Advanced Manufacturing of (5R)-5-Hydroxy Triptolide via Selective Chromium Oxidation
The pharmaceutical industry has long recognized the immense therapeutic potential of triptolide derivatives, particularly for their potent anti-inflammatory and anti-tumor properties. However, the clinical translation of these compounds has been historically hindered by severe toxicity profiles and poor water solubility. A significant breakthrough in addressing these limitations is found in the chemical modification of the triptolide scaffold, specifically through the introduction of a hydroxyl group at the C-5 position. Patent CN110759929B discloses a robust and industrially viable preparation method for (5R)-5-hydroxy triptolide that fundamentally shifts the manufacturing paradigm away from hazardous legacy processes. This technical insight report analyzes the novel synthetic route, which replaces the notorious selenium dioxide oxidation with a safer, chromium-based catalytic system mediated by N-hydroxysuccinimide. For R&D directors and procurement strategists, this patent represents a critical opportunity to secure a supply chain for high-purity pharmaceutical intermediates that is both economically efficient and environmentally compliant.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-hydroxy triptolide derivatives relied heavily on direct oxidation using selenium dioxide (SeO2), a reagent known for its extreme toxicity and environmental persistence. As illustrated in the prior art reaction pathways, this conventional approach suffers from multiple critical deficiencies that render it unsuitable for modern large-scale manufacturing. The use of SeO2 not only introduces severe occupational health risks requiring expensive containment infrastructure but also generates selenium-containing waste streams that are notoriously difficult and costly to treat. Furthermore, the direct oxidation of triptolide lacks sufficient stereocontrol, often producing a racemic mixture or a difficult-to-separate pair of chiral isomers at the C-5 position. This necessitates downstream chiral resolution steps, such as preparative column chromatography, which drastically reduce the overall process yield to approximately 30% and inflate production costs due to excessive solvent consumption and low throughput efficiency.
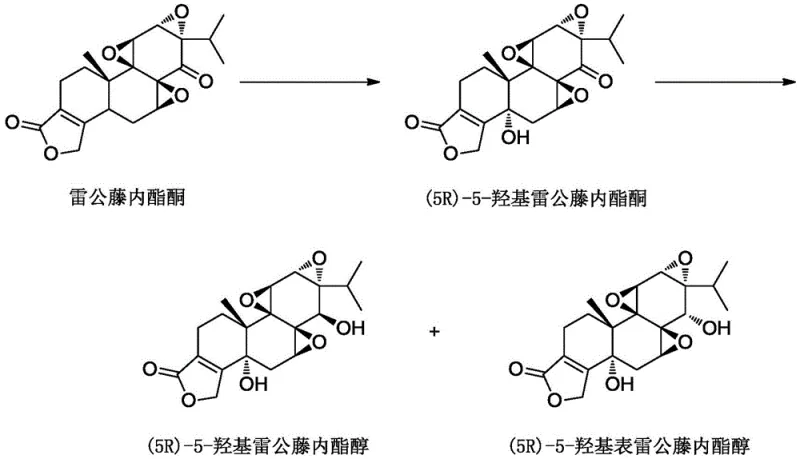
The Novel Approach
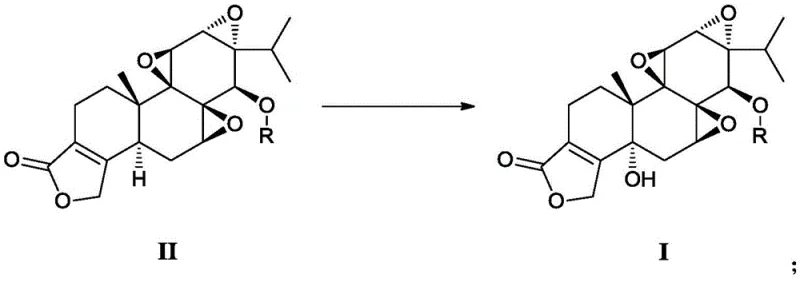
In stark contrast to the hazardous legacy methods, the invention disclosed in CN110759929B introduces a sophisticated three-step strategy centered on a protection-oxidation-deprotection sequence. This novel approach begins by masking the reactive 14-hydroxyl group of triptolide through acylation, creating a stable intermediate that directs subsequent chemical transformations. By shielding this position, the process prevents unwanted side reactions and sets the stage for highly selective oxidation at the C-5 position. The core innovation lies in the use of chromium trioxide (CrO3) in conjunction with N-hydroxysuccinimide (HOSu) or similar catalysts, which enables precise allylic oxidation without compromising the integrity of the sensitive epoxide and lactone rings. This method completely bypasses the need for toxic selenium reagents and, crucially, achieves high stereoselectivity for the desired (5R) configuration, thereby eliminating the need for costly and yield-limiting chiral resolution steps entirely.
