Advanced Ruthenium-Catalyzed Asymmetric Hydrogenation for Scalable Pharmaceutical Intermediate Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce chiral building blocks, particularly optically active aminoalcohols which serve as critical precursors for numerous active pharmaceutical ingredients (APIs). Patent CN101983186B introduces a groundbreaking advancement in this domain by disclosing a novel ruthenium compound designed specifically for the asymmetric reduction of carbonyl compounds. This technology addresses the longstanding challenges associated with traditional catalytic methods, offering a robust solution for the high-yield and high-stereoselective production of optically active aminoalcohol compounds from alpha-aminoketone substrates. The core innovation lies in the specific coordination chemistry of the ruthenium center, which is stabilized by a unique combination of a bidentate phosphine ligand and an optically active diamine ligand. This configuration creates a highly defined chiral environment that dictates the stereochemical outcome of the hydrogenation reaction with remarkable precision. For R&D directors and process chemists, this patent represents a significant opportunity to optimize synthetic routes for complex molecules such as ephedrine derivatives, ensuring both purity and efficiency in the manufacturing pipeline.
The limitations of conventional methods for producing optically active alcohol compounds have long been a bottleneck in industrial organic synthesis. Historically, processes relied heavily on rhodium or iridium coordination compounds, which, while effective, suffer from significant drawbacks including the high cost of the noble metals and relatively low hydrogenation activity. These traditional catalysts often necessitate harsh reaction conditions, such as elevated temperatures or excessively high hydrogen pressures, to achieve acceptable conversion rates. Furthermore, alternative methods utilizing ruthenium catalysts with hydrogen transfer agents like formic acid introduce operational complexities and higher costs compared to direct hydrogenation with molecular hydrogen. Another critical issue with prior art ruthenium catalysts is their dependence on expensive, multi-substituted bidentate phosphine ligands and difficult-to-synthesize diamine ligands to achieve good results. Without these sophisticated and costly ligand systems, the stereoselectivity and yield often drop precipitously, rendering the process economically unviable for large-scale applications. These factors collectively contribute to increased production costs and supply chain vulnerabilities for manufacturers relying on older technologies.
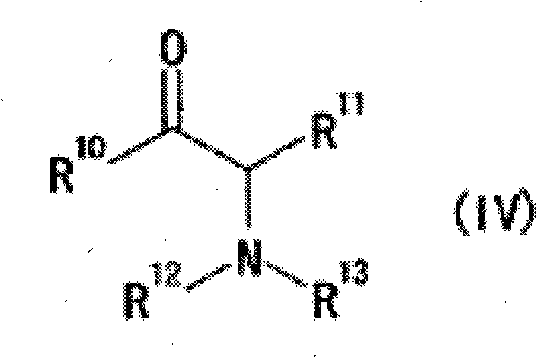
The novel approach detailed in CN101983186B effectively circumvents these historical constraints by employing a specifically engineered ruthenium complex that balances activity, selectivity, and cost. The new catalyst system utilizes a bidentate phosphine ligand represented by Formula (II), where the phosphorus atoms are bonded to substituted phenyl groups, creating a sterically demanding yet electronically tuned environment around the metal center. This is coupled with an optically active diamine ligand, which works in concert with the phosphine to establish a rigid chiral pocket. This dual-ligand strategy allows for the use of inexpensive hydrogen gas as the reducing agent, eliminating the need for costly hydrogen donors like formic acid. The result is a catalytic system that operates with high efficiency under milder conditions, typically between 25°C and 40°C, and at moderate hydrogen pressures ranging from 3 to 50 atmospheres. This shift not only reduces energy consumption but also simplifies the reactor requirements, making the process inherently safer and more scalable for industrial operations.
