Advanced Asymmetric Synthesis of Chiral Beta-Hydroxy Phosphates for Pharmaceutical Applications
Advanced Asymmetric Synthesis of Chiral Beta-Hydroxy Phosphates for Pharmaceutical Applications
The development of efficient routes to optically active organophosphorus compounds remains a critical challenge in modern medicinal chemistry, particularly for the production of enzyme inhibitors and bioactive intermediates. Patent CN109096329B introduces a groundbreaking asymmetric synthesis method for chiral beta-hydroxy phosphates, utilizing a sophisticated transfer hydrogenation protocol that bypasses traditional high-pressure hydrogenation limitations. This technology leverages stable chiral diamine ligands coordinated with transition metals such as ruthenium, rhodium, or iridium to achieve exceptional enantioselectivity under mild conditions. The resulting compounds serve as pivotal scaffolds for renin inhibitors, HIV protease inhibitors, and various agrochemical agents, representing a significant value-add for supply chains focused on high-purity pharmaceutical intermediates. By employing sodium formate as a safe hydrogen donor in trifluoroethanol-water mixtures, this process aligns perfectly with green chemistry principles while delivering robust yields suitable for industrial scaling.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-hydroxy phosphonates has relied heavily on stoichiometric chiral reducing agents or high-pressure catalytic hydrogenation, both of which present substantial operational and economic drawbacks for large-scale manufacturing. Stoichiometric reductions often generate excessive waste streams and require costly chiral auxiliaries that cannot be recovered, leading to inflated production costs and complex purification workflows that erode profit margins. Furthermore, traditional high-pressure hydrogenation necessitates specialized reactor infrastructure capable of withstanding significant safety risks associated with compressed hydrogen gas, creating bottlenecks in facilities lacking such dedicated equipment. The sensitivity of phosphorus-containing substrates to harsh reducing conditions can also lead to side reactions, such as dephosphorylation or over-reduction, which compromise the integrity of the final active pharmaceutical ingredient and complicate the impurity profile.
The Novel Approach
The methodology disclosed in CN109096329B revolutionizes this landscape by employing asymmetric transfer hydrogenation (ATH) using formate salts, which operates effectively at atmospheric pressure and moderate temperatures ranging from 35°C to 50°C. This approach utilizes readily available and inexpensive chiral diamine ligands that exhibit superior stability compared to traditional phosphine ligands, ensuring consistent catalytic performance over extended reaction cycles without rapid degradation. The use of a trifluoroethanol and water solvent system not only enhances the solubility of polar intermediates but also facilitates easier product isolation through simple aqueous workups, drastically reducing solvent consumption and waste disposal costs. By achieving enantiomeric excess values exceeding 98% in optimized examples, this novel route provides a direct, atom-economical pathway to high-value chiral building blocks that was previously inaccessible through standard reduction techniques.
Mechanistic Insights into Monosulfonyl Chiral Diamine-Metal Catalyzed Reduction
The core of this technological breakthrough lies in the precise coordination environment created by the monosulfonyl chiral diamine ligands bound to Group 8-10 transition metals, specifically Ruthenium, Rhodium, and Iridium complexes. These catalysts function through a concerted outer-sphere mechanism where the metal-hydride species transfers a hydride ion to the carbonyl carbon of the beta-keto phosphate substrate while a proton is simultaneously delivered from the ligand's NH moiety. This bifunctional activation mode ensures rigorous stereocontrol, as the bulky aryl groups on the diamine backbone create a well-defined chiral pocket that directs the approach of the substrate, favoring the formation of one enantiomer over the other with high fidelity. The electron-withdrawing nature of the sulfonyl group on the ligand modulates the acidity of the NH proton and the electrophilicity of the metal center, fine-tuning the catalytic activity to match the specific electronic demands of the phosphorylated ketone substrate.
