams evaluating the integration of this technology into existing production pipelines or for R&D groups exploring new analogues based on this scaffold.
Q: What is the key catalytic innovation in this Triptonide synthesis route?
A: The core innovation is the use of a visible-light promoted transition metal catalyzed hydrogen atom transfer (HAT) reaction to construct the cyclic framework, replacing traditional multi-step methods.
Q: How does this method improve supply chain reliability for Triptolide precursors?
A: By utilizing a concise synthetic route starting from (-)-Taniguchi lactone with high atom economy, the method reduces dependency on low-yield natural extraction, ensuring consistent availability.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions involve simple operations such as standard alkylation and photochemical steps that are amenable to scale-up from laboratory to industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triptonide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric synthesis technologies described in CN113292630A for the global pharmaceutical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this metal-catalyzed HAT process are translated into robust manufacturing realities. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of Triptonide or Triptolide intermediate delivered meets the highest international standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge synthesis for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
