Advanced Palladium-Catalyzed Synthesis of Polysubstituted Bisquinolines for Commercial Scale-Up
The landscape of organic synthesis for high-value heterocyclic compounds is undergoing a significant transformation, driven by the urgent demand for more efficient and scalable routes in the electronics and pharmaceutical sectors. Patent CN112898297A, filed in June 2021, introduces a groundbreaking methodology for the preparation of polysubstituted bisquinoline compounds, a class of molecules characterized by their large conjugated planar structures and versatile functionalization potential. This technology represents a pivotal advancement for manufacturers seeking a reliable OLED material supplier, as it addresses long-standing challenges in constructing complex nitrogen-containing heterocycles. The core innovation lies in the utilization of a palladium-catalyzed cascade reaction involving symmetric or asymmetric ortho-amino alkynes and isonitriles, which allows for the direct assembly of the bisquinoline skeleton in a single operational step. 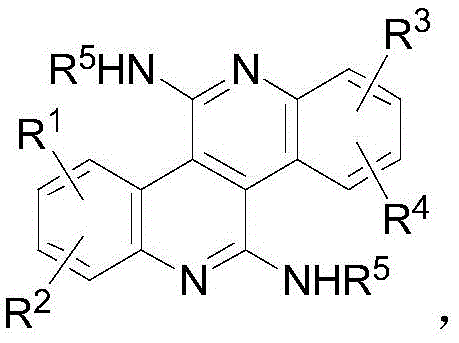 By enabling the introduction of diverse substituents such as methyl, methoxy, cyano, chlorine, and bromine at multiple positions, this method provides an unprecedented level of structural tunability essential for optimizing the optoelectronic properties of next-generation display materials.
By enabling the introduction of diverse substituents such as methyl, methoxy, cyano, chlorine, and bromine at multiple positions, this method provides an unprecedented level of structural tunability essential for optimizing the optoelectronic properties of next-generation display materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted quinoline and bisquinoline derivatives has been plagued by significant inefficiencies that hinder large-scale commercial adoption. Traditional pathways often rely on multi-step sequences involving harsh reaction conditions, such as strong acids or high temperatures, which can lead to the decomposition of sensitive functional groups and result in poor overall yields. Furthermore, many existing catalytic systems struggle with the simultaneous insertion of multiple isonitrile molecules due to the high reactivity of isonitriles themselves, making the control of selectivity extremely difficult. This lack of control frequently results in complex mixtures of by-products, necessitating costly and time-consuming purification processes that erode profit margins. For procurement managers focused on cost reduction in electronic chemical manufacturing, these inefficiencies translate into higher raw material consumption and extended production cycles, rendering many conventional routes economically unviable for mass production.
The Novel Approach
In stark contrast, the methodology disclosed in CN112898297A offers a streamlined and robust alternative that effectively circumvents these traditional bottlenecks. The novel approach leverages a sophisticated palladium-catalyzed mechanism where an alkenyl palladium intermediate undergoes isonitrile migration followed by the insertion of a second isonitrile molecule, ultimately leading to the formation of the bisquinoline core through reductive elimination. 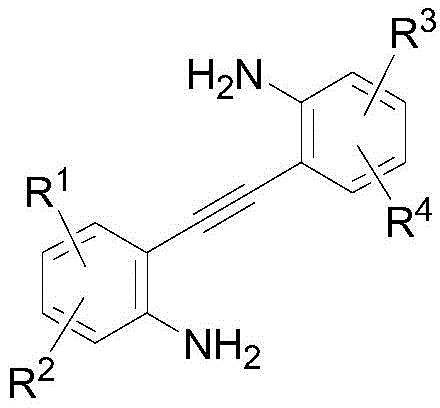 This one-pot strategy not only simplifies the operational workflow but also exhibits remarkable substrate applicability, tolerating a wide array of functional groups without the need for protective group chemistry. The ability to utilize cheap and readily available starting materials, combined with mild reaction conditions ranging from 80°C to 120°C, ensures that the process is both energy-efficient and environmentally friendlier than legacy methods. For supply chain heads, this translates to a more resilient production model with reduced dependency on exotic reagents and simplified logistics.
This one-pot strategy not only simplifies the operational workflow but also exhibits remarkable substrate applicability, tolerating a wide array of functional groups without the need for protective group chemistry. The ability to utilize cheap and readily available starting materials, combined with mild reaction conditions ranging from 80°C to 120°C, ensures that the process is both energy-efficient and environmentally friendlier than legacy methods. For supply chain heads, this translates to a more resilient production model with reduced dependency on exotic reagents and simplified logistics.
Mechanistic Insights into Pd-Catalyzed Isonitrile Insertion Cyclization
To fully appreciate the technical depth of this innovation, one must examine the intricate catalytic cycle that drives the formation of the polysubstituted bisquinoline scaffold. The reaction initiates with the activation of the ortho-amino alkyne by the palladium salt, likely forming a reactive alkenyl palladium species. This intermediate then undergoes a crucial migratory insertion with the first molecule of isonitrile, a step that is typically challenging to control in multi-component reactions. However, the specific ligand environment and reaction conditions optimized in this patent facilitate a second, sequential insertion of another isonitrile molecule. This dual insertion capability is the hallmark of the invention, allowing for the rapid buildup of molecular complexity. The cycle concludes with an intramolecular cyclization and reductive elimination step that releases the final bisquinoline product while regenerating the active palladium catalyst. This mechanistic elegance ensures high atom economy and minimizes the generation of waste, aligning perfectly with modern green chemistry principles.