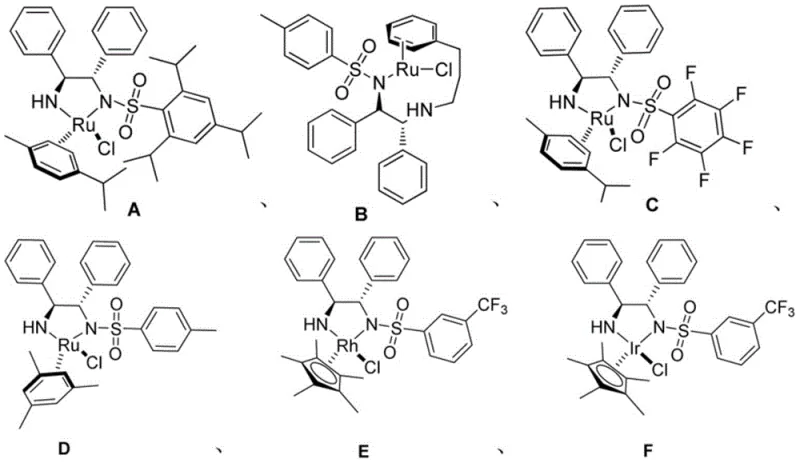
Impurity control in this system is inherently managed by the mild reaction conditions and the high chemoselectivity of the transfer hydrogenation mechanism, which selectively targets the ketone functionality without affecting the sensitive phosphate ester bonds. Unlike strong hydride reagents that might attack the phosphorus center or cleave ester linkages, the formate-mediated hydride transfer is gentle enough to preserve the structural integrity of the molecule while reducing the carbonyl group. The use of water as a co-solvent further aids in suppressing non-specific side reactions by stabilizing charged intermediates and facilitating the removal of inorganic byproducts like sodium salts during the workup phase. This results in a crude product profile that is significantly cleaner than those obtained from conventional methods, reducing the burden on downstream purification steps such as chromatography and crystallization, which is critical for maintaining cost efficiency in commercial production.
How to Synthesize Chiral Beta-Hydroxy Phosphates Efficiently
To implement this synthesis effectively, manufacturers should adhere to the optimized protocols outlined in the patent examples, which demonstrate that catalyst loadings as low as 1 mol% are sufficient to drive the reaction to completion within 4 hours. The process begins with the preparation of the catalyst solution, often generated in situ by mixing the chiral diamine ligand with a metal precursor like [Ru(cymene)]2Cl2 in a halogenated solvent before introducing it to the main reaction vessel containing the substrate and sodium formate. Maintaining an inert nitrogen atmosphere is crucial to prevent oxidation of the active metal-hydride species, and the reaction temperature should be strictly controlled around 40°C to balance reaction rate with enantioselectivity. Detailed standardized synthetic steps for specific substrate variations are provided in the guide below to ensure reproducibility and quality consistency across different batches.
- Prepare the reaction mixture by adding the beta-carbonyl phosphate substrate, sodium formate hydrogen source, and chiral diamine-metal catalyst to a vessel.
- Add the solvent system, typically a mixture of trifluoroethanol and water, and purge the system with nitrogen to ensure an inert atmosphere.
- Heat the reaction mixture to 35-50°C for 3-5 hours, then perform aqueous workup and extraction to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this asymmetric transfer hydrogenation technology offers transformative benefits that directly impact the bottom line and operational resilience of pharmaceutical manufacturing networks. The shift away from high-pressure hydrogen gas to solid sodium formate eliminates the need for expensive high-pressure autoclaves and the associated safety certifications, allowing production to be shifted to more versatile multipurpose reactors that are commonly available in contract manufacturing organizations. This flexibility significantly reduces capital expenditure barriers and shortens the lead time required to establish new production lines for chiral intermediates, enabling faster response to market demands for novel drug candidates. Additionally, the stability and commercial availability of the chiral diamine ligands ensure a reliable supply of critical catalytic materials, mitigating the risk of production stoppages due to specialized reagent shortages that often plague processes relying on exotic phosphine ligands.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the use of inexpensive sodium formate as a hydrogen source drastically lower the operational costs associated with reactor maintenance and safety compliance. Furthermore, the ability to use water-miscible solvents like trifluoroethanol simplifies solvent recovery and recycling processes, leading to substantial reductions in raw material consumption and waste treatment expenses. The high selectivity of the catalyst minimizes the formation of diastereomers and byproducts, which increases the overall yield of the desired active isomer and reduces the volume of material lost during purification, thereby maximizing the return on investment for every kilogram of starting material purchased.
