Revolutionizing Amisulpride Intermediate Production: A Deep Dive into Patent CN112521318A
Revolutionizing Amisulpride Intermediate Production: A Deep Dive into Patent CN112521318A
The global pharmaceutical landscape for antipsychotic medications continues to evolve, with Amisulpride remaining a cornerstone therapy for treating schizophrenia and related disorders. At the heart of manufacturing this critical active pharmaceutical ingredient lies the efficient production of its key precursor, methyl 4-amino-5-thiophenyl-2-methoxybenzoate. Recent intellectual property developments, specifically patent CN112521318A filed in March 2021, have introduced a transformative preparation method that addresses long-standing inefficiencies in synthetic chemistry. This patent discloses a novel pathway starting from methyl 4-amino-2-methoxy-5-thiocyanatobenzoate, utilizing a mild salt-forming neutralization reaction that significantly simplifies the operational complexity traditionally associated with this intermediate. For R&D directors and procurement strategists alike, understanding this technological shift is paramount, as it promises not only enhanced chemical purity but also a more robust and economically viable supply chain for high-volume antipsychotic drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
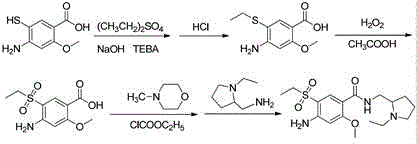
Historically, the synthesis of 4-amino-5-ethylsulfonyl-2-methoxybenzoic acid derivatives has been plagued by cumbersome multi-step processes that rely on hazardous reagents and difficult purification protocols. Traditional routes often commence with 4-amino-2-methoxy-5-mercaptobenzoic acid or require the alkylation of unprotected amines, leading to significant challenges in controlling regioselectivity and managing toxic byproducts. As illustrated in prior art reaction schemes, these legacy methods frequently involve the use of aggressive alkylating agents under harsh conditions, which can degrade sensitive functional groups and result in complex impurity profiles that are costly to remove. Furthermore, the direct handling of free thiol intermediates poses severe safety risks due to their potent odor and instability, necessitating specialized containment equipment that drives up capital expenditure. The inability to isolate pure intermediate species in these conventional workflows often compromises the overall yield of the final API, creating bottlenecks that ripple through the entire production schedule and inflate the cost of goods sold for pharmaceutical manufacturers.
The Novel Approach
In stark contrast to these archaic methodologies, the invention detailed in CN112521318A proposes a streamlined strategy that leverages the stability of a thiocyanate precursor to bypass the pitfalls of free thiol handling. This innovative approach initiates with methyl 4-amino-2-methoxy-5-thiocyanatobenzoate, a stable solid that can be easily manipulated without the safety concerns associated with mercaptans. The core of this novelty lies in a selective reduction step where the thiocyanate group is converted into a thiolate salt in situ, followed by a precise acidification to release the desired thiol product. By decoupling the formation of the sulfur-carbon bond from the final isolation step, this method allows for the removal of insoluble impurities prior to product precipitation, ensuring a level of purity that is difficult to achieve via direct alkylation. The use of benign alcohol-water mixed solvents further enhances the environmental profile of the process, replacing toxic organic solvents with greener alternatives that facilitate easier waste treatment and regulatory compliance.
Mechanistic Insights into Thiocyanate Reduction and Salt Formation
The chemical elegance of this new process is rooted in the nucleophilic attack of sulfide ions on the electrophilic carbon of the thiocyanate group, a transformation that proceeds with high fidelity under controlled thermal conditions. In the first stage of the reaction, sodium sulfide acts as both a source of nucleophilic sulfur and a base, facilitating the cleavage of the carbon-nitrogen triple bond in the thiocyanate moiety. This results in the formation of a soluble sodium thiolate salt intermediate, which remains in the solution phase while insoluble polymeric byproducts or unreacted starting materials can be mechanically filtered off. This physical separation step is critical, as it prevents the carryover of impurities into the final crystallization, thereby acting as a built-in purification mechanism that reduces the need for extensive chromatographic cleaning. The reaction kinetics are carefully managed by maintaining temperatures between 5°C and 90°C, allowing operators to balance reaction rate with selectivity to minimize side reactions such as over-reduction or hydrolysis of the ester group.
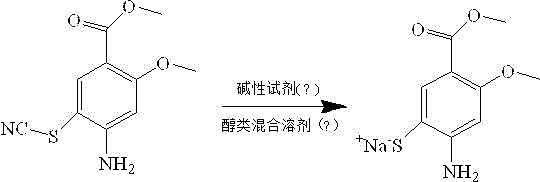
Following the formation of the thiolate salt, the process employs a precise acidification protocol to regenerate the free thiol functionality required for subsequent sulfonylation steps in the Amisulpride synthesis. By adjusting the pH of the filtrate to a specific range of 2 to 6 using acidic reagents like hydrochloric acid or acetic acid, the solubility of the product is drastically reduced, causing it to precipitate as a yellow solid. This pH-controlled precipitation is a powerful tool for impurity rejection, as many acidic or basic contaminants remain dissolved in the mother liquor at these specific pH values. The result is a crude product with significantly higher purity compared to oil-based extractions, simplifying downstream processing. This mechanistic control ensures that the methyl ester group remains intact throughout the reduction, preserving the structural integrity needed for the final coupling reactions in the API synthesis.
