Advanced Manufacturing of Erlotinib Hydrochloride: A Green Chemistry Approach for Scalable API Production
Advanced Manufacturing of Erlotinib Hydrochloride: A Green Chemistry Approach for Scalable API Production
The pharmaceutical industry is constantly evolving towards greener, more efficient synthetic pathways, particularly for high-value kinase inhibitors like Erlotinib Hydrochloride. Patent CN102584719A introduces a significant technological breakthrough in the preparation of this critical oncology drug, offering a robust alternative to legacy manufacturing processes. This innovation focuses on utilizing 2-nitro-4,5-bis(2-methoxyethoxy)benzonitrile as a key starting material, streamlining the synthesis through a novel reduction and cyclization strategy. For R&D directors and procurement managers alike, this patent represents a pivotal shift away from hazardous chlorination chemistry towards a more sustainable and operationally feasible protocol. By addressing the inherent limitations of traditional routes, this technology promises to enhance supply chain stability while maintaining the stringent purity standards required for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
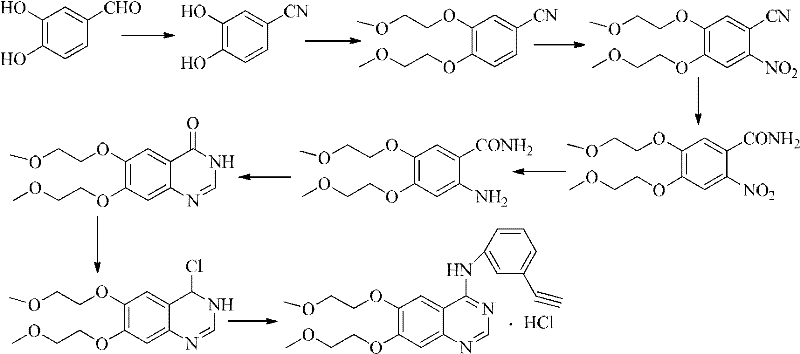
Historically, the industrial synthesis of Erlotinib has relied heavily on aggressive chlorination steps that pose significant environmental and operational challenges. As illustrated in the background technology of the patent, conventional routes often involve the conversion of quinazoline precursors using potent chlorinating reagents such as phosphorus oxychloride (POCl3), thionyl chloride (SOCl2), or oxalyl chloride. These reagents are not only highly corrosive and toxic but also generate substantial amounts of hazardous waste that require complex and costly disposal procedures. Furthermore, the use of such harsh chemicals often leads to the formation of difficult-to-remove chlorinated impurities, complicating the downstream purification process and potentially impacting the final drug safety profile. The reliance on these forbidden or restricted substances creates a bottleneck for manufacturers aiming to comply with increasingly strict global environmental regulations.
The Novel Approach
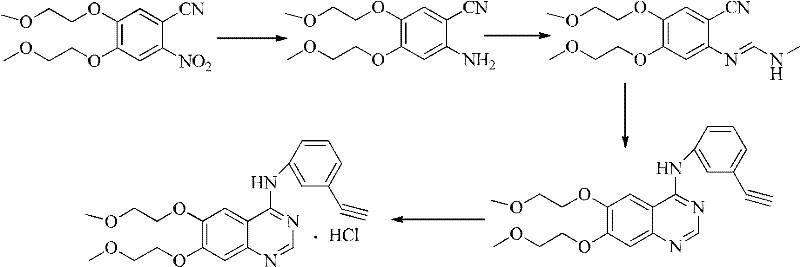
In stark contrast, the methodology disclosed in CN102584719A circumvents these issues by employing a direct cyclization strategy that eliminates the need for chlorinating agents entirely. The process begins with the reduction of the nitro group in 2-nitro-4,5-bis(2-methoxyethoxy)benzonitrile to an amino group, followed by a condensation reaction with DMF-DMA or a Schiff base of 3-ethynylaniline. This approach allows for the construction of the quinazoline core under much milder conditions, typically ranging between 120°C and 140°C. By avoiding the formation of reactive acid chlorides, the process significantly reduces the risk of equipment corrosion and simplifies the workup procedure. This novel pathway not only shortens the overall reaction sequence but also aligns perfectly with the principles of green chemistry, making it an attractive option for reliable erlotinib hydrochloride supplier networks seeking to modernize their production capabilities.
Mechanistic Insights into Nitro Reduction and Cyclization
The core of this technological advancement lies in the efficient transformation of the nitro precursor into the reactive amine species, which serves as the nucleophile for the subsequent ring closure. The patent details the use of versatile reducing agents such as V-Brite B, zinc powder, or iron powder in an aqueous acidic medium. This reduction step is critical as it must proceed with high selectivity to avoid affecting other functional groups, such as the nitrile or the ether linkages. Once the 2-amino-4,5-bis(2-methoxyethoxy)benzonitrile is formed, it undergoes a condensation with DMF-DMA to generate an enamine intermediate. This intermediate is highly reactive towards nucleophilic attack by the amine group of 3-ethynylaniline. The subsequent intramolecular cyclization forms the quinazoline ring system directly, bypassing the need for a separate chlorination-amination sequence. This mechanistic elegance ensures that the carbon skeleton is assembled with minimal side reactions, thereby enhancing the overall atom economy of the synthesis.
