Revolutionizing Steroid Intermediate Production: The Green One-Pot Synthesis of 16-Dehydropregnenolone Acetate
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, particularly in the synthesis of high-value steroid intermediates. Patent CN101974057A introduces a groundbreaking green preparation method for 16-dehydropregnenolone acetate and its congeners, addressing critical environmental and economic challenges associated with traditional steroid synthesis. This technology leverages a sophisticated vanadium-catalyzed oxidation system combined with phase transfer catalysis to achieve a highly efficient 'one-pot' transformation. By replacing hazardous chromium-based oxidants with benign hydrogen peroxide and utilizing water as the primary reaction medium, this method offers a compelling value proposition for manufacturers seeking to optimize their production workflows while adhering to strict environmental standards. The ability to conduct simultaneous oxidation and elimination reactions without the need for volatile organic solvents represents a significant technological leap forward.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 16-dehydropregnenolone acetate, a pivotal precursor for corticosteroids and sex hormones, has relied heavily on the Marker degradation method. This conventional route typically involves the oxidation of pseudo-diosgenin using chromium trioxide in acetic acid, followed by a separate elimination step under alkaline conditions. This multi-step process is fraught with inefficiencies, including the generation of large volumes of toxic chromium waste which poses severe disposal challenges and environmental risks. Furthermore, the reliance on organic solvents like acetic acid necessitates complex recovery systems to prevent atmospheric pollution and reduce raw material costs. The overall yield of such traditional methods often hovers around 65%, and the requirement for distinct reaction vessels for oxidation and elimination increases capital expenditure and operational complexity, making it less attractive for modern, high-throughput manufacturing facilities.
The Novel Approach
In stark contrast, the novel methodology described in the patent data utilizes a streamlined one-pot strategy that integrates oxidation and elimination into a single operational unit. By employing pseudo-steroidal sapogenin acetates as starting materials and reacting them with hydrogen peroxide in the presence of vanadium and quaternary ammonium catalysts, the process achieves superior conversion rates directly in an aqueous environment. This approach not only eliminates the need for toxic chromium reagents but also drastically reduces the consumption of organic solvents, as water serves as the effective reaction medium. The structural versatility of this method allows for the synthesis of various congeners, including 3β-acetoxy-5α-pregna-16(17)-ene-20-one, by simply adjusting the starting sapogenin substrate. 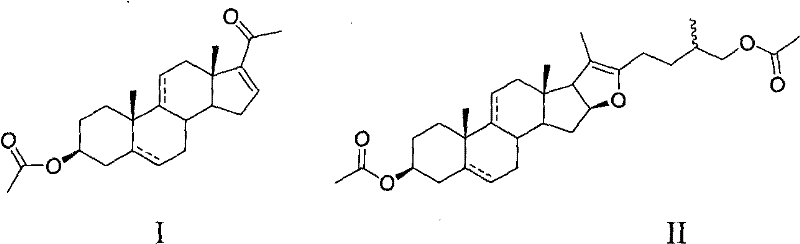
Mechanistic Insights into Vanadium-Catalyzed Oxidative Elimination
The core of this technological advancement lies in the synergistic interaction between the vanadium-containing compound catalyst and the quaternary ammonium phase transfer catalyst. The vanadium species, which can range from simple oxides like V2O5 to complex coordination complexes, act as the primary activators for hydrogen peroxide, generating reactive oxygen species capable of selectively oxidizing the steroid side chain. Simultaneously, the quaternary ammonium salts function as phase transfer agents, facilitating the transport of hydrophobic steroid substrates into the aqueous phase where the oxidation occurs. This dual-catalyst system ensures that the reaction proceeds efficiently despite the immiscibility of the organic substrate and the aqueous oxidant, maintaining high reaction kinetics throughout the process duration of 3 to 24 hours at moderate temperatures ranging from 55°C to 100°C.
From an impurity control perspective, this mechanism offers distinct advantages over harsh chemical oxidation methods. The mild yet effective nature of the vanadium-peroxide system minimizes over-oxidation and side reactions that typically degrade the steroid nucleus, thereby preserving the integrity of the sensitive double bonds within the molecule. The use of specific quaternary ammonium structures, such as those depicted in Formula IV, allows for fine-tuning of the interfacial properties, further enhancing selectivity. 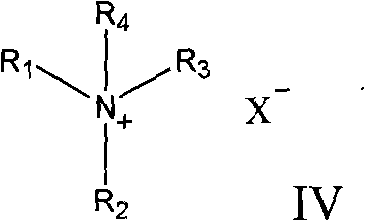
How to Synthesize 16-Dehydropregnenolone Acetate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and catalyst longevity. The process begins with the suspension of the pseudo-steroidal sapogenin acetate in water, followed by the sequential addition of the vanadium catalyst, the quaternary ammonium phase transfer catalyst, and hydrogen peroxide. Mechanical stirring is critical to maintain a homogeneous suspension and ensure effective mass transfer between the phases. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined below to guide R&D teams in replicating this high-efficiency protocol.