How to Synthesize Methyl 4-amino-5-thiophenyl-2-methoxybenzoate Efficiently
Implementing this synthesis route requires careful attention to solvent composition and temperature gradients to maximize the efficiency of the thiocyanate reduction. The patent outlines a robust procedure where the substrate is first dissolved in a mixture of water and an alcohol such as methanol or ethanol, creating a homogeneous phase that supports rapid mass transfer. The addition of the alkaline reagent must be controlled to manage the exotherm and ensure complete conversion to the salt form before proceeding to filtration.
- Dissolve methyl 4-amino-2-methoxy-5-thiocyanatobenzoate in an alcohol-water mixed solvent and add an alkaline reagent like sodium sulfide at controlled temperatures.
- Maintain the reaction mixture between 5-90°C for 0.5 to 9 hours to form the soluble sodium salt intermediate while filtering out insoluble impurities.
- Adjust the pH of the filtrate to 2-6 using an acidic reagent to precipitate the final yellow solid product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology represents a strategic opportunity to optimize the cost structure and reliability of the Amisulpride supply chain. The shift away from complex alkylation routes towards a direct reduction of commercially available thiocyanates eliminates the need for expensive and hazardous alkylating agents, which are often subject to strict regulatory controls and price volatility. By utilizing commodity chemicals like sodium sulfide and common solvents like ethanol, manufacturers can significantly reduce raw material costs while mitigating the risks associated with the storage and transport of dangerous goods. Furthermore, the simplified purification process reduces the consumption of energy and auxiliary materials required for distillation and chromatography, contributing to a lower overall carbon footprint and operational expenditure.
- Cost Reduction in Manufacturing: The elimination of multi-step protection and deprotection sequences inherently lowers the cost of goods by reducing labor hours, reactor occupancy time, and solvent usage. Since the process avoids the use of precious metal catalysts or exotic reagents, the dependency on volatile global markets for specialized chemicals is minimized, leading to more predictable budgeting. The high yield reported in the patent examples suggests that less starting material is wasted, directly improving the atom economy and reducing the cost per kilogram of the final intermediate. Additionally, the ability to filter impurities before the final product isolation reduces the load on wastewater treatment facilities, lowering environmental compliance costs.
- Enhanced Supply Chain Reliability: Sourcing stable thiocyanate precursors is generally more reliable than sourcing unstable mercaptans, which often have limited suppliers due to their hazardous nature. This diversification of the raw material base enhances supply security, ensuring that production schedules are not disrupted by shortages of niche reagents. The robustness of the reaction conditions, which tolerate a wide temperature range and use standard equipment, means that the process can be easily transferred between different manufacturing sites without extensive re-validation. This flexibility is crucial for maintaining continuity of supply in the face of geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The use of water-alcohol solvent systems makes this process inherently safer and easier to scale from pilot plant to commercial tonnage compared to processes relying on chlorinated or aromatic solvents. The absence of heavy metals and the generation of manageable salt byproducts simplify the waste stream, facilitating easier disposal and adherence to increasingly stringent environmental regulations. The straightforward work-up procedure, involving simple filtration and pH adjustment, is highly amenable to continuous processing technologies, offering a clear path for future capacity expansion to meet growing global demand for antipsychotic medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route, based on the specific data and advantages disclosed in the patent literature. Understanding these details is essential for technical teams evaluating the feasibility of adopting this new method for large-scale production.
Q: What is the primary advantage of using the thiocyanate route over traditional mercapto routes?
A: The thiocyanate route avoids the handling of unstable and odorous free mercaptans in early stages, utilizing a stable thiocyanate precursor that is selectively reduced in situ, leading to safer operations and easier purification.
Q: How does the new method improve yield compared to conventional alkylation methods?
A: By eliminating complex protection-deprotection sequences and harsh alkylation conditions that often lead to side reactions, this method achieves yields up to 85% in laboratory examples through a direct and mild reduction pathway.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes common solvents like water and ethanol/methanol and inexpensive reagents like sodium sulfide, making it highly scalable, cost-effective, and environmentally friendlier than routes requiring toxic alkylating agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 4-amino-5-thiophenyl-2-methoxybenzoate Supplier
As the pharmaceutical industry demands ever-higher standards of purity and efficiency, NINGBO INNO PHARMCHEM stands ready to leverage advanced synthetic methodologies like the one described in CN112521318A to deliver superior intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and compliant. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Amisulpride intermediate meets the exacting requirements of global regulatory bodies, providing our partners with the confidence needed to accelerate their own drug development timelines.
We invite forward-thinking pharmaceutical companies to collaborate with us to explore the full potential of this cost-effective and environmentally friendly synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, along with specific COA data and route feasibility assessments. Let us help you secure a sustainable and competitive supply of high-quality psychiatric drug intermediates, driving value and innovation across your entire product portfolio.
