Revolutionizing C-S Bond Formation: A Scalable, Metal-Free Route for High-Purity Pharmaceutical Intermediates
Introduction to Patent CN113683559B: A Breakthrough in Sulfur Chemistry
The landscape of organic synthesis is constantly evolving, driven by the need for more sustainable and cost-effective methodologies. Patent CN113683559B introduces a transformative approach to constructing carbon-sulfur bonds, specifically targeting the synthesis of heterocyclic o-iodosulfides. This innovation addresses a longstanding challenge in the industry: the difficulty of performing C-S couplings without relying on expensive and sensitive transition metal catalysts. By utilizing a simple yet effective system involving sodium hydride (NaH) and o-diiodobenzene, this method achieves high atom utilization and operates under remarkably mild conditions. For R&D directors and process chemists, this represents a paradigm shift away from complex catalytic cycles towards robust, base-mediated nucleophilic substitutions that are easier to control and optimize.
The significance of this technology extends beyond the laboratory bench; it offers tangible benefits for supply chain stability and manufacturing economics. The ability to synthesize these valuable intermediates without transition metals eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical ingredients. Furthermore, the retention of the ortho-iodine functionality provides a strategic handle for downstream diversification, allowing medicinal chemists to rapidly access a wide array of 1,2-disubstituted benzene derivatives. As we delve deeper into the technical specifics, it becomes clear why this patent is a cornerstone for reliable pharmaceutical intermediate supplier strategies aiming to reduce dependency on precious metals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl sulfide compounds has relied heavily on metal-catalyzed cross-coupling reactions involving palladium, copper, iron, or nickel. While these methods are theoretically sound, they face severe practical limitations in industrial settings. The primary issue is catalyst deactivation; sulfur atoms possess lone pair electrons that coordinate strongly with transition metal centers, often leading to irreversible catalyst poisoning. To overcome this, conventional protocols frequently demand harsh reaction conditions, including high temperatures ranging from 80°C to 140°C, strong bases like sodium tert-butoxide, and specialized, air-sensitive ligands that drive up costs significantly. These stringent requirements not only increase energy consumption but also complicate the purification process, as removing trace metal residues to meet regulatory standards adds multiple processing steps and reduces overall yield.
The Novel Approach
In stark contrast, the methodology disclosed in CN113683559B bypasses these hurdles entirely by employing a transition-metal-free strategy. The core innovation lies in the activation of heterocyclic thiophenols using sodium hydride, a cheap and readily available inorganic base. This generates a highly reactive thiolate anion in situ, which then undergoes a nucleophilic aromatic substitution with o-diiodobenzene. The reaction proceeds smoothly at room temperature, typically completing within 1 to 4 hours, which is a drastic improvement over the thermal energy input required by traditional methods. This mildness preserves sensitive functional groups on the heterocyclic ring, expanding the substrate scope to include molecules that would decompose under vigorous metal-catalyzed conditions. The result is a cleaner reaction profile with fewer byproducts, directly translating to simplified workup procedures and higher purity final products.
Mechanistic Insights into NaH-Mediated Nucleophilic Substitution
Understanding the mechanistic underpinnings of this reaction is crucial for process optimization. Unlike oxidative addition/reductive elimination cycles seen in palladium catalysis, this transformation follows a direct nucleophilic substitution pathway. The sodium hydride acts as a potent deprotonating agent, stripping the proton from the thiol group of the heterocyclic thiophenol to form a sodium thiolate species. This anionic sulfur center is a powerful nucleophile that attacks the electron-deficient aromatic ring of the o-diiodobenzene. The presence of the iodine atom at the ortho-position facilitates this attack, likely through an addition-elimination mechanism or a benzyne intermediate pathway, although the exact trajectory can depend on the specific electronic nature of the heterocycle. Crucially, the reaction conditions are tuned to ensure mono-substitution, leaving the second iodine atom intact for future functionalization.
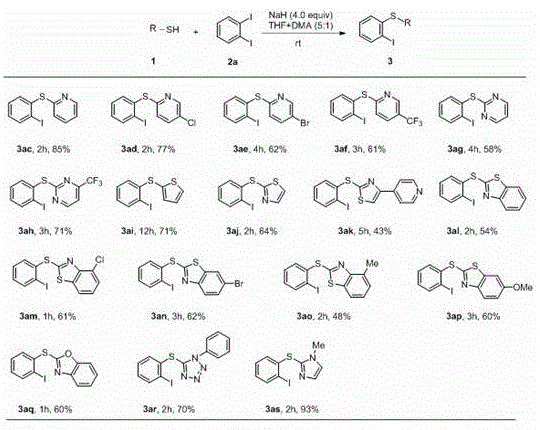
From an impurity control perspective, this mechanism offers distinct advantages. Since no transition metals are involved, there is no risk of metal-ligand complexes forming difficult-to-remove impurities. The primary side reactions are minimized by controlling the stoichiometry of the base and the electrophile. The patent data indicates that using 4.0 equivalents of NaH relative to the thiophenol ensures complete deprotonation and drives the reaction to completion without excessive degradation of the starting materials. Furthermore, the choice of solvent system, specifically a mixture of THF and DMA, plays a vital role in solubilizing both the inorganic base and the organic substrates, ensuring a homogeneous reaction environment that promotes consistent kinetics. This level of control is essential for maintaining batch-to-batch consistency in a commercial manufacturing setting.
