Advanced Meta-Selective C-H Olefination for High-Value Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access complex molecular architectures, particularly those required for next-generation therapeutics. Patent CN109535084B introduces a groundbreaking methodology for the synthesis of meta-alkenyl phenylacetic acid compounds, addressing a long-standing challenge in organic synthesis: the selective functionalization of the meta-position on aromatic rings. Traditionally, achieving meta-selectivity in electrophilic aromatic substitution or transition metal-catalyzed C-H activation has been notoriously difficult due to steric and electronic factors that favor ortho- or para-substitution. This invention leverages a sophisticated U-shaped template strategy, employing a pyrimidine moiety as a novel directing group template. By coordinating with a transition metal center, this template forms a macrocyclic intermediate that precisely positions the catalyst at the meta-carbon, enabling direct olefination. This technological leap not only simplifies the synthetic route by eliminating the need for pre-functionalized halogenated precursors but also significantly enhances the atom economy and overall efficiency of producing high-value pharmaceutical intermediates used in anti-inflammatory and analgesic drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of meta-substituted phenylacetic acid derivatives has relied on multi-step sequences that are both time-consuming and costly. Conventional approaches often involve the initial halogenation of the aromatic ring, followed by cross-coupling reactions such as Suzuki-Miyaura or Heck couplings to introduce the desired alkenyl group. These traditional pathways suffer from several critical drawbacks, including the generation of stoichiometric amounts of toxic waste, the requirement for harsh reaction conditions, and the inherent difficulty in controlling regioselectivity without extensive protecting group manipulation. Furthermore, the reliance on pre-halogenated starting materials increases the raw material costs and complicates the supply chain, as these intermediates often require specialized synthesis themselves. The cumulative effect of these inefficiencies is a higher cost of goods sold (COGS) and a longer time-to-market for new drug candidates, creating a significant bottleneck for R&D teams aiming to scale up production of complex API intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN109535084B offers a streamlined, direct C-H functionalization pathway. By utilizing a pyrimidine-based U-shaped template, the process achieves exceptional meta-selectivity in a single catalytic step. This approach bypasses the need for pre-installed leaving groups, thereby reducing the number of synthetic operations and minimizing waste generation. The reaction operates under relatively mild conditions, typically employing palladium catalysis with simple amino acid-derived ligands and silver oxidants in fluorinated alcohol solvents. This shift from multi-step functional group interconversion to direct C-H activation represents a paradigm shift in process chemistry, offering a robust platform for the rapid diversification of phenylacetic acid scaffolds. The ability to directly install versatile alkenyl handles at the meta-position opens up new avenues for late-stage functionalization of drug-like molecules, providing medicinal chemists with powerful tools for structure-activity relationship (SAR) studies.
Mechanistic Insights into Pd-Catalyzed Meta-Selective C-H Activation
The core of this innovation lies in the unique mechanistic pathway facilitated by the pyrimidine directing group. Unlike conventional bidentate ligands that typically promote ortho-C-H activation through five- or six-membered palladacycles, the extended linker in the U-shaped template forces the formation of a larger macrocyclic transition state. This macrocycle brings the palladium center into close proximity with the meta-C-H bond, lowering the activation energy for cleavage at this specific position while disfavoring ortho-interaction due to geometric constraints. The catalytic cycle initiates with the coordination of the pyrimidine nitrogen to the Pd(II) species, followed by the concerted metalation-deprotonation (CMD) of the meta-C-H bond assisted by the carboxylate ligand, such as N-acetylglycine. Subsequent olefin insertion into the Pd-C bond and beta-hydride elimination release the meta-alkenylated product and regenerate the active Pd(II) catalyst, often with the aid of a silver oxidant to re-oxidize any reduced palladium species. This precise control over the catalytic cycle ensures high regioselectivity and minimizes the formation of ortho- or para-isomers, which are common impurities in non-directed C-H functionalization reactions.
From an impurity control perspective, this mechanism offers distinct advantages for manufacturing high-purity intermediates. The high selectivity inherent in the U-shaped template strategy means that the crude reaction mixture contains significantly fewer regioisomeric byproducts compared to traditional electrophilic substitution methods. This simplifies the downstream purification process, often allowing for straightforward column chromatography or crystallization to achieve pharmaceutical-grade purity. Moreover, the compatibility of the reaction with a wide range of functional groups on both the phenylacetic acid substrate and the olefin coupling partner—such as esters, nitriles, aldehydes, and phosphonates—demonstrates the robustness of the catalytic system. The use of mild oxidants like silver carbonate and solvents like hexafluoroisopropanol (HFIP) further contributes to a cleaner reaction profile, reducing the risk of side reactions such as polymerization of the olefin or over-oxidation of sensitive functional groups, thereby ensuring a consistent and reliable quality profile for the final product.
