Advanced Copper-Catalyzed Synthesis of 3,3'-Disubstituted-2-Indolones for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of novel therapeutic agents has placed 2-indolone (oxindole) scaffolds at the forefront of drug discovery, serving as the core structure for FDA-approved medications such as Sunitinib and Nintedanib. Addressing the critical need for efficient access to these privileged structures, patent CN109956894B discloses a groundbreaking synthetic methodology that directly constructs 3,3'-disubstituted-2-indolone compounds from simple aniline precursors. This innovation represents a paradigm shift from traditional multi-step sequences to a streamlined, copper-catalyzed direct cyclization strategy. By leveraging readily available aniline derivatives and ethyl bromoacetates, this process not only simplifies the synthetic route but also enhances the economic viability of producing high-value pharmaceutical intermediates. The reaction proceeds through a robust catalytic cycle that tolerates a wide array of functional groups, ensuring versatility for medicinal chemists designing next-generation kinase inhibitors and other bioactive molecules.
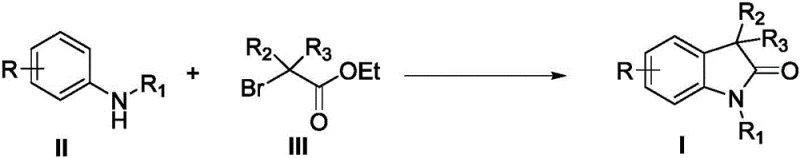
For procurement managers and supply chain directors, the implications of this technology are profound. The ability to bypass complex pre-functionalization steps translates directly into reduced lead times and lower inventory costs for starting materials. Unlike legacy methods that require the synthesis of specialized N-arylpropenamides or malonates, this approach utilizes commodity chemicals that are globally sourced and price-stable. Furthermore, the use of earth-abundant copper catalysts instead of precious metals like palladium drastically lowers the cost of goods sold (COGS), making the final active pharmaceutical ingredient (API) more competitive in the global market. This method stands as a testament to how process chemistry innovation can drive substantial cost reduction in pharmaceutical intermediate manufacturing while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,3-disubstituted indolones has been plagued by inefficiencies inherent to classical organic synthesis strategies. Conventional routes often necessitate the preparation of highly functionalized starting materials, such as N-arylpropenamides, alkynamides, or specific haloalkylamides, which themselves require multiple synthetic steps to generate. These multi-step sequences not only accumulate waste and increase solvent consumption but also introduce significant yield losses at each stage, ultimately depressing the overall throughput of the manufacturing process. Additionally, many established methods rely on expensive transition metal catalysts or harsh reaction conditions that pose safety risks and complicate waste treatment protocols. The reliance on precious metals further exacerbates cost volatility, as the price of catalysts like palladium can fluctuate wildly based on geopolitical factors, creating uncertainty in long-term supply contracts and budget forecasting for large-scale production campaigns.
The Novel Approach
In stark contrast, the methodology described in CN109956894B offers a direct, atom-economical pathway that circumvents these historical bottlenecks. By employing a simple coupling of substituted anilines with ethyl bromoacetates, the invention achieves the construction of the indolone core in a single operational step. This telescoped approach eliminates the need for isolating unstable or costly intermediates, thereby streamlining the workflow and reducing the physical footprint required for manufacturing. The reaction utilizes a cost-effective copper catalytic system, specifically copper(II) acetate paired with bipyridine ligands, which provides high turnover numbers and excellent selectivity. This novel route not only accelerates the timeline from benchtop discovery to pilot plant validation but also aligns perfectly with green chemistry principles by minimizing waste generation and energy consumption, offering a sustainable solution for the modern chemical industry.
Mechanistic Insights into Copper-Catalyzed Cyclization
The success of this transformation hinges on the precise orchestration of the copper catalytic cycle, which facilitates the activation of the C-H bond and subsequent ring closure. The mechanism likely initiates with the coordination of the copper(II) species to the nitrogen atom of the aniline substrate, enhancing its nucleophilicity towards the electrophilic carbon of the bromoacetate. Following the initial alkylation, an intramolecular oxidative cyclization occurs, driven by the redox properties of the copper center and the presence of the base, potassium carbonate. The 2,2'-bipyridine ligand plays a crucial role in stabilizing the copper oxidation states throughout the cycle, preventing catalyst deactivation and ensuring consistent performance across extended reaction times. This mechanistic robustness allows the reaction to proceed efficiently even with sterically hindered substrates, a common challenge in heterocyclic synthesis that often leads to incomplete conversions or side reactions in less optimized systems.
From an impurity control perspective, the specificity of this copper-mediated pathway is exceptional. The reaction conditions are tuned to favor the formation of the five-membered lactam ring over potential competing pathways such as N-alkylation without cyclization or polymerization of the bromoacetate. The use of acetonitrile as the solvent provides an optimal polarity balance that solubilizes the ionic intermediates while maintaining the stability of the organic substrates. Furthermore, the thermal window of 110-130°C is sufficient to overcome the activation energy barrier for cyclization without inducing thermal degradation of the sensitive indolone product. This high degree of chemoselectivity ensures that the crude reaction mixture contains minimal byproducts, simplifying downstream purification and enabling the isolation of products with purity levels that meet stringent regulatory requirements for pharmaceutical applications.