- Enhanced Supply Chain Reliability: By utilizing robust catalyst systems that tolerate a wide range of functional groups, manufacturers can source diverse aromatic ketone precursors from multiple global suppliers without fearing compatibility issues that could halt production. The mild reaction conditions reduce the thermal stress on equipment, extending the lifespan of reactor vessels and agitation systems, which contributes to higher asset utilization rates and more predictable maintenance schedules. This reliability is crucial for long-term supply agreements, as it ensures that production timelines remain consistent even when scaling up from pilot plant quantities to multi-ton commercial campaigns, providing partners with the confidence needed to plan their own downstream formulation activities.
- Scalability and Environmental Compliance: The aqueous workup procedure inherent to this method aligns with increasingly stringent environmental regulations regarding volatile organic compound emissions, making it easier to obtain necessary permits for expansion in regulated jurisdictions. The process generates minimal hazardous waste compared to stoichiometric reductions, simplifying the disposal workflow and reducing the environmental footprint of the manufacturing site. Scalability is further enhanced by the exothermic nature of the transfer hydrogenation being manageable at 40°C, preventing thermal runaways that are common in large-scale hydrogenations, thus allowing for safe and efficient production of complex organophosphorus compounds at the 100 MT annual scale required by major pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this asymmetric synthesis platform, derived directly from the experimental data and scope defined in the patent literature. Understanding these nuances is essential for R&D teams looking to adapt this chemistry to their specific proprietary substrates or for quality assurance personnel verifying process parameters. The answers reflect the robustness of the catalyst system and the versatility of the reaction conditions across various electronic and steric environments found in drug-like molecules.
Q: What represents the primary advantage of using sodium formate over hydrogen gas in this synthesis?
A: Using sodium formate as a hydrogen source eliminates the need for high-pressure hydrogenation equipment, significantly enhancing operational safety and reducing capital expenditure for reactor infrastructure while maintaining high enantioselectivity.
Q: Which metal centers are most effective for this asymmetric transfer hydrogenation?
A: The patent specifies that complexes of Ruthenium (Ru), Rhodium (Rh), and Iridium (Ir) coordinated with monosulfonyl chiral diamine ligands provide superior catalytic activity and stereocontrol for beta-carbonyl phosphate reduction.
Q: Can this method tolerate diverse functional groups on the aromatic ring?
A: Yes, the methodology demonstrates excellent substrate scope, successfully accommodating electron-donating groups like methoxy and methyl, as well as electron-withdrawing groups such as halogens and trifluoromethyl substituents without compromising optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Hydroxy Phosphate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of securing a stable supply of high-enantiopurity intermediates for the development of next-generation antiviral and antihypertensive therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and compliant with international quality standards. We utilize stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC capabilities to guarantee that every batch of chiral beta-hydroxy phosphate meets the exacting requirements of global regulatory bodies. Our commitment to process excellence means we can replicate the high enantioselectivity demonstrated in patent CN109096329B while optimizing the workflow for maximum cost efficiency and throughput.
We invite potential partners to engage with our technical procurement team to discuss how this advanced asymmetric transfer hydrogenation technology can be integrated into your specific supply chain to drive down costs and accelerate time-to-market. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this formate-based reduction method for your specific project needs. We encourage you to contact us today to obtain specific COA data for our catalog of chiral phosphonates and to receive comprehensive route feasibility assessments tailored to your unique molecular targets, ensuring a partnership built on transparency, quality, and shared success.