How to Synthesize Heterocyclic O-Iodosulfide Efficiently
Implementing this synthesis route requires careful attention to reagent quality and addition rates, particularly when handling reactive hydrides. The general procedure involves suspending the base in an anhydrous solvent to prevent premature decomposition by moisture, followed by the controlled addition of the thiol. Once the thiolate is formed, the electrophile is introduced to initiate the coupling. The simplicity of the workup—quenching with ice water and extracting with standard organic solvents—makes this protocol highly attractive for pilot plant operations. Below is the structured guide derived from the patent examples, detailing the precise operational parameters for achieving optimal yields.
- Suspend Sodium Hydride (NaH, 4.0 equiv) in anhydrous THF under magnetic stirring at room temperature.
- Slowly add the heterocyclic thiophenol (1.0 equiv, dissolved in DMA) to the suspension and stir for 3 minutes to generate the thiolate anion.
- Add o-diiodobenzene (2.0 equiv, dissolved in THF) to the mixture, stir at room temperature until TLC indicates completion, then quench and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this metal-free methodology represents a significant opportunity for cost reduction in API manufacturing. The elimination of precious metal catalysts such as palladium or rhodium removes a major variable cost component that is subject to volatile market pricing. Additionally, the removal of expensive phosphine ligands further decreases the raw material bill. Because the reaction runs at room temperature, there is no need for energy-intensive heating systems, leading to substantial utility savings over large-scale production campaigns. The simplified purification process, devoid of metal scavenging steps, reduces the consumption of silica gel and solvents, thereby lowering waste disposal costs and improving the overall environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The economic benefits are driven primarily by the substitution of high-cost catalytic systems with inexpensive commodity chemicals like sodium hydride and o-diiodobenzene. Traditional methods often require catalyst loadings of 1-5 mol% of expensive metals, whereas this method uses stoichiometric amounts of cheap inorganic salts. Furthermore, the absence of metal residues means that costly purification steps dedicated to reducing metal levels below ppm thresholds are rendered unnecessary. This streamlining of the downstream processing significantly shortens the production cycle time and reduces the labor and material inputs associated with quality control testing for heavy metals.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, commodity-grade raw materials. Sodium hydride and o-diiodobenzene are produced by numerous global suppliers, reducing the risk of single-source bottlenecks that often plague specialized catalyst markets. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality or environmental factors, ensuring consistent output even when sourcing from different vendors. This reliability is critical for maintaining continuous production schedules and meeting the just-in-time delivery expectations of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety and environmental challenges, but this methodology mitigates many of them. Operating at ambient temperature reduces the thermal load on reactors, minimizing the risk of runaway exotherms. Moreover, the avoidance of toxic heavy metals aligns perfectly with increasingly stringent environmental regulations regarding wastewater discharge and solid waste disposal. The process generates less hazardous waste, simplifying compliance reporting and reducing the fees associated with hazardous waste treatment. This green chemistry profile enhances the corporate sustainability metrics of any organization adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these details is essential for making informed decisions about process adoption and vendor selection.
Q: Why is this metal-free method superior to traditional Pd-catalyzed C-S coupling?
A: Traditional methods often suffer from catalyst poisoning due to the strong coordination of sulfur with transition metals like Palladium or Copper. This new NaH-mediated protocol eliminates the need for expensive, air-sensitive metal catalysts and ligands, operating efficiently at room temperature without deactivation issues.
Q: What are the scalability advantages of using Sodium Hydride for this reaction?
A: Sodium Hydride is a commercially abundant and inexpensive reagent. The reaction proceeds under mild conditions (room temperature) without the need for high-pressure equipment or extreme heating, significantly simplifying the engineering requirements for scaling up from grams to tons.
Q: Can the remaining iodine atom on the product be utilized for further synthesis?
A: Yes, a key feature of this methodology is the retention of the ortho-iodine atom. This allows the resulting heterocyclic o-iodosulfide to serve as a versatile building block for subsequent cross-coupling reactions (e.g., Suzuki, Sonogashira) or intramolecular cyclizations to build complex drug scaffolds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Heterocyclic O-Iodosulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic routes like the one described in CN113683559B. Our team of expert process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this metal-free chemistry are fully realized in a GMP-compliant manufacturing environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of heterocyclic o-iodosulfide meets the highest quality standards required for pharmaceutical applications. Our commitment to technical excellence ensures that your supply of critical intermediates remains uninterrupted and compliant.
We invite you to collaborate with us to leverage this advanced technology for your specific drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your project's volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can accelerate your timeline to market while optimizing your overall production costs.