How to Synthesize 3,3'-Disubstituted-2-Indolones Efficiently
To implement this synthesis effectively, operators must adhere to precise stoichiometric ratios and thermal profiles as validated in the patent examples. The process begins with the careful charging of the reactor with the aniline derivative and the bromoacetate building block in a molar ratio of approximately 1:2, ensuring an excess of the alkylating agent to drive the equilibrium forward. The addition of the copper catalyst and ligand must be performed under controlled atmospheric conditions to prevent premature oxidation or moisture ingress, which could inhibit catalytic activity. Once the reagents are combined with the base and solvent, the system is heated to a target temperature of 120°C, a parameter that has been empirically determined to maximize yield while minimizing reaction time. Monitoring the reaction progress via TLC or GC is essential to determine the exact endpoint, typically achieved within 12 hours, after which standard workup procedures involving vacuum concentration and silica gel chromatography yield the pure target compound.
- Charge a reactor with diphenylamine derivative, ethyl bromoacetate derivative, Cu(OAc)2 catalyst, 2,2'-bipyridine ligand, and K2CO3 base.
- Add acetonitrile solvent and heat the mixture to 120°C for 12 hours under sealed conditions to drive the cyclization.
- Concentrate the reaction mixture under vacuum and purify the crude residue via silica gel column chromatography to isolate the target indolone.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, this synthetic route offers compelling strategic advantages that extend beyond mere chemical efficiency. The substitution of precious metal catalysts with abundant copper salts represents a direct and significant reduction in raw material expenditure, insulating the supply chain from the volatility of the platinum group metals market. Moreover, the simplicity of the reagent profile means that sourcing is no longer a bottleneck; anilines and bromoacetates are commodity chemicals produced by numerous global vendors, ensuring redundancy in the supply base and mitigating the risk of single-source dependency. This diversification of supply sources is critical for maintaining uninterrupted production schedules, especially in the face of global logistical disruptions or regional manufacturing shutdowns.
- Cost Reduction in Manufacturing: The elimination of multi-step precursor synthesis drastically reduces labor, utility, and solvent costs associated with intermediate isolation and purification. By consolidating the synthesis into a single pot, manufacturers can achieve higher throughput per batch without expanding facility capacity. The use of inexpensive copper catalysts further lowers the variable cost per kilogram of product, allowing for more competitive pricing strategies in contract manufacturing negotiations. Additionally, the simplified workup procedure reduces the consumption of silica gel and eluents during purification, contributing to overall operational expense savings.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that production can be scaled rapidly to meet surging demand without long lead times for custom synthesis of exotic reagents. The robustness of the reaction conditions allows for flexibility in manufacturing locations, as the process does not require specialized high-pressure equipment or cryogenic cooling systems that limit where a reaction can be run. This geographical flexibility empowers supply chain managers to optimize logistics networks and reduce transportation costs by manufacturing closer to end markets or API formulation sites.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional methods that utilize stoichiometric amounts of toxic reagents or generate heavy metal contamination. The copper catalyst, while a heavy metal, is used in low loading and can potentially be recovered or treated more easily than palladium residues, simplifying wastewater treatment compliance. The high atom economy of the direct cyclization means less organic waste is sent for incineration, aligning with corporate sustainability goals and reducing the carbon footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a realistic framework for evaluating the feasibility of this route for your specific project needs. Understanding these nuances is essential for R&D teams planning scale-up activities and procurement officers assessing vendor capabilities.
Q: What are the key advantages of this copper-catalyzed method over traditional transition metal routes?
A: This method eliminates the need for expensive palladium or rhodium catalysts and complex pre-functionalized substrates like N-arylpropenamides. It utilizes inexpensive copper salts and readily available anilines, significantly reducing raw material costs and simplifying the supply chain.
Q: Is this synthesis method scalable for industrial production of API intermediates?
A: Yes, the process operates under relatively mild thermal conditions (110-130°C) using common organic solvents like acetonitrile. The robustness of the copper catalytic system and the high yields reported across diverse substrates indicate strong potential for commercial scale-up from kilogram to multi-ton batches.
Q: What is the substrate scope for the aniline and bromoacetate components?
A: The methodology demonstrates excellent tolerance for various substituents. Anilines can bear electron-donating or withdrawing groups such as halogens, alkyls, and alkoxy groups. Similarly, the bromoacetate component accepts diverse R-groups including alkyl, allyl, and ester functionalities, allowing for the synthesis of a wide library of 3,3'-disubstituted derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Disubstituted-2-Indolones Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with state-of-the-art reactors capable of handling the thermal and pressure requirements of copper-catalyzed cyclizations, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards for pharmaceutical intermediates.
We invite you to leverage our technical expertise to optimize this synthesis for your specific application. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this route or need specific COA data to validate the quality of our pilot batches, our team is ready to assist. Please contact our technical procurement team today to request route feasibility assessments and discuss how we can support your supply chain with high-purity, cost-effective 3,3'-disubstituted-2-indolones.