How to Synthesize Meta-Alkenyl Phenylacetic Acid Efficiently
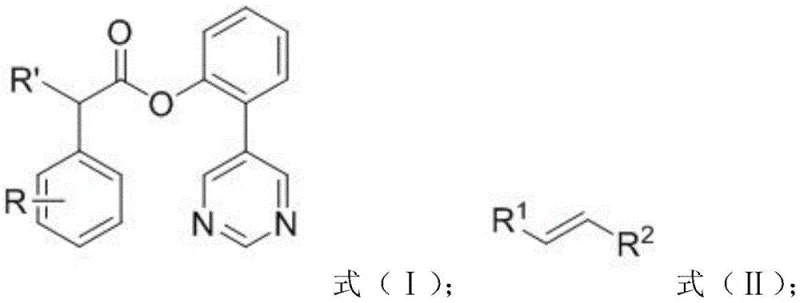
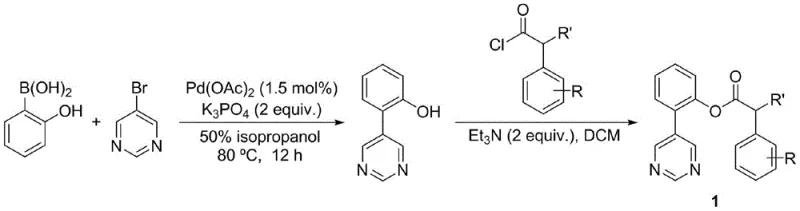
The practical implementation of this synthesis involves a straightforward protocol that is amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the key phenylacetic acid derivative bearing the pyrimidine directing group, which can be synthesized via a Suzuki coupling between 5-bromopyrimidine and 2-hydroxyphenylboronic acid, followed by esterification with the appropriate aryl acetyl chloride. Once the substrate is prepared, the meta-alkenylation is performed by combining the substrate, the chosen olefin, palladium acetate, the amino acid ligand, and silver carbonate in HFIP. The reaction mixture is heated to 80°C for approximately 24 hours under a nitrogen atmosphere. Detailed standardized synthesis steps, including specific molar ratios and workup procedures for various substrates, are outlined in the guide below.
- Prepare the reaction mixture by adding the phenylacetic acid derivative (Formula I), olefin compound (Formula II), Pd(OAc)2 catalyst, Ac-Gly-OH ligand, and Ag2CO3 oxidant into a reactor.
- Add HFIP solvent, vacuum the reactor, and replace with nitrogen. Heat the mixture to 80°C and stir for 24 hours to facilitate the meta-alkenylation.
- Upon completion, remove the solvent via rotary evaporation and purify the crude product using column chromatography with petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this meta-alkenylation technology translates into tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the substantial simplification of the supply chain for raw materials. By eliminating the need for pre-halogenated aromatic building blocks, which are often subject to price volatility and limited supplier availability, manufacturers can source simpler, commodity-grade phenylacetic acid derivatives and olefins. This shift reduces dependency on specialized intermediate suppliers and mitigates the risk of supply disruptions. Furthermore, the reduction in synthetic steps directly correlates with a decrease in overall processing time and resource consumption, leading to significant cost reductions in manufacturing. The ability to produce complex meta-substituted scaffolds in fewer steps enhances the overall throughput of the production facility, allowing for faster response times to market demands and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The direct C-H activation strategy eliminates the need for expensive halogenation reagents and the associated waste treatment costs. By streamlining the synthesis from multiple steps to a single catalytic transformation, the process drastically reduces solvent usage, energy consumption, and labor hours. The use of earth-abundant palladium catalysts in low loadings, combined with recyclable ligands, further optimizes the cost structure. This efficiency gain allows for a more competitive pricing model for high-purity pharmaceutical intermediates, providing a clear margin advantage in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions and the wide availability of starting materials contribute to a more resilient supply chain. Unlike processes requiring cryogenic conditions or highly sensitive organometallic reagents, this method operates at moderate temperatures and uses stable reagents that are easy to handle and store. This reduces the logistical complexity and safety risks associated with transportation and storage of hazardous materials. Consequently, manufacturers can maintain higher safety stock levels and ensure continuous production schedules, safeguarding against potential bottlenecks and ensuring timely delivery to downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reaction vessels and workup procedures that translate well from gram-scale to ton-scale production. The high atom economy of the direct olefination reaction aligns with green chemistry principles, minimizing the generation of hazardous byproducts and reducing the environmental footprint of the manufacturing process. This compliance with increasingly stringent environmental regulations not only avoids potential fines and shutdowns but also enhances the corporate sustainability profile, which is becoming a critical factor in supplier selection for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this meta-alkenylation technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners and stakeholders.
Q: What is the key innovation in this meta-alkenylation method?
A: The method utilizes a novel U-shaped template strategy with a pyrimidine directing group. This allows the formation of a macrocyclic metal complex that specifically targets the meta-position of the phenylacetic acid derivative, overcoming the traditional difficulty of meta-selectivity in C-H activation.
Q: Can the pyrimidine directing group be removed after the reaction?
A: Yes, the patent indicates that the pyrimidine-based template can be removed under mild conditions. This feature is crucial for pharmaceutical applications, allowing the functionalized core to be integrated into final drug molecules like ibuprofen derivatives without retaining the auxiliary group.
Q: What types of olefins are compatible with this synthesis?
A: The process demonstrates broad substrate scope, successfully reacting with various electron-deficient olefins including ethyl acrylate, acrylonitrile, acrolein, vinyl diethyl phosphate, and phenyl vinyl sulfone, yielding diverse meta-alkenylated products with good to excellent yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meta-Alkenyl Phenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative synthetic routes like the one described in CN109535084B can be seamlessly transferred from the lab to the plant. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of meta-alkenyl phenylacetic acid intermediate meets the highest industry standards for potency and impurity profiles. We are committed to leveraging our technical expertise to optimize these processes for maximum efficiency and cost-effectiveness, serving as a trusted extension of your R&D and supply chain teams.
We invite you to explore how our capabilities can support your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis platforms can drive value and speed for your pharmaceutical pipeline.
