Advanced Manufacturing of Riociguat Intermediate via Palladium-Catalyzed Cyanation
Advanced Manufacturing of Riociguat Intermediate via Palladium-Catalyzed Cyanation
The pharmaceutical landscape for treating chronic thromboembolic pulmonary hypertension (CTEPH) has been significantly advanced by the development of Riociguat, a soluble guanylate cyclase stimulator. Central to the efficient production of this life-saving medication is the robust synthesis of its key intermediate, 1-(2-fluorobenzyl)-1H-pyrazolo[3,4-b]pyridin-3-formamidine hydrochloride. Patent CN102491974A introduces a groundbreaking methodology that addresses long-standing inefficiencies in the supply chain of this critical pharmaceutical intermediate. By leveraging a novel sequence starting from 3-iodo-1H-pyrazolo[3,4-b]pyridine, this technology offers a pathway that is not only chemically elegant but also commercially viable for high-volume manufacturing. The structural complexity of the target molecule, as seen in the final drug substance below, demands precision in every synthetic step to ensure purity and safety.
For R&D directors and process chemists, the significance of this patent lies in its departure from legacy routes that were plagued by low throughput and hazardous reagents. The disclosed method optimizes the construction of the pyrazolo[3,4-b]pyridine core, ensuring that the final active pharmaceutical ingredient (API) meets the rigorous quality standards required for cardiovascular therapeutics. As a reliable pharmaceutical intermediate supplier, understanding these nuanced synthetic improvements is crucial for maintaining a competitive edge in the global market. The following analysis dissects the technical merits and commercial implications of this innovative synthesis strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in US20020173514 and related literature (e.g., Chem. Med. Chem. 2009), presented substantial hurdles for industrial adoption. These conventional routes often relied on starting materials that were either prohibitively expensive or difficult to source on a commercial scale, creating bottlenecks in the supply chain. Furthermore, the synthetic efficiency was suboptimal; for instance, the initial two steps in older protocols reportedly achieved a cumulative yield of only 49.9%, which is economically unsustainable for large-scale API production. A critical operational flaw in these legacy methods was the dependence on column chromatography for purification after the second step. In an industrial setting, column chromatography is a major impediment to scalability due to high solvent consumption, slow processing times, and difficulties in waste management. These factors collectively resulted in high manufacturing costs and inconsistent batch quality, limiting the ability to meet the growing global demand for Riociguat.
The Novel Approach
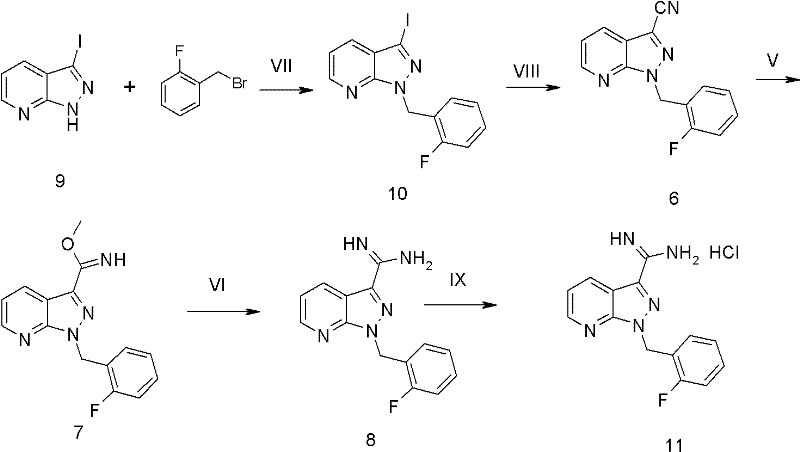
The methodology outlined in Patent CN102491974A represents a paradigm shift in the synthesis of this intermediate, specifically designed to overcome the aforementioned limitations. The new route initiates with 3-iodo-1H-pyrazolo[3,4-b]pyridine, a raw material that is both cheap and readily available, immediately addressing the sourcing issues of previous methods. The synthetic sequence is streamlined into four distinct transformations that avoid the need for chromatographic purification entirely. As illustrated in the reaction scheme below, the process flows logically from N-alkylation to cyanation, followed by amidination and final salt formation. This linear progression minimizes unit operations and reduces the overall environmental footprint of the manufacturing process. By eliminating complex purification steps and utilizing high-yielding reactions, this approach facilitates cost reduction in API manufacturing while ensuring a consistent supply of high-purity material.
