Advanced Palladium-Catalyzed Synthesis of High-Purity Quinoline Derivatives for Global Pharmaceutical Supply Chains
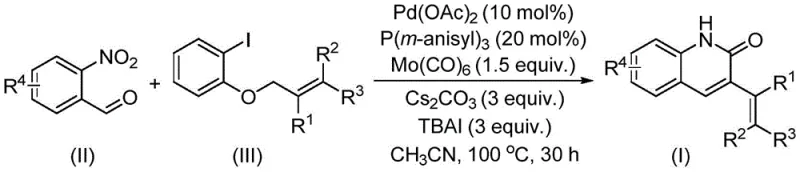
The Chinese patent CN114478375A introduces a groundbreaking palladium-catalyzed reductive aminocarbonylation process that fundamentally transforms the synthesis of pharmacologically significant quinoline derivatives. This innovative methodology addresses critical limitations in traditional approaches by utilizing o-nitrobenzaldehyde as both nitrogen source and formyl donor—a dual functionality that eliminates multi-step synthetic sequences previously required to construct these complex heterocyclic frameworks. The process demonstrates exceptional versatility through its broad substrate scope accommodating diverse functional groups including halogens and alkyl substituents while maintaining high reaction efficiency under mild conditions. Crucially, this advancement directly responds to industry demands for more sustainable pharmaceutical intermediate production by replacing hazardous electrophiles with naturally derived allyl aryl ethers that offer superior handling characteristics and reduced environmental impact. The patent establishes a new paradigm in heterocyclic chemistry by demonstrating how strategic catalyst selection—specifically palladium acetate with tris(3-methoxyphenyl)phosphine—enables previously unattainable transformations while maintaining operational simplicity suitable for industrial implementation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to quinoline derivatives typically rely on harsh reaction conditions involving toxic transition metal catalysts or expensive stoichiometric reagents that generate significant waste streams requiring complex purification protocols. Conventional carbonylation methodologies predominantly employ activated electrophiles like allyl chlorides or acetates which present substantial handling hazards due to their moisture sensitivity and corrosive nature—creating major obstacles for large-scale pharmaceutical manufacturing where safety and environmental compliance are paramount concerns. Furthermore, existing processes often suffer from narrow functional group tolerance necessitating extensive protection/deprotection steps that dramatically increase production costs and reduce overall yield through multiple intermediate isolations. The reliance on specialized nitrogen sources also introduces additional synthetic complexity and impurity profiles that complicate regulatory approval pathways for active pharmaceutical ingredients. Most critically for supply chain managers, these conventional approaches demonstrate poor scalability due to exothermic reaction profiles and stringent temperature control requirements that become increasingly difficult to maintain when transitioning from laboratory to commercial production volumes.
The Novel Approach
This patented methodology overcomes these limitations through an elegant palladium-catalyzed reductive aminocarbonylation process that leverages o-nitrobenzaldehyde as a dual-functionality reagent—simultaneously providing both the nitrogen atom and formyl group required for quinoline ring formation—thereby eliminating the need for separate nitrogen sources and reducing synthetic steps by approximately one-third compared to conventional routes. The strategic use of allyl aryl ethers as electrophiles represents a significant advancement over traditional halide-based systems; these compounds offer superior stability during storage and handling while maintaining excellent reactivity under optimized catalytic conditions. The carefully engineered catalyst system comprising palladium acetate with tris(3-methoxyphenyl)phosphine ligand and molybdenum carbonyl co-catalyst operates efficiently at moderate temperatures of exactly 100°C without requiring specialized equipment or hazardous reagents. This approach demonstrates remarkable functional group tolerance across diverse substrates including fluorinated and methoxylated derivatives while consistently delivering high yields between approximately seventy percent and ninety-nine percent as documented in the patent examples—establishing a robust foundation for reliable commercial production of these critical pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Reductive Aminocarbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the carbon-iodine bond of allyl aryl ether substrates—a step facilitated by the electron-rich tris(3-methoxyphenyl)phosphine ligand which stabilizes the active Pd(II) species while preventing premature catalyst decomposition. This generates an organopalladium intermediate that undergoes migratory insertion with carbon monoxide derived from molybdenum carbonyl decomposition under thermal conditions at precisely controlled temperatures. The key innovation lies in the dual role of o-nitrobenzaldehyde which first participates in nitro group reduction through palladium-mediated hydrogen transfer from the solvent system before undergoing condensation with the acylpalladium intermediate to form the critical imine precursor. Subsequent intramolecular cyclization followed by rearomatization completes the quinoline ring formation with concomitant release of palladium(0) back into the catalytic cycle—this elegant sequence avoids stoichiometric oxidants or reductants while maintaining high atom economy throughout the transformation.
