Scalable Electrocatalytic Production of 2,4,5-Trisubstituted Oxazole Derivatives for Pharma
Introduction to Electrocatalytic Oxazole Synthesis
The pharmaceutical and fine chemical industries are constantly seeking sustainable methodologies that align with green chemistry principles while maintaining high efficiency and selectivity. Patent CN115233243A introduces a groundbreaking preparation method for 2,4,5-trisubstituted oxazole derivatives under electrocatalysis, representing a significant paradigm shift from traditional thermal catalysis. This technology leverages electrical energy to drive the oxidative cyclization of alkynamide derivatives with nitrile solvents, effectively bypassing the need for stoichiometric oxidants or expensive transition metal catalysts. For R&D directors and process chemists, this approach offers a compelling alternative for constructing the oxazole scaffold, a privileged structure found in numerous bioactive molecules targeting neurological and infectious diseases. The ability to perform this transformation under mild conditions opens new avenues for the synthesis of complex pharmaceutical intermediates with improved environmental profiles.
Furthermore, the strategic importance of this patent lies in its operational simplicity and robustness, which are critical factors for industrial adoption. Unlike conventional methods that often demand stringent anhydrous and anaerobic conditions, this electrocatalytic protocol tolerates air and moisture, significantly reducing the engineering controls required for production. The use of readily available nitrile solvents as both the reaction medium and the carbon source for the oxazole ring adds another layer of atom economy to the process. As a reliable pharmaceutical intermediates supplier, understanding such innovative synthetic routes is essential for offering clients cost-effective and scalable solutions. This report delves into the technical nuances of this electrocatalytic system, highlighting its potential to redefine the manufacturing landscape for high-value heterocyclic compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenyloxazole and related derivatives has relied heavily on transition metal catalysis, involving reagents such as palladium, copper, or silver complexes. These traditional pathways, while effective, suffer from inherent drawbacks that pose significant challenges for large-scale manufacturing and regulatory compliance. Firstly, the reliance on precious metals introduces substantial raw material costs and creates a dependency on volatile commodity markets. Secondly, and perhaps more critically for the pharmaceutical sector, the presence of residual heavy metals in the final active pharmaceutical ingredient (API) or intermediate is strictly regulated under guidelines like ICH Q3D. Removing trace metals to parts-per-million levels often necessitates additional purification steps, such as scavenging treatments or repeated recrystallizations, which drastically reduce overall yield and increase processing time. Moreover, many conventional protocols require harsh reaction conditions, including high temperatures and strong bases, which can compromise the integrity of sensitive functional groups on the substrate.
The Novel Approach
In stark contrast, the methodology disclosed in CN115233243A utilizes an electrocatalytic strategy that replaces chemical oxidants with electrons, offering a cleaner and more direct route to 2,4,5-trisubstituted oxazoles. By employing a selenide compound as a redox mediator and carbon cloth electrodes, the system facilitates the oxidative cyclization at room temperature without the need for external metal catalysts. This transition from chemical to electrochemical driving force not only eliminates the risk of heavy metal contamination but also simplifies the reaction setup by allowing it to proceed in an open air atmosphere. The versatility of this approach is demonstrated by its compatibility with a wide range of substituents, including halogens, alkoxy groups, and alkyl chains, ensuring broad applicability across different drug scaffolds. 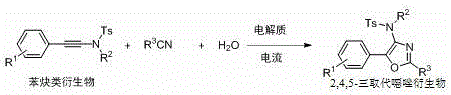
The operational advantages extend beyond the reaction vessel to the downstream processing stages. Since the reaction does not generate heavy metal waste streams, the environmental burden is significantly reduced, aligning with modern sustainability goals. The use of electricity as a reagent allows for precise control over the reaction rate and selectivity by adjusting the current density, providing a level of tunability that is difficult to achieve with thermal methods. For procurement managers, this translates to cost reduction in pharmaceutical intermediates manufacturing by lowering the total cost of ownership associated with catalyst recovery and waste disposal. The streamlined workflow, from mixing reagents to obtaining the purified product via standard column chromatography, underscores the practical viability of this technology for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Electrocatalytic Cyclization
The core of this innovation lies in the anodic oxidation mechanism mediated by the selenide species. Upon applying a constant current, the selenide catalyst undergoes oxidation at the anode to generate a reactive selenium cation or radical species. This electrophilic selenium intermediate then activates the alkyne moiety of the alkynamide derivative, facilitating a nucleophilic attack by the nitrogen atom of the nitrile solvent. This sequence initiates a cascade of intramolecular cyclization events that construct the oxazole ring system with high regioselectivity. The electrochemical regeneration of the active selenium species ensures that only catalytic amounts of the mediator are required, enhancing the atom economy of the process. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction parameters such as current density, electrode material, and electrolyte concentration to maximize yield and minimize side reactions.