Mechanistic Insights into Palladium-Catalyzed Cyanation
A cornerstone of this synthetic strategy is the transformation of the iodo-intermediate (compound 10) into the corresponding nitrile (compound 6) via a palladium-catalyzed cyanation. This step replaces traditional, harsher cyanation methods (such as the Rosenmund-von Braun reaction using copper cyanide) with a more modern and selective catalytic system. The reaction employs a robust catalyst system comprising tetrakis(triphenylphosphine)palladium(0) [Pd(PPh3)4] and 1,1'-bis(diphenylphosphino)ferrocene (dppf) as a ligand. Crucially, the inclusion of zinc powder acts as a scavenger or activator, enhancing the turnover of the catalytic cycle and preventing catalyst deactivation. The use of zinc cyanide [Zn(CN)2] as the cyanide source is particularly advantageous; it is less toxic and easier to handle than alkali metal cyanides, aligning with green chemistry principles. The reaction is conducted in N,N-dimethylacetamide (DMAC) at elevated temperatures ranging from 100°C to 150°C, which provides the necessary activation energy to drive the oxidative addition and reductive elimination steps efficiently.
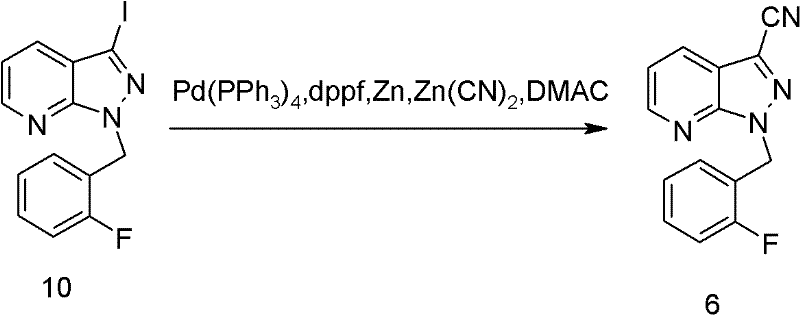
From an impurity control perspective, this catalytic system offers superior selectivity. The specific ligand environment created by dppf stabilizes the palladium center, minimizing side reactions such as homocoupling of the aryl halide or hydrodehalogenation, which are common pitfalls in cross-coupling chemistry. The patent data indicates a yield of 63.7% for this specific transformation, which, when combined with the high efficiency of the subsequent steps, contributes to a highly favorable overall mass balance. For quality assurance teams, the absence of heavy metal contaminants like copper (from traditional methods) simplifies the downstream purification and ensures that the final intermediate meets stringent heavy metal specifications required for pharmaceutical grade materials. This mechanistic refinement is a key driver for the process's scalability and regulatory compliance.
How to Synthesize 1-(2-Fluorobenzyl)-1H-Pyrazolo[3,4-b]Pyridin-3-Formamidine Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly during the cyanation and amidination stages. The process begins with a straightforward N-alkylation at room temperature, followed by the high-temperature cyanation described above. The subsequent conversion of the nitrile to the amidine involves a Pinner-like reaction sequence using sodium methoxide and ammonium chloride in methanol, followed by acidification. This sequence effectively installs the formamidine functionality with high fidelity. The final step involves the precipitation of the hydrochloride salt using hydrogen chloride gas, which serves as both a purification and isolation step. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- React 3-iodo-1H-pyrazolo[3,4-b]pyridine with 2-fluorobenzyl bromide using potassium carbonate in DMF at room temperature to yield compound 10.
