Scalable Metal-Free Synthesis of 6-Aryl-benzofuranocarbazoles for Advanced Optical Materials
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for advanced functional materials. Patent CN110041340B introduces a groundbreaking approach to synthesizing 6-aryl-benzofuranocarbazole compounds, a class of molecules with significant potential in the realm of fluorescent probes and organic electronics. This innovation leverages a unique multicomponent reaction strategy that operates under exceptionally mild conditions, specifically utilizing the induction effect of ortho-hydroxyl groups to drive the cyclization process efficiently. Unlike traditional methods that often rely on harsh conditions or expensive catalysts, this protocol achieves high atom economy and structural diversity through a simple one-pot procedure. The core structural framework of these novel compounds, as depicted in the general formula, allows for extensive modification at multiple positions, enabling chemists to fine-tune electronic and optical properties for specific applications. 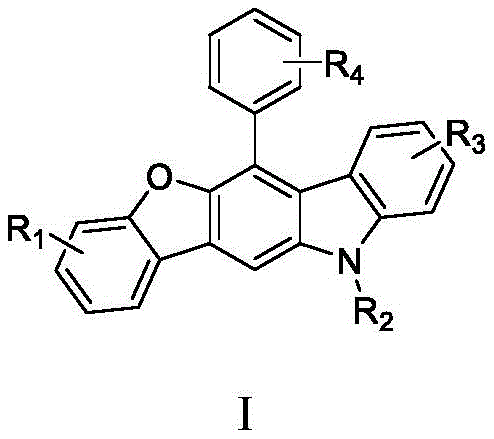 This technological advancement represents a significant leap forward for reliable pharmaceutical intermediate supplier networks looking to integrate high-value fluorescent scaffolds into their portfolios without the burden of complex processing requirements.
This technological advancement represents a significant leap forward for reliable pharmaceutical intermediate supplier networks looking to integrate high-value fluorescent scaffolds into their portfolios without the burden of complex processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of complex fused heterocyclic systems like benzofuranocarbazoles has been fraught with synthetic challenges that hinder large-scale commercial adoption. Conventional strategies often depend heavily on transition metal catalysis, which introduces significant complications regarding residual metal contamination, a critical quality attribute for any active pharmaceutical ingredient or electronic material. Furthermore, many existing protocols, such as those reported by W.-J. Xiao involving sulfur ylides and nitroalkenes, typically require specific catalysts like thiourea and DMAP or involve intricate cascade sequences that are sensitive to reaction conditions. 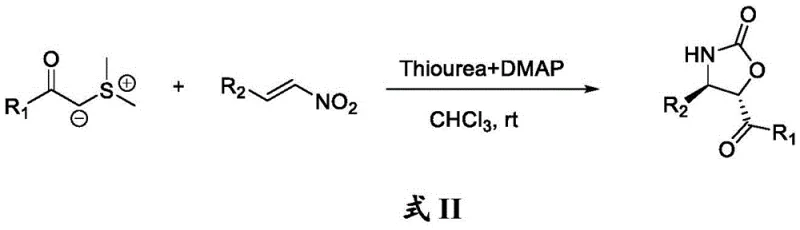 These traditional pathways frequently suffer from limited substrate scope, where the presence of certain functional groups can poison the catalyst or lead to undesirable side reactions, thereby reducing overall yield and purity. The necessity for strict anhydrous conditions or elevated temperatures in many of these older methods also escalates energy consumption and operational costs, making them less attractive for cost reduction in electronic chemical manufacturing. Additionally, the multi-step nature of some conventional syntheses increases the generation of chemical waste, posing environmental compliance challenges that modern supply chains strive to minimize.
These traditional pathways frequently suffer from limited substrate scope, where the presence of certain functional groups can poison the catalyst or lead to undesirable side reactions, thereby reducing overall yield and purity. The necessity for strict anhydrous conditions or elevated temperatures in many of these older methods also escalates energy consumption and operational costs, making them less attractive for cost reduction in electronic chemical manufacturing. Additionally, the multi-step nature of some conventional syntheses increases the generation of chemical waste, posing environmental compliance challenges that modern supply chains strive to minimize.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN110041340B offers a streamlined, metal-free alternative that fundamentally reshapes the economic and technical feasibility of producing these valuable intermediates. By exploiting the intrinsic reactivity of (E)-2-(2-nitrovinyl)phenols, the new method utilizes the ortho-hydroxyl group as an internal directing group to facilitate the cyclization without external metal promoters. This strategic design eliminates the need for expensive and toxic transition metals, thereby removing the costly and time-consuming heavy metal scavenging steps that typically bottleneck production timelines. The reaction proceeds smoothly at room temperature in common solvents like 1,2-dichloroethane, demonstrating remarkable tolerance to a wide array of substituents including halogens, electron-withdrawing nitro groups, and electron-donating methoxy moieties. This broad compatibility ensures that the commercial scale-up of complex polymer additives or pharmaceutical precursors can be achieved with consistent quality across diverse batches. The simplicity of the one-pot operation not only enhances safety by reducing handling of reactive intermediates but also drastically improves the overall process mass intensity, aligning perfectly with green chemistry principles demanded by today's regulatory environment.