Mechanistic Insights into CrO3-HOSu Mediated Selective Oxidation
The heart of this technological advancement is the mechanistic interplay between the chromium oxidant and the organic catalyst during the conversion of the protected intermediate. In the absence of a mediator, chromium trioxide can be overly aggressive, leading to over-oxidation or degradation of the complex diterpene skeleton. However, the addition of N-hydroxysuccinimide (HOSu) creates a synergistic catalytic cycle that moderates the oxidation potential. The HOSu likely functions by generating an active oxoammonium or radical species in situ, which selectively abstracts a hydrogen atom from the allylic C-5 position. This radical intermediate is then trapped by the chromium species to form the hydroxyl group with high fidelity. The presence of the protecting group at the C-14 position is equally vital, as it sterically and electronically biases the reaction towards the 5-position, ensuring that the oxidation occurs exclusively where intended. This level of control is what allows the process to achieve yields significantly superior to the uncontrolled direct oxidation methods of the past.
Furthermore, the impurity profile of this reaction is markedly cleaner than that of selenium-based routes. The specific solvent system employed, typically a mixture of acetonitrile and water, facilitates the solubility of both the organic substrate and the inorganic oxidant while maintaining a homogeneous reaction environment that promotes consistent kinetics. The use of mild reaction temperatures, generally ranging from 20°C to 50°C, further suppresses the formation of thermal degradation by-products. Post-reaction workup involves simple extraction and washing with sodium bisulfite to quench excess oxidant, a standard and scalable unit operation. This mechanistic precision ensures that the resulting protected 5-hydroxy intermediate possesses high purity, which simplifies the final deprotection step and reduces the burden on downstream purification processes, ultimately delivering a final API intermediate that meets stringent regulatory specifications for heavy metals and residual solvents.
How to Synthesize (5R)-5-Hydroxy Triptolide Efficiently
The practical execution of this synthesis involves a logical progression of unit operations designed for scalability and reproducibility. The process initiates with the acylation of triptolide using acetic anhydride or benzoyl chloride in the presence of a base like pyridine, yielding the protected ester in high conversion. This intermediate is then subjected to the critical oxidation step described previously, utilizing the CrO3-HOSu system in an aqueous acetonitrile medium. Following the oxidation, the crude protected product undergoes a mild hydrolysis to reveal the free hydroxyl group. The detailed standardized synthetic steps, including specific molar ratios, temperature controls, and workup procedures validated in the patent examples, are outlined below to guide process engineers in replicating this high-yield route.
- Protect the 14-hydroxyl group of Triptolide using acetic anhydride or benzoyl chloride to form a stable ester intermediate.
- Perform selective oxidation at the C-5 position using Chromium Trioxide (CrO3) mediated by N-hydroxysuccinimide in an acetonitrile-water solvent system.
