Revolutionizing Conjugated Diyne Production with Stable Ortho-Carborane Copper Catalysts for Commercial Scale-Up
Revolutionizing Conjugated Diyne Production with Stable Ortho-Carborane Copper Catalysts for Commercial Scale-Up
The chemical industry is constantly seeking more efficient and sustainable pathways for constructing carbon-carbon bonds, particularly for high-value intermediates used in pharmaceuticals and advanced materials. A significant breakthrough in this domain is detailed in patent CN113105301A, which introduces a novel method for preparing conjugated diyne compounds using a specialized copper complex. This technology leverages an ortho-carborane Schiff base ligand to stabilize the copper center, enabling highly efficient Glaser coupling reactions under remarkably mild conditions. For R&D directors and process chemists, this represents a paradigm shift from traditional methods that often rely on stoichiometric oxidants or extreme thermal conditions. The ability to utilize atmospheric air as the sole oxidant not only enhances safety profiles but also drastically reduces the generation of hazardous waste streams. As a reliable pharma intermediate supplier, understanding such technological advancements is crucial for maintaining a competitive edge in the synthesis of complex molecular architectures.
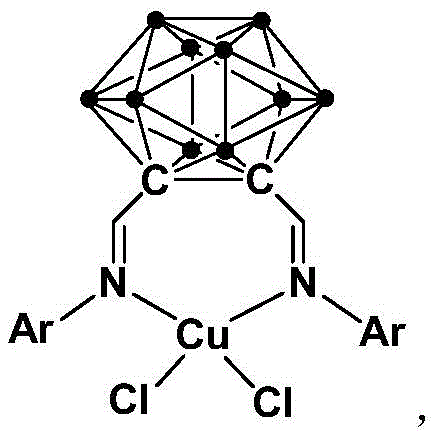
The structural integrity of the catalyst is paramount for its performance. As illustrated in the provided structural diagram, the copper center is coordinated by a rigid ortho-carborane cage functionalized with Schiff base moieties. This unique architecture provides exceptional steric protection and electronic modulation, preventing the catalyst from decomposing or aggregating during the reaction cycle. For procurement managers, the stability of this catalyst translates directly into cost reduction in pharma intermediate manufacturing, as lower catalyst loadings are required to achieve high conversion rates. Furthermore, the robustness of the carborane cage ensures that the catalyst remains active even in the presence of air and moisture, eliminating the need for expensive inert gas setups and anhydrous solvents. This operational simplicity is a key factor in reducing the overall cost of goods sold (COGS) for large-scale production runs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of conjugated diynes via Glaser coupling has been plagued by several significant drawbacks that hinder industrial adoption. Traditional protocols frequently employ palladium or copper salts in conjunction with strong, stoichiometric oxidants such as TMEDA/CuCl systems or require high-pressure oxygen environments. These conditions often lead to poor selectivity, resulting in complex mixtures of homocoupling and oligomerization byproducts that are difficult to separate. Moreover, the use of harsh oxidants generates substantial amounts of toxic waste, posing severe environmental compliance challenges and increasing disposal costs. For supply chain heads, the reliance on sensitive catalysts that degrade rapidly upon exposure to air necessitates stringent storage conditions and specialized handling equipment, which complicates logistics and increases lead time for high-purity intermediates. Additionally, many conventional methods struggle with aliphatic alkynes, yielding poor results and limiting the scope of accessible chemical space for drug discovery programs.
The Novel Approach
In stark contrast, the methodology disclosed in CN113105301A offers a transformative solution by utilizing a pre-formed copper complex containing an ortho-carborane Schiff base ligand. This innovative approach allows the Glaser coupling reaction to proceed smoothly at room temperature using nothing more than ambient air as the oxidant. The reaction system is incredibly forgiving, tolerating a wide variety of functional groups without the need for protective group strategies. This mildness is particularly advantageous for the commercial scale-up of complex polymer additives or electronic chemicals where thermal sensitivity is a concern. The high turnover number (TON) achieved with minimal catalyst loading (as low as 0.01 mmol) ensures that residual metal contamination in the final product is negligible, thereby simplifying the purification process. By eliminating the need for external oxidants and high-energy inputs, this novel route aligns perfectly with green chemistry principles while delivering superior yields and purity profiles.