Mechanistic Insights into Ru-Catalyzed Asymmetric Hydrogenation
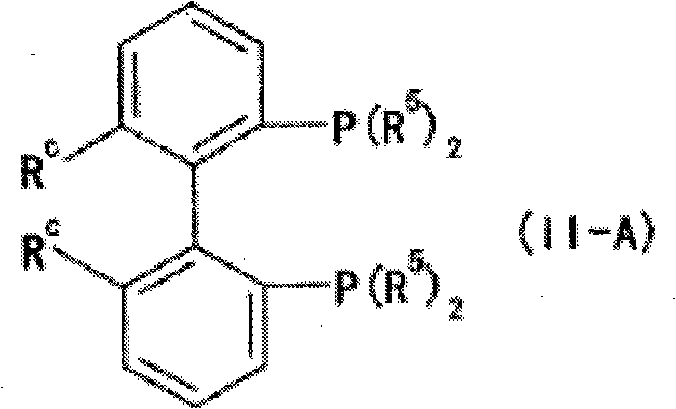
The mechanistic superiority of this ruthenium-catalyzed system stems from the precise spatial arrangement of the ligands around the central metal atom, which governs the approach of the substrate during the hydrogenation event. The bidentate phosphine ligand, particularly the preferred Formula (II-A) featuring a 2,2'-bis[bis(3,5-dimethylphenyl)phosphino]-6,6'-dimethyl-1,1'-biphenyl backbone, provides substantial steric bulk. This bulkiness restricts the rotational freedom of the complex and forces the alpha-aminoketone substrate to bind in a specific orientation that favors the formation of one enantiomer over the other. The 3,5-dimethylphenyl groups on the phosphorus atoms play a crucial role in shielding specific quadrants of the metal center, thereby directing the hydride transfer to a specific face of the carbonyl group. Simultaneously, the optically active diamine ligand, such as 1,1-bis(4-methoxyphenyl)-2-isopropyl-1,2-ethylenediamine, reinforces this chiral induction through secondary interactions, potentially involving hydrogen bonding or dipole alignment with the substrate's amino group. This synergistic effect between the phosphine and diamine ligands ensures that the transition state for the desired stereoisomer is significantly lower in energy than that of the undesired isomer.
Impurity control is another critical aspect where this mechanistic design excels, directly impacting the downstream purification burden for pharmaceutical manufacturers. The high stereoselectivity of the catalyst minimizes the formation of unwanted diastereomers and enantiomers, which are often difficult and expensive to separate from the desired product. In the examples provided within the patent, the reaction consistently yields a syn/anti diastereomer ratio greater than 20:1, with some optimized conditions pushing this selectivity even higher. Furthermore, the enantiomeric excess (ee) frequently exceeds 99%, indicating that the catalyst effectively suppresses the non-selective background reaction pathways that typically lead to racemic mixtures. By preventing the formation of these structural impurities at the source, the process reduces the need for extensive chromatographic purification or recrystallization steps. This not only improves the overall mass balance and yield of the final API intermediate but also ensures a cleaner impurity profile, which is a stringent requirement for regulatory approval in the pharmaceutical sector. The robustness of the catalyst against functional group tolerance further ensures that side reactions, such as dehalogenation or over-reduction, are kept to a minimum.
How to Synthesize Optically Active Aminoalcohols Efficiently
The synthesis of these high-value chiral intermediates using the patented ruthenium technology involves a streamlined sequence that is amenable to both laboratory optimization and commercial scale-up. The process begins with the preparation of the active catalyst species, which can be generated either by isolating the ruthenium complex beforehand or by forming it in situ within the reaction vessel. The in situ method offers logistical advantages by reducing the number of handling steps and exposure of the sensitive catalyst to air or moisture. Once the catalyst system is established, the alpha-aminoketone substrate is introduced into the reaction mixture along with a suitable solvent, typically an alcohol like 2-propanol, which also serves as a proton source in certain mechanistic pathways. A base, such as potassium tert-butoxide, is added to activate the catalyst and facilitate the heterolytic cleavage of the hydrogen molecule. The detailed standardized synthesis steps for implementing this technology are outlined in the guide below.
- Preparation of the Ruthenium Catalyst Complex by reacting a divalent ruthenium-halide complex with a bidentate phosphine ligand and an optically active diamine.
- Setup of the hydrogenation reaction by dissolving the alpha-aminoketone substrate and the prepared catalyst in a suitable solvent such as 2-propanol.