- Execute mild hydrolysis using hydrazine hydrate or lithium hydroxide at low temperatures to remove the protecting group and yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented methodology offers transformative advantages in terms of cost structure and operational reliability. The most immediate impact is the drastic reduction in raw material hazards; by eliminating selenium dioxide, companies avoid the substantial costs associated with handling, storing, and disposing of highly toxic heavy metal wastes. This shift not only lowers direct operational expenditures but also mitigates regulatory risks and insurance premiums associated with hazardous chemical manufacturing. Additionally, the removal of chiral resolution steps translates directly into increased throughput capacity, as facilities no longer need to dedicate significant time and solvent resources to separating enantiomers. This efficiency gain allows for faster batch turnover and a more responsive supply chain capable of meeting fluctuating market demands for this valuable oncology intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic route and the improvement in overall yield. By avoiding the 30% total yield ceiling imposed by the old selenium method and its requisite chiral separation, the new route potentially doubles the output per kilogram of starting triptolide. The elimination of expensive chiral chromatography columns and the vast quantities of solvents required for such separations results in substantial cost savings. Furthermore, the reagents used, such as chromium trioxide and N-hydroxysuccinimide, are commodity chemicals with stable pricing and widespread availability, ensuring that the cost of goods sold remains predictable and competitive in the global marketplace.
- Enhanced Supply Chain Reliability: Supply continuity is significantly bolstered by the robustness of the new reaction conditions. The tolerance of the CrO3-HOSu system to minor variations in temperature and stoichiometry makes it less prone to batch failures compared to the sensitive selenium oxidations. Moreover, the reliance on commercially available starting materials and catalysts reduces the risk of supply bottlenecks that often plague specialized reagent chains. The ability to produce the intermediate in high purity without complex purification trains means that manufacturing slots can be utilized more effectively, reducing lead times for high-purity pharmaceutical intermediates and ensuring a steady flow of material for downstream drug development programs.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is far superior for commercial scale-up. The absence of selenium eliminates the need for specialized wastewater treatment protocols required to reduce soluble selenium to insoluble forms before discharge. The waste streams generated are primarily organic and chromium-based, which are well-understood and manageable within standard pharmaceutical waste treatment frameworks. This compliance advantage accelerates the regulatory approval process for manufacturing sites and aligns with the increasing corporate mandates for green chemistry and sustainable manufacturing practices, making the facility more attractive to top-tier pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of the method for potential licensees and manufacturing partners.
Q: Why is the new CrO3 method preferred over the traditional Selenium Dioxide route?
A: The traditional Selenium Dioxide (SeO2) route poses severe environmental and safety hazards due to selenium toxicity and generates difficult-to-treat waste streams. Furthermore, the SeO2 method often yields a mixture of chiral isomers requiring complex and costly resolution steps. The new CrO3 method described in CN110759929B utilizes a protective group strategy that directs oxidation specifically to the 5R position, eliminating the need for chiral separation and significantly improving process safety and total yield.
Q: What role does N-hydroxysuccinimide (HOSu) play in the oxidation step?
A: N-hydroxysuccinimide acts as a crucial catalytic mediator in the chromium-mediated oxidation. It facilitates the generation of active oxygen species that selectively target the C-5 allylic position while minimizing over-oxidation or degradation of the sensitive epoxide and lactone rings present in the triptolide scaffold. This catalytic effect allows for reduced loading of the primary oxidant (CrO3) and enhances the stereochemical purity of the resulting 5-hydroxy derivative.
Q: How does this process impact the cost of goods for pharmaceutical development?
A: By avoiding the use of highly toxic selenium reagents, manufacturers save significantly on hazardous waste disposal and specialized containment equipment costs. Additionally, the elimination of chiral chromatography steps, which are traditionally required to separate 5R and 5S isomers in older methods, drastically reduces solvent consumption and processing time. The higher overall yield of the protected oxidation route further lowers the effective cost per kilogram of the active pharmaceutical ingredient.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (5R)-5-Hydroxy Triptolide Supplier
As the demand for next-generation anti-cancer and immunosuppressive agents continues to rise, securing a dependable source of high-quality intermediates like (5R)-5-hydroxy triptolide is paramount for pharmaceutical developers. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in complex natural product modification to deliver solutions that bridge the gap between academic innovation and commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the exacting standards required for clinical trial materials and commercial API production.
We invite R&D directors and procurement executives to engage with us to explore how this advanced synthetic route can optimize your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and comprehensive quotations that demonstrate the tangible value of our manufacturing capabilities.