Mechanistic Insights into Metal-Free Multicomponent Cyclization
The mechanistic elegance of this synthesis lies in its ability to orchestrate a sequence of bond-forming events through a carefully designed cascade initiated by the unique electronic properties of the starting materials. The reaction begins with the interaction between the arylacetylthio ylide and the nitroalkene component, where the strong electron-withdrawing nature of the nitro group polarizes the conjugated double bond, making it highly susceptible to nucleophilic attack. 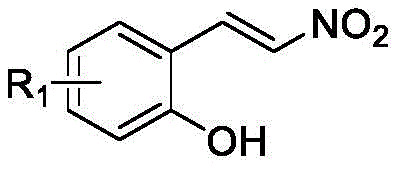 This initial Michael addition generates a key intermediate that is stabilized by the adjacent carbonyl and sulfonium groups, setting the stage for the subsequent ring-closing steps. The presence of the ortho-hydroxyl group on the phenol ring is critical, as it acts as an internal nucleophile that attacks the activated species, triggering an intracyclic nucleophilic进攻 that forms the benzofuran moiety. This is followed by a [4+2] cycloaddition or similar annulation with the indole component, driven by the acidic conditions provided by trifluoroacetic acid, which protonates key intermediates to lower activation barriers. The entire sequence is a testament to the power of organocatalysis and substrate-controlled reactivity, avoiding the randomness often associated with radical processes. Understanding this mechanism allows process chemists to predict impurity profiles more accurately, ensuring that the final high-purity OLED material meets stringent specifications for optical clarity and performance.
This initial Michael addition generates a key intermediate that is stabilized by the adjacent carbonyl and sulfonium groups, setting the stage for the subsequent ring-closing steps. The presence of the ortho-hydroxyl group on the phenol ring is critical, as it acts as an internal nucleophile that attacks the activated species, triggering an intracyclic nucleophilic进攻 that forms the benzofuran moiety. This is followed by a [4+2] cycloaddition or similar annulation with the indole component, driven by the acidic conditions provided by trifluoroacetic acid, which protonates key intermediates to lower activation barriers. The entire sequence is a testament to the power of organocatalysis and substrate-controlled reactivity, avoiding the randomness often associated with radical processes. Understanding this mechanism allows process chemists to predict impurity profiles more accurately, ensuring that the final high-purity OLED material meets stringent specifications for optical clarity and performance.
Controlling the impurity profile in such complex multicomponent reactions is paramount for ensuring the reliability of the final product, especially when destined for sensitive applications like biological imaging or display technologies. The metal-free nature of this reaction inherently reduces the risk of metal-induced degradation or coloration, which is a common issue in fluorescent materials where trace metals can quench emission efficiency. Furthermore, the mild acidic conditions promote the desired cyclization pathway while minimizing side reactions such as polymerization of the nitroalkene or decomposition of the sensitive ylide reagent. The use of trifluoroacetic acid serves a dual purpose: it activates the electrophiles for the indole addition and helps in the elimination of the dimethyl sulfide leaving group from the ylide, driving the equilibrium towards the product. This precise control over the reaction trajectory ensures that byproducts are minimal and easily separable, facilitating a straightforward purification process via standard column chromatography. For R&D teams, this means that scaling from milligram to kilogram quantities does not require a complete re-optimization of the purification train, significantly de-risking the technology transfer phase.
How to Synthesize 6-Aryl-benzofuranocarbazole Efficiently
The practical implementation of this synthesis is designed for ease of execution, requiring standard laboratory equipment and readily available reagents that are common in most chemical manufacturing facilities. The protocol involves dissolving the nitroalkene and ylide precursors in a solvent such as 1,2-dichloroethane and allowing them to react at ambient temperature for a short period to generate the reactive intermediate in situ. Once this initial transformation is confirmed, the indole derivative and a catalytic amount of trifluoroacetic acid are introduced to the mixture, initiating the final cyclization cascade that constructs the fused carbazole core. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and timing required to maximize yield and purity. This operational simplicity makes it an ideal candidate for rapid prototyping of new analogues, allowing medicinal chemists to explore structure-activity relationships without being bogged down by complex synthetic logistics.
- Dissolve (E)-2-(2-nitrovinyl)phenol and arylacetylthio ylide in 1,2-dichloroethane and react at 25°C for 0.5 to 2 hours to form the intermediate.
- Add the indole derivative and trifluoroacetic acid to the reaction mixture once the intermediate is formed.
