Advanced One-Pot Synthesis of Polysubstituted Aminopyridines and Aminoisoquinolines for Commercial Applications
Advanced One-Pot Synthesis of Polysubstituted Aminopyridines and Aminoisoquinolines for Commercial Applications
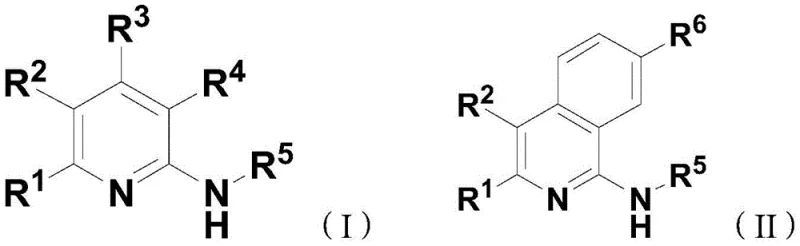
The development of efficient synthetic routes for nitrogen-containing aromatic heterocycles is a critical priority for the modern pharmaceutical and fine chemical industries. Patent CN110204481B introduces a groundbreaking preparation method for polysubstituted nitrogen-containing aromatic heterocyclic compounds, specifically targeting the synthesis of versatile aminopyridine and aminoisoquinoline scaffolds. These structures are indispensable in the creation of fluorescent molecular markers and active pharmaceutical ingredients due to their high fluorescence conversion rates and stable molecular architectures. The disclosed technology utilizes a transition metal-catalyzed, one-pot multicomponent reaction that combines alkenyl azides, isonitriles, and alkyne or benzyne precursors to construct these complex cores with exceptional efficiency. By leveraging this innovative approach, manufacturers can achieve high atom economy while minimizing the environmental footprint associated with traditional multi-step syntheses. This report analyzes the technical merits and commercial viability of this process for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted aminopyridine cores has been plagued by significant inefficiencies that hinder large-scale production and increase costs. Traditional pathways often rely on starting from pre-formed aminopyridines and modifying the ring through electrophilic or nucleophilic reactions, followed by separate coupling steps to functionalize the amino group, which inherently lacks step economy. Alternative methods involving the amination of pyridine oxides require equivalent amounts of phosphorus catalysts, leading to poor atomic economy and the generation of substantial phosphorus-containing by-products that are difficult to remove. Furthermore, certain established protocols necessitate the use of highly toxic malononitrile compounds, posing severe safety risks and regulatory hurdles for industrial facilities. These conventional strategies frequently suffer from insufficient substrate universality, limiting the diversity of fluorescent properties achievable in the final marker molecules. Consequently, the industry has long sought a more direct, safer, and environmentally benign synthetic strategy.
The Novel Approach
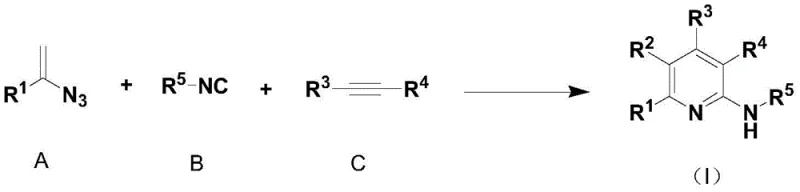
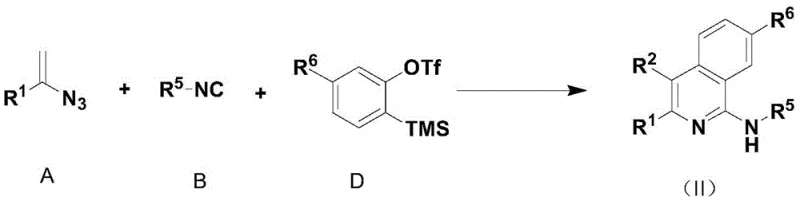
The methodology outlined in the patent represents a paradigm shift by employing a direct, one-pot assembly of the heterocyclic ring from simple, readily available building blocks. Instead of modifying an existing ring, this novel approach constructs the pyridine or isoquinoline skeleton de novo using alkenyl azides, isonitriles, and alkynes under rhodium catalysis. As illustrated in the reaction schemes below, the process allows for the simultaneous formation of multiple bonds and the introduction of diverse substituents in a single operational sequence. This strategy eliminates the need for isolation of intermediates, drastically reducing solvent consumption and labor hours associated with multi-step purifications. The reaction exhibits remarkable tolerance to various functional groups, enabling the incorporation of esters, halogens, and aryl groups without protecting group chemistry. By generating nitrogen gas as the sole byproduct, the process aligns perfectly with green chemistry principles, offering a sustainable alternative to legacy manufacturing techniques.
