Scalable n-Butyllithium Catalyzed Hydroboration for High-Purity Pharmaceutical Intermediates
Introduction to Advanced Catalytic Reduction Technologies
The landscape of organic synthesis is constantly evolving, driven by the urgent need for safer, more efficient, and economically viable pathways to produce essential chemical building blocks. A pivotal advancement in this domain is detailed in patent CN109467498B, which introduces a groundbreaking method for preparing alcohol compounds from aromatic carboxylic acids using n-butyllithium as a highly efficient catalyst. This technology represents a paradigm shift away from traditional, hazardous reduction methods towards a more atom-economical and operationally simple hydroboration strategy. By leveraging the unique reactivity of commercial n-butyllithium, this process achieves exceptional catalytic activity at room temperature, utilizing merely 0.5 mol% of the catalyst relative to the carboxylic acid substrate. For R&D directors and process chemists, this innovation offers a compelling solution to the longstanding challenges associated with reducing carboxylic acids, particularly regarding selectivity, safety, and scalability in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of carboxylic acids to their corresponding alcohols has been fraught with significant technical and safety hurdles that complicate industrial manufacturing. Traditional reagents such as Lithium Aluminum Hydride (LiAlH4) and Sodium Borohydride (NaBH4) are widely known but present severe safety risks due to their pyrophoric nature and violent reactions with moisture, necessitating rigorous anhydrous conditions and specialized handling equipment. Furthermore, alternative systems like SmI2-H2O-Et3N require substantial excesses of reagents, leading to poor atom economy and generating massive amounts of waste that are costly to treat. Perhaps most critically, existing transition metal complex systems often demand harsh reaction conditions, typically requiring temperatures around 60°C and extended reaction times exceeding 24 hours. These legacy methods also frequently rely on catalysts that are difficult to synthesize and expensive to procure, creating a bottleneck for cost reduction in API manufacturing and limiting the feasibility of large-scale operations.
The Novel Approach
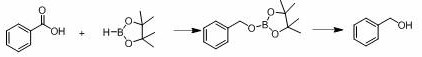
In stark contrast to these cumbersome legacy protocols, the novel approach disclosed in the patent utilizes commercial n-butyllithium to catalyze the hydroboration of carboxylic acids with pinacol borane under remarkably mild conditions. This method eliminates the need for high temperatures and pressures, proceeding efficiently at room temperature within a mere 40 to 50 minutes. The use of a commercially available reagent like n-butyllithium, rather than a bespoke transition metal complex, drastically simplifies the supply chain and reduces raw material costs. The reaction demonstrates broad substrate scope, accommodating various electronic effects and substituent positions on the aromatic ring, which is crucial for the diverse needs of fine chemical synthesis. Moreover, the post-treatment process is exceptionally straightforward, involving a simple hydrolysis step with silica gel and methanol, which facilitates easy isolation of the product and minimizes downstream processing burdens.
Mechanistic Insights into n-Butyllithium Catalyzed Hydroboration
The mechanistic elegance of this transformation lies in the ability of n-butyllithium to activate the hydroboration reaction between the carboxylic acid and the borane species with high specificity. Unlike nucleophilic additions to aldehydes or ketones, carboxylic acids present a unique challenge due to the equivalence of the two oxygen atoms and the steric hindrance imposed by the carboxyl group. The presence of hydrogen bonding in carboxylic acids increases the electron cloud density of the carbonyl group, making it less susceptible to nucleophilic attack by standard reagents. However, the n-butyllithium catalyst effectively overcomes these electronic and steric barriers, facilitating the formation of the borate ester intermediate with high turnover frequency. This catalytic cycle avoids the formation of stable transition states that typically require high energy inputs, allowing the reaction to proceed rapidly at ambient temperatures without the need for external heating sources.
From an impurity control perspective, this mechanism offers distinct advantages over transition metal catalysis. Traditional methods often suffer from side reactions or incomplete conversions that lead to complex impurity profiles, requiring extensive purification steps. The n-butyllithium system, operating under mild conditions, minimizes thermal degradation and side reactions, resulting in a cleaner crude reaction mixture. The subsequent hydrolysis step is equally controlled; by adding silica gel and methanol at 50°C for 2 hours, the borate ester is cleanly converted to the alcohol compound. This two-step sequence ensures that the final product maintains high purity levels, which is a critical parameter for reliable pharmaceutical intermediate suppliers who must meet stringent regulatory specifications for residual metals and organic impurities.