From an impurity control perspective, the mechanism offers distinct advantages that are critical for R&D directors concerned with purity specifications. The high selectivity of the palladium catalyst towards the desired cascade pathway suppresses the formation of oligomeric by-products that often arise from uncontrolled isonitrile polymerization. Additionally, the tolerance for halogen substituents (chlorine and bromine) suggests that the catalytic system does not promote unwanted dehalogenation side reactions, which is a common pitfall in transition metal catalysis. This stability allows for the synthesis of halogenated derivatives that can serve as valuable handles for further cross-coupling reactions, thereby expanding the utility of these intermediates in the synthesis of even more complex organic semiconductors. The rigorous control over the reaction pathway ensures that the final product profile is clean, reducing the burden on downstream purification units.
How to Synthesize Polysubstituted Bisquinoline Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the stoichiometry and reaction parameters outlined in the patent data. The process is designed to be operationally simple, involving the dispersion of the palladium catalyst and base in a solvent like acetonitrile, followed by the addition of the substrates. The detailed standardized synthesis steps below provide a clear roadmap for replicating the high yields and purity reported in the examples, ensuring that technical teams can validate the feasibility of this route for their specific applications.
- Disperse the palladium salt catalyst (e.g., allyl palladium chloride dimer) and alkali base (e.g., potassium carbonate) in a suitable solvent such as acetonitrile.
- Add the ortho-amino alkyne substrate and the isonitrile reactant (e.g., tert-butyl isonitrile) to the reaction mixture under stirring.
- Heat the reaction mixture to 80-120°C for 0.5 to 2 hours, then perform extraction and column chromatography purification to isolate the final bisquinoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the shift to this novel synthetic methodology offers profound strategic benefits that extend beyond mere technical novelty. The adoption of this Pd-catalyzed route directly addresses key pain points in the supply chain of specialty chemicals, particularly regarding cost structure and scalability. By replacing complex, multi-step syntheses with a concise one-pot procedure, manufacturers can drastically reduce the number of unit operations required, leading to significant savings in labor, energy, and equipment usage. This efficiency gain is compounded by the use of commodity-grade solvents and bases, which are universally available and inexpensive, thereby insulating the production process from the volatility of niche reagent markets.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the elimination of expensive transition metal removal steps and the reduction of solvent consumption associated with intermediate isolations. Since the reaction proceeds with high conversion and selectivity, the need for extensive chromatographic purification is minimized, or at least simplified to standard flash chromatography using common eluent systems like petroleum ether and ethyl acetate. Furthermore, the avoidance of cryogenic conditions or ultra-high pressures means that existing standard reactor infrastructure can be utilized without the need for capital-intensive upgrades. These factors collectively contribute to a substantially lower cost of goods sold (COGS), enhancing the competitiveness of the final electronic materials in the global market.
- Enhanced Supply Chain Reliability: A critical advantage of this method is its reliance on ortho-amino alkynes and isonitriles, which are commercially accessible building blocks with stable supply chains. Unlike specialized precursors that may be sourced from a single vendor, these starting materials are produced by multiple suppliers worldwide, mitigating the risk of supply disruptions. The robustness of the reaction conditions also implies a wider operating window, reducing the likelihood of batch failures due to minor fluctuations in temperature or mixing rates. For supply chain heads, this reliability ensures consistent delivery schedules and the ability to scale production volumes rapidly in response to market demand without compromising quality.
- Scalability and Environmental Compliance: The environmental footprint of chemical manufacturing is increasingly scrutinized, and this process offers a greener alternative to traditional methods. The high atom efficiency and the potential for solvent recycling align with stringent environmental regulations, reducing the costs associated with waste disposal and treatment. The simplicity of the workup procedure, involving basic extraction and drying, facilitates easier scale-up from gram to kilogram and ton scales. This scalability is essential for meeting the growing demand for high-performance OLED materials, ensuring that production can keep pace with the expansion of the display and lighting industries while maintaining compliance with global sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this polysubstituted bisquinoline synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this Pd-catalyzed method over conventional quinoline synthesis?
A: Unlike conventional methods that often suffer from harsh conditions and limited substrate scope, this novel approach utilizes mild reaction temperatures (80-120°C) and demonstrates exceptional tolerance for diverse functional groups including halogens, cyano, and methoxy groups, enabling the one-step construction of complex bisquinoline scaffolds.
Q: Can this synthesis method be scaled for industrial production of OLED materials?
A: Yes, the patent explicitly highlights the method's suitability for industrial production due to the use of cheap and readily available raw materials, simple operational procedures, and the elimination of complex multi-step sequences, which significantly streamlines the manufacturing process for electronic chemicals.
Q: What specific applications do these polysubstituted bisquinolines serve?
A: These compounds serve as critical intermediates in the field of organic light-emitting materials (OLEDs) and function as highly sensitive fluorescent probes for the detection of metal ions, particularly palladium ions, leveraging their large conjugated planar molecular structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Bisquinoline Supplier
The technological potential of polysubstituted bisquinolines in the realm of organic electronics and fluorescence detection is immense, yet realizing this potential requires a manufacturing partner with deep expertise in complex heterocycle synthesis. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise requirements of palladium-catalyzed reactions, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that the performance of OLED materials is critically dependent on the absence of trace impurities, and our dedicated analytical teams employ advanced characterization techniques to guarantee product integrity.
We invite forward-thinking enterprises to collaborate with us to leverage this cutting-edge synthesis route for their product pipelines. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your transition to these advanced materials is seamless, cost-effective, and strategically sound.