- Execution of the asymmetric reduction under hydrogen pressure (1-200 atm) at controlled temperatures (25-40°C) with the addition of a base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ruthenium-catalyzed technology translates into tangible strategic advantages regarding cost structure and supply reliability. The primary driver for cost reduction lies in the catalyst's exceptional turnover number and frequency. The patent data indicates that the catalyst can operate effectively at substrate-to-catalyst (S/C) molar ratios as high as 10,000 while maintaining complete conversion and high stereoselectivity. This high efficiency means that significantly less of the expensive ruthenium metal and specialized ligands are required per kilogram of product produced, drastically lowering the raw material cost contribution of the catalyst system. Furthermore, the ability to use molecular hydrogen instead of stoichiometric chemical reducing agents eliminates the cost and waste disposal issues associated with reagents like sodium borohydride or formic acid. The operational simplicity of the process, which runs at near-ambient temperatures and moderate pressures, also reduces energy consumption and extends the lifespan of reactor equipment, contributing to lower overhead costs in the manufacturing facility.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high catalytic efficiency and the use of commodity chemicals. By achieving high yields with minimal catalyst loading, the direct material costs are significantly optimized. The elimination of expensive noble metals like rhodium and iridium in favor of the more abundant ruthenium further stabilizes the cost basis against market volatility. Additionally, the high selectivity reduces the loss of valuable starting materials into byproduct streams, maximizing the atom economy of the synthesis. The simplified workup procedure, resulting from the clean reaction profile, lowers the consumption of solvents and stationary phases required for purification, leading to substantial savings in waste treatment and utility expenses.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the accessibility of the raw materials required for this synthesis. The ligands and ruthenium precursors are commercially available or can be synthesized via established routes, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions means that the process is less susceptible to disruptions caused by utility fluctuations, such as variations in cooling water temperature or steam pressure. Moreover, the high stability of the catalyst system allows for longer batch cycles or potential continuous flow processing, which enhances throughput capacity without requiring proportional increases in capital investment. This reliability ensures that downstream API production schedules can be met consistently, minimizing the risk of stockouts for critical pharmaceutical products.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology aligns well with modern green chemistry principles. The use of hydrogen gas as the reductant produces water as the only byproduct, avoiding the generation of heavy metal waste or toxic organic residues associated with other reduction methods. The mild reaction conditions reduce the carbon footprint of the manufacturing process by lowering energy demand. Scalability is facilitated by the fact that the reaction kinetics remain favorable even at higher concentrations, allowing for smaller reactor volumes to produce the same amount of product. This intensification capability makes it easier to transition from pilot plant trials to full commercial production, ensuring that the supply chain can rapidly respond to increased market demand for the final pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing manufacturing workflows. The responses cover aspects ranging from catalyst performance metrics to operational parameters, providing a comprehensive overview for decision-makers.
Q: What are the advantages of this ruthenium catalyst over traditional rhodium or iridium catalysts?
A: The novel ruthenium compound offers significantly higher hydrogenation activity, allowing for milder reaction conditions and lower costs compared to expensive noble metals like rhodium and iridium.
Q: What level of stereoselectivity can be achieved with this method?
A: The method demonstrates exceptional stereoselectivity, achieving enantiomeric excess (ee) values greater than 99% and syn/anti diastereomer ratios exceeding 20:1 in optimized examples.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes inexpensive hydrogen gas as a hydrogen source and operates under scalable conditions, making it highly suitable for commercial production of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Aminoalcohol Supplier
The technological potential demonstrated in CN101983186B underscores the importance of having a manufacturing partner with deep expertise in organometallic catalysis and process development. NINGBO INNO PHARMCHEM stands ready to leverage this advanced ruthenium-catalyzed methodology to deliver high-quality chiral intermediates for the global pharmaceutical market. As a seasoned CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of optically active aminoalcohol meets the exacting standards required for API synthesis. We understand the critical nature of chirality in drug efficacy and safety, and our processes are designed to consistently deliver the high enantiomeric excess promised by this patent technology.
We invite you to collaborate with us to explore how this innovative catalytic route can enhance your supply chain and reduce your overall manufacturing costs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific target molecules, evaluating the economic impact of switching to this ruthenium-based process. We encourage you to contact our technical procurement team to request specific COA data from our pilot runs and to discuss route feasibility assessments for your project. By partnering with NINGBO INNO PHARMCHEM, you gain access to cutting-edge chemical technology combined with reliable, scalable manufacturing capacity, securing your position in the competitive pharmaceutical landscape.