Impurity control is achieved through multiple synergistic mechanisms inherent to this catalytic system; the mild reaction conditions prevent common side reactions such as over-reduction or polymerization that plague traditional methods using stronger reducing agents. The precise stoichiometric balance between cesium carbonate base and tetrabutylammonium iodide additive creates an optimal microenvironment that suppresses hydrolysis pathways while promoting selective imine formation at the ortho position relative to the nitro group. Crucially, the patent demonstrates how substituent effects on both coupling partners can be strategically manipulated to direct regioselectivity—electron-donating groups on the aryl ether component enhance nucleophilicity at specific positions while halogen substituents on the aldehyde component modulate electrophilicity through resonance effects. This level of mechanistic understanding enables precise control over impurity profiles without requiring additional purification steps beyond standard column chromatography as documented in the experimental section.
How to Synthesize Quinoline Derivatives Efficiently
This patented methodology provides an industrially viable pathway for producing high-purity quinoline intermediates through a carefully optimized catalytic process that addresses longstanding challenges in heterocyclic synthesis. The procedure leverages readily available starting materials under precisely controlled conditions to deliver consistent results across diverse substrate combinations while maintaining operational simplicity suitable for commercial manufacturing environments. Detailed standardized synthesis steps are provided below to ensure reliable implementation across different production scales while maintaining stringent quality specifications required by pharmaceutical clients.
- Precisely combine palladium acetate catalyst with tris(3-methoxyphenyl)phosphine ligand and molybdenum carbonyl co-catalyst in acetonitrile solvent under inert atmosphere
- Introduce o-nitrobenzaldehyde as dual nitrogen/formyl source alongside allyl aryl ether substrate at controlled stoichiometric ratios
- Execute reaction at precisely maintained temperature of 100°C for optimized duration followed by silica gel filtration and column chromatography purification
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points associated with traditional intermediate production methods. The process eliminates reliance on hazardous or specialized reagents while utilizing commercially available starting materials that benefit from established global supply networks—significantly enhancing sourcing flexibility and reducing vulnerability to single-source dependencies that frequently disrupt pharmaceutical manufacturing pipelines.
- Cost Reduction in Manufacturing: The elimination of multi-step protection/deprotection sequences combined with simplified purification protocols substantially reduces production costs through decreased solvent consumption and lower energy requirements during processing. The use of cost-effective palladium acetate catalyst at optimized loadings minimizes precious metal consumption while maintaining high turnover numbers—this strategic catalyst selection directly translates to significant operational savings without compromising product quality or yield consistency.
- Enhanced Supply Chain Reliability: By utilizing naturally derived allyl aryl ethers instead of moisture-sensitive halide electrophiles, this process enables more stable raw material inventories with extended shelf lives—reducing waste from material degradation during storage and transportation. The broad functional group tolerance allows manufacturers to maintain consistent production schedules even when facing fluctuations in specific substrate availability since multiple alternative starting materials can achieve equivalent results without process revalidation.
- Scalability and Environmental Compliance: The mild reaction conditions operating at standard atmospheric pressure eliminate requirements for specialized high-pressure equipment while generating minimal waste streams through high atom economy—this facilitates straightforward scale-up from laboratory to commercial production volumes without major capital investments. The simplified purification process using standard column chromatography techniques significantly reduces solvent waste compared to traditional methods requiring multiple crystallization steps—aligning with increasingly stringent environmental regulations while lowering disposal costs across global manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of the patented methodology's implementation parameters and performance characteristics as documented in CN114478375A. These insights reflect practical experience from scaling similar catalytic processes within our manufacturing facilities while maintaining compliance with global regulatory standards.
Q: Why does this method outperform conventional approaches for synthesizing quinoline derivatives?
A: This process uniquely utilizes o-nitrobenzaldehyde as dual nitrogen/formyl source while employing allyl aryl ethers—previously underutilized electrophiles—resulting in superior functional group tolerance and elimination of multi-step protection/deprotection sequences required in traditional methods.
Q: How does the catalytic system ensure high purity without expensive purification steps?
A: The palladium/tris(3-methoxyphenyl)phosphine/molybdenum carbonyl system enables direct reductive aminocarbonylation with minimal byproducts; combined with simple filtration and standard column chromatography as described in the patent's post-processing protocol.
Q: What makes this process suitable for commercial scale-up in pharmaceutical manufacturing?
A: The method uses readily available starting materials at economical ratios with robust reaction conditions that maintain high yields across diverse substrates—critical for consistent large-scale production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivative Supplier
We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required by global regulatory authorities. Our dedicated technical teams apply rigorous QC labs and advanced analytical capabilities to ensure consistent quality across all batches—providing pharmaceutical clients with complete confidence in supply continuity regardless of volume requirements or regulatory jurisdiction changes. This patented methodology represents exactly the type of innovative chemistry we specialize in transforming from laboratory discovery into reliable commercial manufacturing processes.
Request our Customized Cost-Saving Analysis today to understand how this technology can optimize your specific production requirements; our technical procurement team stands ready to provide detailed COA data and route feasibility assessments tailored to your manufacturing needs.