Mechanistic Insights into Rhodium-Catalyzed Cyclization
The core of this transformative synthesis lies in the precise orchestration of a transition metal-catalyzed cascade reaction, typically utilizing a rhodium catalyst such as {Rh(COD)Cl}2 paired with a 2,2'-bipyridyl ligand. The mechanism initiates with the activation of the alkenyl azide and isonitrile components at room temperature, forming a reactive metallacycle intermediate that sets the stage for ring closure. Upon the subsequent addition of the alkyne or benzyne precursor and heating to temperatures ranging from -10°C to 150°C, the system undergoes a concerted cyclization to forge the aromatic heterocyclic framework. The choice of ligand and catalyst loading, which can be as low as 0.05% to 0.1% equivalent, is critical for maintaining high turnover numbers and ensuring complete conversion of the starting materials. This catalytic system is robust enough to tolerate a wide array of electronic environments on the substrates, from electron-deficient trifluoromethyl groups to electron-rich methoxy substituents. The mild reaction conditions prevent the decomposition of sensitive functional groups, thereby preserving the integrity of complex molecular architectures required for advanced applications.
Impurity control in this process is inherently superior due to the high selectivity of the rhodium catalyst and the clean nature of the byproducts. Unlike phosphorus-mediated reactions that generate stoichiometric waste, this method produces nitrogen gas, which simply vents from the reaction mixture, leaving behind a cleaner crude product profile. The absence of harsh reagents and the use of common organic solvents like 1,4-dioxane or toluene further simplify the downstream purification process, often requiring only standard column chromatography or recrystallization. This high level of chemoselectivity ensures that the final polysubstituted aminopyridines and aminoisoquinolines meet stringent purity specifications essential for pharmaceutical and diagnostic applications. The ability to fine-tune the reaction temperature and molar ratios allows process chemists to optimize yield and minimize side reactions, resulting in a highly reproducible manufacturing protocol. Such mechanistic clarity provides confidence in scaling the process from laboratory benchtop to industrial reactor volumes.
How to Synthesize Polysubstituted Nitrogen Heterocycles Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and temperature control to maximize the formation of the desired heterocyclic core. The process begins by establishing the catalytic environment, followed by the room temperature reaction of the azide and isonitrile components to form the key intermediate. Once this initial phase is complete, the third component is introduced, and the system is heated to drive the final cyclization to completion. Detailed standardized operating procedures regarding specific molar ratios, solvent choices, and workup protocols are critical for ensuring consistent quality across different batches. For a comprehensive guide on the exact experimental steps and conditions optimized for various substrates, please refer to the technical documentation below.
- Prepare the reaction vessel by adding a transition metal catalyst (such as {Rh(COD)Cl}2), a 2,2'-bipyridyl ligand, and an organic solvent like 1,4-dioxane.
- Introduce raw material A (alkenyl azide) and raw material B (isonitrile) to the mixture and allow them to react fully at room temperature.