- Prepare the reaction mixture by suspending pseudo-steroidal sapogenin acetate in water with a vanadium-containing compound catalyst and a quaternary ammonium phase transfer catalyst.
- Add hydrogen peroxide as the oxidant and maintain mechanical stirring at temperatures between 55°C and 100°C for 3 to 24 hours to facilitate simultaneous oxidation and elimination.
- Perform post-treatment by separating the aqueous layer, extracting the product with organic solvent, and recrystallizing to obtain the final high-purity 16-dehydropregnenolone acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this green synthesis technology translates into tangible strategic benefits that extend beyond mere regulatory compliance. The elimination of chromium trioxide removes the necessity for expensive heavy metal scavenging and specialized waste treatment infrastructure, leading to a significant reduction in overall operational expenditures. Furthermore, the ability to recycle the catalytic system for multiple batches without regeneration creates a closed-loop material flow that stabilizes raw material costs and insulates the production line from volatility in reagent pricing. This robustness ensures a more predictable cost structure for long-term manufacturing contracts.
- Cost Reduction in Manufacturing: The transition to an aqueous-based system fundamentally alters the cost dynamics of steroid intermediate production by removing the dependency on vast quantities of organic solvents. Traditional methods require extensive solvent recovery units which are energy-intensive and prone to losses; in contrast, this method utilizes water which is inexpensive and easily separated. Additionally, the high turnover number of the recyclable catalysts means that the cost per kilogram of the active catalytic components is amortized over a much larger production volume, driving down the variable cost of goods sold substantially.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and safety of the reagents involved. Hydrogen peroxide and vanadium compounds are commodity chemicals with stable global supply chains, unlike specialized or regulated toxic oxidants which may face shipping restrictions or supply disruptions. The simplified process flow, which combines two reaction steps into one, also reduces the total processing time and equipment footprint, allowing for faster batch turnover and improved responsiveness to market demand fluctuations without the need for massive capital expansion.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is facilitated by the use of standard stainless steel reactors and conventional mechanical stirring equipment, avoiding the need for exotic corrosion-resistant materials often required for acidic chromium processes. From an environmental standpoint, the absence of heavy metal effluents and volatile organic compound (VOC) emissions simplifies the permitting process and ensures continuous operation even under tightening environmental regulations, thereby mitigating the risk of production stoppages due to compliance issues.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this vanadium-catalyzed green synthesis method. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a clear understanding of the process capabilities and limitations for potential partners and stakeholders evaluating this technology for integration into their existing manufacturing portfolios.
Q: How does this green synthesis method improve upon the traditional Marker degradation process?
A: Unlike the traditional Marker method which relies on toxic chromium trioxide and requires separate oxidation and elimination steps in organic solvents, this novel approach utilizes a vanadium-catalyzed one-pot system in aqueous media. This eliminates the need for hazardous heavy metals, significantly reduces solvent consumption, and streamlines the workflow by combining oxidation and elimination into a single reaction vessel, thereby lowering both environmental impact and production costs.
Q: Can the catalysts used in this process be recycled for multiple batches?
A: Yes, a key advantage of this technology is the recyclability of the catalytic system. The patent data demonstrates that both the vanadium-containing compounds and the quaternary ammonium phase transfer catalysts can be recovered and reused for more than 10 consecutive cycles without significant loss in catalytic activity or product yield, ensuring consistent production efficiency and minimizing raw material waste.
Q: What represent the primary commercial benefits for large-scale manufacturing of this steroid intermediate?
A: The primary commercial benefits include substantial cost reduction through the elimination of expensive organic solvents and toxic chromium reagents, alongside simplified waste treatment protocols due to the use of water as the primary reaction medium. Furthermore, the robust nature of the catalyst system allows for reliable scale-up from laboratory to industrial production, ensuring supply chain continuity and compliance with increasingly stringent environmental regulations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16-Dehydropregnenolone Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting sustainable and efficient synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering high-purity steroid intermediates that meet stringent purity specifications, supported by our rigorous QC labs which utilize advanced analytical techniques to verify identity and assay every batch.
We invite you to collaborate with us to leverage this cutting-edge green synthesis technology for your supply chain needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can drive value and reliability for your organization.