From an impurity control perspective, this mechanism offers distinct advantages over traditional methods. In conventional chlorination routes, the presence of excess POCl3 or SOCl2 can lead to over-chlorination or the formation of stable chloro-impurities that are structurally similar to the target molecule, making them notoriously difficult to separate via crystallization or chromatography. By contrast, the byproducts generated in this new route are primarily organic amines or simple salts that are far more soluble in aqueous washes or can be easily removed during the recrystallization of the free base. The absence of halogenated solvents and reagents also means that the residual solvent profile of the final API is cleaner, reducing the burden on analytical quality control teams. This inherent purity advantage is crucial for meeting the rigorous specifications demanded by regulatory bodies for oncology therapeutics.
How to Synthesize Erlotinib Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control during the cyclization phase and pH adjustment during the isolation steps. The patent outlines a clear operational path where the reduction is conducted at moderate temperatures (50-70°C), followed by a reflux step for the DMF-DMA reaction. The subsequent coupling with 3-ethynylaniline is performed at elevated temperatures (120-140°C) to drive the cyclization to completion. Detailed standard operating procedures for scaling this reaction from laboratory to pilot plant are essential to maintain consistency. For a comprehensive guide on the specific stoichiometry, solvent choices, and workup protocols validated in the patent examples, please refer to the technical breakdown below.
- Reduce 2-nitro-4,5-bis(2-methoxyethoxy)benzonitrile to the corresponding amino derivative using V-Brite B, zinc powder, or iron powder under acidic conditions.
- React the resulting amine with DMF-DMA to form an intermediate, followed by cyclization with 3-ethynylaniline at 120-140°C.
- Purify the crude Erlotinib base and convert to the hydrochloride salt using hydrochloric acid or HCl gas in an alcoholic solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology translates into tangible strategic benefits beyond mere chemical efficiency. The elimination of hazardous chlorinating agents removes a significant logistical and compliance burden, as these substances often require special handling permits and dedicated storage facilities. By switching to benign reagents like iron powder or zinc, manufacturers can drastically simplify their raw material sourcing and reduce the overhead costs associated with environmental health and safety (EHS) management. This shift facilitates cost reduction in pharmaceutical intermediates manufacturing by lowering waste disposal fees and minimizing the need for specialized corrosion-resistant reactor linings. Furthermore, the shorter reaction sequence implies fewer unit operations, which directly correlates to reduced energy consumption and shorter batch cycle times, ultimately enhancing the throughput capacity of existing production assets.
- Cost Reduction in Manufacturing: The removal of expensive and toxic reagents like POCl3 and SOCl2 significantly lowers the direct material costs per kilogram of product. Additionally, the simplified workup procedure reduces the consumption of solvents and neutralizing agents required for quenching aggressive acids. This leaner process flow minimizes utility usage and labor hours, leading to substantial cost savings that can be passed down the supply chain or reinvested into further process optimization initiatives.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals such as iron powder, zinc, and common organic solvents ensures a stable supply of raw materials, unlike specialized chlorinating agents which may be subject to regulatory restrictions or supply volatility. This robustness reduces lead time for high-purity API intermediates by mitigating the risk of production stoppages due to reagent shortages. Manufacturers can maintain continuous production schedules with greater confidence, ensuring consistent delivery to downstream formulation partners.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of noxious gas evolution make this process inherently safer and easier to scale up to commercial volumes. Facilities can achieve commercial scale-up of complex kinase inhibitors without requiring major capital expenditure on scrubber systems or hazardous waste incineration units. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening environmental laws but also enhances the corporate sustainability profile, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new preparation technology is vital for stakeholders evaluating its potential for integration into their supply chains. The following questions address common inquiries regarding the practical implementation, safety profile, and quality attributes of the process described in CN102584719A. These insights are derived directly from the patent's experimental data and beneficial effect analysis, providing a factual basis for decision-making.
Q: What are the primary advantages of this new Erlotinib synthesis route over conventional methods?
A: The primary advantage is the elimination of toxic chlorinating agents such as POCl3 and SOCl2, which are environmentally hazardous and complicate industrial waste treatment. This new route utilizes milder conditions and avoids the formation of chlorinated byproducts.
Q: Which reducing agents are suitable for the initial nitro reduction step?
A: The patent specifies several effective reducing agents including V-Brite B, zinc powder, and iron powder. This flexibility allows manufacturers to select the most cost-effective and operationally convenient reagent for their specific facility capabilities.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrial scalability. It features short reaction routes, mild reaction temperatures (120-140°C for cyclization), and avoids hazardous reagents, making it safer and more feasible for ton-scale manufacturing compared to traditional chlorination methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Erlotinib Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the growing global demand for high-quality oncology treatments. Our technical team has thoroughly analyzed the pathway described in CN102584719A and confirmed its viability for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against international pharmacopoeia standards.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us on leveraging this greener synthesis route for Erlotinib Hydrochloride. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional regulatory landscape. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can drive value and reliability in your supply chain.