From an impurity control perspective, the absence of transition metals fundamentally alters the impurity profile of the final product. Traditional metal-catalyzed reactions often produce metal-complexed byproducts or homocoupling products derived from the metal center. In this electrocatalytic system, the primary byproducts are likely to be organic in nature, arising from over-oxidation or competing nucleophilic attacks, which are generally easier to separate using standard chromatographic techniques. The mild reaction conditions (0-40°C) further suppress thermal degradation pathways, preserving the structural integrity of sensitive substituents. This clean reaction profile supports the production of high-purity oxazole derivatives, meeting the stringent quality specifications required for clinical trial materials. The ability to fine-tune the electrochemical potential offers an additional handle for suppressing specific side reactions, providing a robust platform for process development.
How to Synthesize 2,4,5-Trisubstituted Oxazoles Efficiently
The practical implementation of this electrocatalytic method is straightforward, requiring standard electrochemical equipment and commercially available reagents. The process begins with the preparation of the alkynamide starting materials, which can be efficiently synthesized from arylacetylene bromides and sulfonamides, ensuring a reliable supply chain for the precursors. 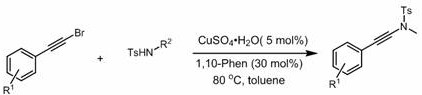
- Mix alkynamide derivatives, selenide compounds (catalyst), electrolyte, and nitrile solvent in a reactor.
- Apply constant current (e.g., 10 mA) using carbon cloth electrodes under air atmosphere at room temperature.
- After reaction completion, concentrate the mixture and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this electrocatalytic technology presents a multitude of strategic benefits that directly impact the bottom line and operational resilience. The elimination of precious metal catalysts removes a significant variable cost component and mitigates the supply risk associated with sourcing rare earth elements or noble metals. Furthermore, the simplified purification process reduces the consumption of solvents and adsorbents typically used for metal scavenging, leading to substantial cost savings in raw materials and waste management. The robustness of the reaction under air atmosphere lowers the capital expenditure required for specialized reactors capable of maintaining inert environments, making it an attractive option for retrofitting existing manufacturing facilities. These factors collectively contribute to a more agile and cost-efficient supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and ligands significantly lowers the direct material costs associated with the synthesis. Additionally, the avoidance of rigorous metal removal steps reduces the consumption of auxiliary chemicals and shortens the overall production cycle time. This streamlined process flow enhances throughput and reduces the energy footprint per kilogram of product, driving down the unit cost of goods sold. The use of electricity as a clean reagent further optimizes the cost structure by replacing costly chemical oxidants. These efficiencies allow for competitive pricing strategies while maintaining healthy margins in a challenging market environment.
- Enhanced Supply Chain Reliability: By relying on abundant and commercially available reagents such as nitriles and selenides, the supply chain becomes less vulnerable to geopolitical disruptions affecting rare metal supplies. The simplicity of the reaction conditions means that production can be easily distributed across multiple manufacturing sites without the need for highly specialized infrastructure. This decentralization capability enhances supply security and reduces lead times for high-purity oxazole derivatives. Moreover, the stability of the reagents and the tolerance to ambient conditions simplify logistics and storage requirements, ensuring consistent availability of key intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable, as reaction rates can be controlled by adjusting electrode surface area and current, facilitating a smooth transition from laboratory to pilot and commercial scales. The green nature of this synthesis, characterized by the absence of heavy metal waste and the use of mild conditions, aligns perfectly with increasingly strict environmental regulations. This compliance reduces the regulatory burden and potential liabilities associated with hazardous waste disposal. The ability to produce high-quality intermediates with a lower environmental impact strengthens the company's sustainability profile, appealing to eco-conscious partners and stakeholders in the global pharmaceutical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrocatalytic synthesis of oxazole derivatives, based on the detailed specifications provided in the patent literature. These insights are designed to clarify the feasibility and advantages of implementing this technology in a GMP environment. Understanding these details helps stakeholders make informed decisions about integrating this novel methodology into their existing portfolios. The answers reflect the current state of the art as described in the intellectual property documentation.
Q: Does this electrocatalytic method require expensive transition metal catalysts?
A: No, the method described in patent CN115233243A eliminates the need for expensive transition metal catalysts like palladium or copper, utilizing electricity and a selenide mediator instead.
Q: What are the reaction conditions regarding atmosphere and temperature?
A: The reaction proceeds efficiently under mild conditions, specifically at room temperature (0-40°C) and in an air atmosphere, removing the need for inert gas protection.
Q: How does this method impact the purification process for pharmaceutical intermediates?
A: By avoiding heavy metal catalysts, the downstream purification process is significantly simplified, as there is no need for rigorous metal scavenging steps to meet ICH Q3D guidelines.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trisubstituted Oxazoles Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrocatalytic synthesis in advancing the production of complex heterocyclic intermediates. Our team of expert process chemists is well-versed in adapting innovative academic and patent technologies into robust, GMP-compliant manufacturing processes. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale supply. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of 2,4,5-trisubstituted oxazoles we deliver. We are committed to being a strategic partner who not only supplies chemicals but also provides technical solutions that enhance your R&D efficiency.
We invite you to collaborate with us to leverage this cutting-edge electrocatalytic technology for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Together, we can accelerate your timeline to market while optimizing costs and adhering to the highest standards of quality and sustainability. Let us be your trusted partner in navigating the complexities of modern pharmaceutical synthesis.