- Stir the reaction at room temperature for 24 hours, then concentrate and purify via column chromatography to obtain the final yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free synthesis route offers substantial strategic advantages that directly impact the bottom line and operational resilience. The elimination of transition metal catalysts removes a major cost driver associated with both the purchase of precious metals and the subsequent validation required to prove their absence in the final product. 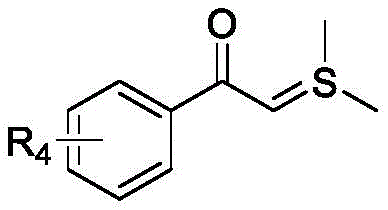 This simplification translates into significantly reduced manufacturing costs, as the expensive scavenging resins and additional filtration steps typically needed for metal removal are rendered unnecessary. Furthermore, the use of room temperature conditions drastically lowers energy consumption compared to processes requiring reflux or cryogenic cooling, contributing to a smaller carbon footprint and lower utility bills. The robustness of the reaction across a wide range of substrates ensures supply chain continuity, as manufacturers are not forced to source exotic or unstable reagents that might be subject to market volatility. This flexibility allows for the rapid substitution of raw materials if supply disruptions occur, maintaining production schedules without compromising on quality. Additionally, the high atom economy and reduced waste generation simplify waste disposal compliance, further enhancing the overall cost-effectiveness of the process for large-scale industrial applications.
This simplification translates into significantly reduced manufacturing costs, as the expensive scavenging resins and additional filtration steps typically needed for metal removal are rendered unnecessary. Furthermore, the use of room temperature conditions drastically lowers energy consumption compared to processes requiring reflux or cryogenic cooling, contributing to a smaller carbon footprint and lower utility bills. The robustness of the reaction across a wide range of substrates ensures supply chain continuity, as manufacturers are not forced to source exotic or unstable reagents that might be subject to market volatility. This flexibility allows for the rapid substitution of raw materials if supply disruptions occur, maintaining production schedules without compromising on quality. Additionally, the high atom economy and reduced waste generation simplify waste disposal compliance, further enhancing the overall cost-effectiveness of the process for large-scale industrial applications.
- Cost Reduction in Manufacturing: The absence of noble metal catalysts such as palladium or rhodium eliminates the need for costly catalyst recovery systems and rigorous heavy metal testing, leading to direct savings in both material and analytical expenses. By operating at room temperature, the process avoids the high energy costs associated with heating or cooling reactors, resulting in substantial operational expenditure reductions over the lifecycle of the product. The one-pot nature of the reaction minimizes solvent usage and intermediate isolation steps, which reduces labor time and consumable costs associated with work-up procedures. These cumulative efficiencies create a leaner manufacturing model that is highly competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials like nitroalkenes and indoles ensures a secure supply base that is less prone to geopolitical or logistical disruptions. The broad substrate tolerance means that if a specific substituted reagent becomes unavailable, alternative analogues can often be substituted without revalidating the entire process, providing crucial flexibility. The simplified purification workflow reduces the dependency on specialized chromatography media or complex distillation setups, making the technology transferable to a wider network of contract manufacturing organizations. This decentralization capability strengthens the supply chain against single-point failures and ensures consistent delivery of high-purity intermediates to downstream customers.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of toxic metal residues make this process inherently safer and easier to scale from pilot plant to full commercial production volumes. The reduced generation of hazardous waste aligns with increasingly strict environmental regulations, minimizing the risk of fines and permitting delays. The high selectivity of the reaction ensures that resource utilization is optimized, supporting sustainability goals and enhancing the corporate social responsibility profile of the manufacturing entity. This environmental compatibility is a key differentiator when bidding for contracts with global partners who prioritize green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this novel synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this chemistry into existing production lines or new product development pipelines.
Q: Does this synthesis require transition metal catalysts?
A: No, the process described in patent CN110041340B is completely metal-free, utilizing trifluoroacetic acid as the promoter, which significantly simplifies downstream purification and eliminates heavy metal residue concerns.
Q: What is the substrate scope for this reaction?
A: The method demonstrates broad substrate tolerance, accommodating various substituents such as halogens, methoxy, nitro, and cyano groups on the phenol, ylide, and indole components, allowing for diverse functionalization.
Q: What are the optical properties of the resulting compounds?
A: The synthesized 6-aryl-benzofuranocarbazoles exhibit significant fluorescence with large Stokes shifts, making them highly suitable for applications in molecular imaging and as potential fluorophores in optoelectronic devices.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Aryl-benzofuranocarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for producing high-performance fluorescent intermediates and pharmaceutical building blocks. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 6-aryl-benzofuranocarbazole delivered meets the highest standards of quality and consistency. We understand the critical nature of supply chain stability and are committed to providing a reliable source of these advanced materials to support your R&D and manufacturing needs globally.
We invite you to engage with our technical procurement team to discuss how this innovative chemistry can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free process for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate the viability of this technology for your portfolio. Let us collaborate to drive efficiency and innovation in your supply chain together.