- Perform palladium-catalyzed cyanation on compound 10 using zinc cyanide, Pd(PPh3)4, and dppf in DMAC at 120°C to obtain nitrile compound 6.
- Convert nitrile compound 6 to amidine compound 8 using sodium methoxide and ammonium chloride in methanol, followed by reflux with acetic acid.
- Treat compound 8 with hydrogen chloride gas in methyl tert-butyl ether to precipitate the final hydrochloride salt product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits. The primary advantage lies in the drastic simplification of the manufacturing workflow. By removing the need for column chromatography, the process significantly reduces solvent usage and processing time, leading to substantial cost savings in utility and waste disposal. Furthermore, the reliance on cheap and readily available starting materials mitigates the risk of supply disruptions caused by the scarcity of exotic reagents. This stability is crucial for maintaining continuous production schedules and meeting delivery commitments to downstream API manufacturers. The robust nature of the chemistry also implies a lower risk of batch failures, enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The elimination of column chromatography is a major economic driver. Chromatography is capital-intensive and slow; replacing it with crystallization or simple filtration (as done in this process) drastically lowers the cost of goods sold (COGS). Additionally, the use of zinc cyanide instead of more hazardous or expensive cyanating agents reduces raw material costs and safety compliance expenses. The high overall yield reported in the patent examples suggests that less raw material is wasted, further optimizing the cost structure for commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The starting material, 3-iodo-1H-pyrazolo[3,4-b]pyridine, is commercially accessible, reducing dependency on custom synthesis for early-stage precursors. The mild conditions of the first step (room temperature alkylation) allow for flexible scheduling and lower energy consumption compared to cryogenic or high-pressure alternatives. This operational flexibility ensures that production can be ramped up quickly to meet market demand without significant lead time delays, securing the supply chain for critical cardiovascular medications.
- Scalability and Environmental Compliance: The process is inherently designed for scale. The use of standard solvents like DMF, DMAC, and Methanol facilitates easy solvent recovery and recycling. The avoidance of heavy metal catalysts like copper simplifies the wastewater treatment process, ensuring compliance with increasingly strict environmental regulations. The high purity of the final product (reported as 99% in the patent examples) reduces the burden on downstream purification, making this route highly attractive for multi-ton production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method. These insights are derived directly from the experimental data and claims within Patent CN102491974A, providing a clear picture of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their supply chain.
Q: What are the primary advantages of this synthesis route over prior art methods?
A: Unlike previous methods (e.g., US20020173514) which suffered from low yields (approx. 50% for initial steps) and required difficult column chromatography, this patented route utilizes readily available raw materials and achieves significantly higher overall yields without complex purification steps, making it ideal for large-scale production.
Q: What are the critical conditions for the cyanation step?
A: The cyanation step (conversion of compound 10 to 6) requires precise control, utilizing a palladium catalyst system with Pd(PPh3)4 and dppf ligand, zinc powder as an activator, and zinc cyanide as the cyanide source in DMAC solvent at temperatures between 100°C and 150°C.
Q: Is this process suitable for commercial scale-up?
A: Yes, the process is explicitly designed for scalability. It avoids expensive and hard-to-source starting materials, operates under relatively mild conditions for the alkylation step (room temperature), and eliminates the need for column chromatography, which is a major bottleneck in industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(2-Fluorobenzyl)-1H-Pyrazolo[3,4-b]Pyridin-3-Formamidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development and production of life-saving medicines like Riociguat. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our facility is equipped to handle the specific reaction conditions required for this synthesis, including high-temperature palladium-catalyzed reactions and safe handling of cyanide sources.
We invite you to collaborate with us to optimize your supply chain for this vital intermediate. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this patented route can reduce your overall procurement costs. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us be your partner in bringing efficient and affordable cardiovascular therapies to the global market.