- Add raw material C (alkyne) or raw material D (benzyne precursor) and heat the reaction mixture to a temperature between -10°C and 150°C to complete the cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this rhodium-catalyzed one-pot method presents a compelling value proposition centered on cost efficiency and operational reliability. The elimination of toxic reagents like malononitrile and the removal of phosphorus waste streams significantly reduce the costs associated with hazardous waste disposal and regulatory compliance. Furthermore, the high atom economy and the ability to use commercially available solvents without special drying or degassing treatments lower the overall raw material expenditure. By consolidating multiple synthetic steps into a single pot, manufacturers can drastically reduce labor costs, energy consumption, and equipment occupancy time, leading to a leaner and more agile production workflow. These factors combine to create a robust supply chain capable of delivering high-quality intermediates with reduced lead times and enhanced consistency.
- Cost Reduction in Manufacturing: The transition to this one-pot methodology eliminates the need for expensive and toxic reagents while minimizing waste generation, directly translating to lower variable costs per kilogram of product. The low catalyst loading requirements mean that precious metal expenses are kept to a minimum, and the simplified workup reduces the consumption of purification media and solvents. Additionally, the avoidance of multi-step sequences removes the yield losses typically accumulated at each isolation stage, effectively increasing the overall throughput of the facility. This streamlined approach allows for significant cost reduction in API manufacturing and fine chemical production without compromising on the quality of the final output.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials such as alkenyl azides and isonitriles ensures a secure supply base that is less susceptible to market volatility. Since the reaction does not demand strict anhydrous or anaerobic conditions, the logistical burden of transporting and storing sensitive reagents is greatly diminished, simplifying inventory management. The robustness of the catalytic system against functional group variations means that supply disruptions for specific substituted precursors can often be mitigated by switching to alternative analogs without re-validating the entire process. This flexibility enhances the resilience of the supply chain, ensuring continuous availability of critical pharmaceutical intermediates even during periods of raw material scarcity.
- Scalability and Environmental Compliance: The mild reaction conditions and the generation of benign nitrogen gas as the only byproduct make this process exceptionally easy to scale from pilot plants to full commercial production. Facilities can expand capacity without needing specialized infrastructure for handling hazardous gases or corrosive wastes, thereby accelerating time-to-market for new products. The alignment with green chemistry principles facilitates easier permitting and regulatory approval, reducing the administrative overhead associated with environmental compliance. This scalability ensures that the technology can meet growing global demand for high-purity heterocyclic compounds while maintaining a sustainable and responsible manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this method into their existing pipelines and for procurement specialists assessing supplier capabilities. The answers provided are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and relevance. For further specific technical data or custom synthesis requests, direct consultation with our technical team is recommended.
Q: What are the primary advantages of this rhodium-catalyzed method over traditional aminopyridine synthesis?
A: Unlike traditional methods that require multiple steps, equivalent phosphorus catalysts, or toxic malononitrile, this one-pot approach offers high atom economy with nitrogen gas as the only byproduct, significantly reducing waste and operational complexity.
Q: Does this synthesis method require strict anhydrous or anaerobic conditions?
A: No, the reaction described in patent CN110204481B does not require strict anhydrous or anaerobic conditions, making it operationally simple and highly suitable for large-scale commercial manufacturing environments.
Q: What is the substrate scope for the R groups in the final heterocyclic products?
A: The method demonstrates excellent functional group tolerance, accommodating a wide range of substituents including aryl, heteroaryl, ester, halogen, and alkyl groups, allowing for the diverse modification of the pyridine or isoquinoline core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Nitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this rhodium-catalyzed technology in advancing the production of high-value pharmaceutical intermediates and fluorescent markers. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory discovery to industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of polysubstituted aminopyridines or aminoisoquinolines delivered meets the highest international standards. We are committed to leveraging our technical expertise to optimize this novel route for your specific application needs, providing a reliable foundation for your drug development or material science projects.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can drive efficiency and cost savings in your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this one-pot process for your specific target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique requirements, ensuring a partnership built on transparency, quality, and mutual success.