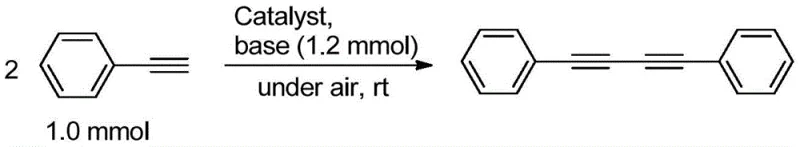
Mechanistic Insights into Ortho-Carborane Copper Complex Catalyzed Glaser Coupling
The exceptional catalytic activity observed in this system can be attributed to the unique electronic and steric properties imparted by the ortho-carborane scaffold. The carborane cage acts as a three-dimensional aromatic system that withdraws electron density from the coordinating nitrogen atoms, thereby tuning the Lewis acidity of the copper center. This electronic modulation facilitates the oxidative addition of the terminal alkyne and stabilizes the key copper-acetylide intermediate. Mechanistically, the reaction likely proceeds through a Cu(I)/Cu(II) redox cycle where molecular oxygen from the air serves to re-oxidize the reduced copper species back to the active state. The rigid geometry of the ligand prevents the formation of inactive polynuclear copper clusters, a common deactivation pathway in simpler copper salt systems. For R&D teams, understanding this mechanism is vital for optimizing reaction parameters such as base selection and solvent polarity to maximize throughput. The stability of the intermediate species ensures that the reaction kinetics remain favorable even at low catalyst concentrations, driving the equilibrium towards the desired diyne product with high selectivity.
Impurity control is another critical aspect where this catalyst excels. In traditional Glaser couplings, the formation of E-enynes or higher-order oligomers is a persistent issue due to uncontrolled radical pathways. However, the well-defined coordination sphere of the ortho-carborane copper complex enforces a specific geometric arrangement that favors the head-to-head coupling of two alkyne units. This stereochemical control minimizes the formation of side products, resulting in a cleaner crude reaction mixture. Consequently, the downstream purification burden is significantly reduced, often requiring only a simple column chromatography or crystallization step to achieve pharmaceutical-grade purity. This level of control over the impurity profile is essential for meeting the stringent regulatory requirements of API manufacturing, ensuring that the final conjugated diyne compounds are free from genotoxic impurities or heavy metal residues that could compromise patient safety.
How to Synthesize Conjugated Diyne Compounds Efficiently
The practical implementation of this technology involves a straightforward two-stage process: first, the preparation of the robust copper catalyst, and second, its application in the coupling reaction. The catalyst synthesis itself is a one-pot procedure involving the condensation of ortho-carborane dicarbaldehyde with various arylamines, followed by complexation with copper(II) chloride. This simplicity allows for the rapid generation of a library of catalyst variants to fine-tune reactivity for specific substrates. Once the catalyst is secured, the coupling reaction is performed by simply mixing the terminal alkyne, a mild base like cesium carbonate, and the catalyst in a solvent such as methanol or toluene. The detailed standardized synthesis steps see the guide below for precise molar ratios and workup procedures tailored for maximum efficiency.
- Prepare the catalyst by reacting ortho-carborane dicarbaldehyde with arylamine in toluene, followed by complexation with CuCl2.
- Dissolve the terminal alkyne substrate and base (e.g., Cs2CO3) in methanol or toluene within a reaction vessel.