How to Synthesize Benzyl Alcohol Efficiently
The synthesis of benzyl alcohol and its derivatives via this patented route is designed for operational simplicity and robustness, making it highly attractive for process development teams looking to optimize their manufacturing workflows. The protocol begins with the mixing of the aromatic carboxylic acid and pinacol borane under an inert gas atmosphere, followed by the precise addition of the n-butyllithium catalyst. The reaction is allowed to stir at room temperature for a short duration, after which the solvent is removed, and the crude borate ester is subjected to hydrolysis. This streamlined approach eliminates the need for complex temperature ramping or high-pressure reactors, significantly reducing the capital expenditure required for implementation. For a detailed breakdown of the standardized synthetic steps, including specific molar ratios and workup procedures, please refer to the guide below.
- Mix aromatic carboxylic acid and pinacol borane in an inert atmosphere, then add 0.5 mol% n-butyllithium catalyst.
- Stir the reaction mixture at room temperature for 40-50 minutes to complete the hydroboration.
- Add silica gel and methanol, heat at 50°C for 2 hours to hydrolyze the borate ester into the final alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this n-butyllithium catalyzed process translates into tangible strategic benefits that directly impact the bottom line and operational resilience. The shift from expensive, hard-to-source transition metal catalysts to a ubiquitous commercial reagent like n-butyllithium fundamentally alters the cost structure of the synthesis. By removing the dependency on proprietary ligands and precious metals, manufacturers can achieve substantial cost savings in raw material procurement while simultaneously mitigating the risks associated with supply chain disruptions for specialized chemicals. Furthermore, the mild reaction conditions imply a drastic reduction in energy consumption, as there is no need for prolonged heating or high-pressure maintenance, contributing to a more sustainable and cost-effective manufacturing footprint.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and complex ligands results in a direct decrease in material costs. Additionally, the short reaction time of 40 to 50 minutes compared to the 24 hours required by older methods significantly increases reactor throughput, allowing for more batches to be produced in the same timeframe without additional capital investment. The simplified workup procedure also reduces labor costs and solvent usage, further enhancing the overall economic efficiency of the process.
- Enhanced Supply Chain Reliability: Utilizing commercial n-butyllithium, a standard reagent available from multiple global suppliers, ensures a stable and secure supply chain. This contrasts sharply with custom-synthesized catalysts that may have long lead times or single-source dependencies. The robustness of the reaction across various substrates means that the same process infrastructure can be used to produce a wide range of alcohol intermediates, providing flexibility to respond quickly to changing market demands.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to commercial production without the need for specialized high-pressure equipment. The use of mild conditions and the absence of heavy metal residues simplify waste treatment and disposal, ensuring compliance with increasingly stringent environmental regulations. This ease of scale-up reduces the time-to-market for new products and lowers the barrier for commercializing complex organic molecules.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydroboration technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production lines.
Q: What are the safety advantages of using n-butyllithium over LiAlH4 for this reduction?
A: Unlike Lithium Aluminum Hydride (LiAlH4), which poses significant safety risks due to its violent reactivity with moisture and requires strict anhydrous handling, the n-butyllithium catalyzed system operates under much milder conditions. The reaction proceeds at room temperature with low catalyst loading, significantly reducing thermal hazards and operational complexity.
Q: How does this method improve cost efficiency compared to transition metal catalysts?
A: Traditional transition metal systems often require expensive, difficult-to-synthesize ligands and high-temperature conditions. This patented method utilizes commercial n-butyllithium at only 0.5 mol% loading, eliminating the need for costly precious metals and complex ligand synthesis, thereby drastically lowering raw material costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. It features a simple workup procedure involving silica gel and methanol hydrolysis, avoids high-pressure equipment, and achieves high yields (>90%) across a wide range of substrates, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the n-butyllithium mediated hydroboration described in CN109467498B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of high-purity pharmaceutical intermediates we deliver meets the exacting standards required by the global healthcare industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this efficient route can optimize your budget. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the market with our reliable supply of high-quality alcohol compounds.