- Add the copper complex catalyst and stir at room temperature under an open air atmosphere for 3 to 8 hours to complete the coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers profound strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the reaction infrastructure. Since the process operates under ambient air and room temperature, there is no need for costly high-pressure reactors, cryogenic cooling systems, or inert gas manifolds. This reduction in capital expenditure (CAPEX) allows manufacturers to utilize existing general-purpose equipment, accelerating the timeline from pilot plant to full commercial production. Furthermore, the use of air as a free and abundant oxidant eliminates the recurring cost of purchasing and storing hazardous chemical oxidants, leading to substantial cost savings in raw material procurement. The stability of the catalyst also means it can be stored for extended periods without degradation, reducing waste associated with expired reagents and ensuring consistent supply continuity.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the extremely low catalyst loading required to achieve high yields. Traditional copper-catalyzed couplings often require stoichiometric or near-stoichiometric amounts of copper salts, which not only increases material costs but also creates a massive burden for metal removal during purification. In this novel system, catalyst loadings as low as 1 mol% or less are sufficient, which significantly lowers the cost per kilogram of the final product. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further driving down utility costs. The simplified workup procedure, often requiring only filtration and solvent removal, minimizes labor hours and solvent usage, contributing to a leaner and more cost-effective manufacturing process overall.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and ease of handling of the ortho-carborane copper complexes. Unlike sensitive organometallic catalysts that require cold chain shipping and glovebox handling, these complexes are stable in air and moisture. This stability simplifies logistics, allowing for standard shipping methods and reducing the risk of spoilage during transit. For global supply chains, this means shorter lead times and fewer disruptions caused by specialized storage requirements. Moreover, the wide substrate scope implies that a single catalyst platform can be used to produce a diverse range of diyne intermediates, reducing the need to source and validate multiple different catalytic systems. This versatility streamlines inventory management and ensures that production lines can be quickly adapted to changing market demands.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this method is inherently designed for scalability. The use of air as an oxidant avoids the accumulation of energetic peroxide species that can pose explosion risks in large batches. From an environmental perspective, the atom economy of the reaction is excellent, with water being the primary byproduct of the oxidation step. This aligns with increasingly stringent global regulations on volatile organic compounds (VOCs) and heavy metal discharge. The ability to run the reaction in greener solvents like methanol or ethanol further enhances the environmental profile, making it easier to obtain necessary environmental permits and maintain a sustainable corporate image. The combination of safety, scalability, and eco-friendliness makes this technology a future-proof choice for long-term manufacturing strategies.
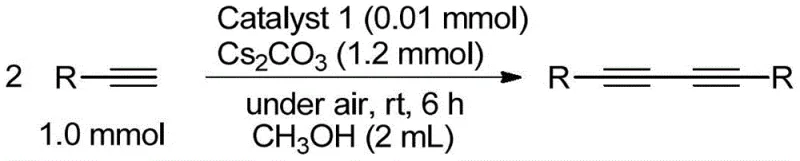
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed Glaser coupling technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear picture of what potential partners can expect regarding performance and operational requirements. Understanding these nuances is critical for making informed decisions about integrating this method into existing production workflows.
Q: What are the advantages of using the ortho-carborane copper complex over traditional Glaser coupling catalysts?
A: Unlike traditional systems that often require harsh oxidants or elevated temperatures, this novel copper complex utilizes ambient air as the sole oxidant and operates efficiently at room temperature, significantly simplifying the process and reducing energy consumption.
Q: Is this catalytic system suitable for large-scale industrial production of diynes?
A: Yes, the catalyst demonstrates high stability and can be used at very low loadings (0.01-0.03 mmol), which minimizes metal contamination and downstream purification costs, making it highly viable for commercial scale-up.
Q: What is the substrate scope for this Glaser coupling method?
A: The method exhibits excellent versatility, successfully coupling a wide range of terminal alkynes including aryl, heteroaryl, and aliphatic substrates with high yields and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Conjugated Diyne Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ortho-carborane copper complex system for producing high-value conjugated diynes. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of conjugated diyne compound meets the highest international standards for pharmaceutical and electronic applications. We are committed to leveraging our technical expertise to optimize these novel routes for your specific needs, ensuring maximum yield and minimal environmental impact.
We invite you to collaborate with us to unlock the full commercial potential of your diyne-based projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this catalytic method can improve your margins. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us be your partner in driving innovation and efficiency in the fine chemical sector.
