Revolutionizing Isoindigo Compound Production: Sustainable Catalytic Process for Commercial Scale-Up of Complex Electronic Materials
Patent CN113773243B introduces a groundbreaking method for preparing isoindigo compounds through organic catalytic oxidation of indole self-condensation, representing a significant advancement in the field of electronic materials synthesis that addresses critical limitations in traditional manufacturing approaches. This innovative approach utilizes commercially available organic bases as catalysts and elemental sulfur as an initiator, enabling the production of functionalized isoindigo compounds under mild conditions ranging from 25 to 100°C over 8 to 24 hours without requiring expensive transition metal catalysts or hazardous gaseous reagents. The process demonstrates exceptional functional group tolerance across diverse substrates while maintaining high reaction efficiency and excellent product purity characteristics essential for demanding optoelectronic applications where material quality directly impacts device performance metrics such as efficiency and longevity. By eliminating metal contamination risks entirely from the catalytic system, this method provides substantial advantages for manufacturers seeking reliable high-purity isoindigo compounds that meet stringent specifications required in advanced electronic material applications including organic solar cells and semiconductor devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing isoindigo compounds primarily rely on acid-catalyzed Aldol condensation of isatin and oxindole molecules which suffer from multiple significant drawbacks including complex reaction procedures requiring multiple steps, low efficiency resulting in inconsistent yields across different substrates, and poor scalability due to specialized equipment requirements that limit commercial viability at larger production volumes. Metal-catalyzed approaches introduce costly precious metal catalysts that create serious metal residue issues compromising product safety and performance in sensitive applications such as pharmaceuticals and optoelectronic devices where even trace metal contamination can significantly degrade device functionality and longevity. The previously developed method using carbonyl sulfide (COS) as a vulcanizing agent addresses metal residue concerns but presents new challenges including the high cost of COS gas which increases raw material expenses substantially, sensitivity to environmental conditions like moisture and oxygen which creates supply chain vulnerabilities through inconsistent reaction performance, and specialized handling requirements that increase operational complexity without delivering proportional benefits for large-scale manufacturing operations.
The Novel Approach
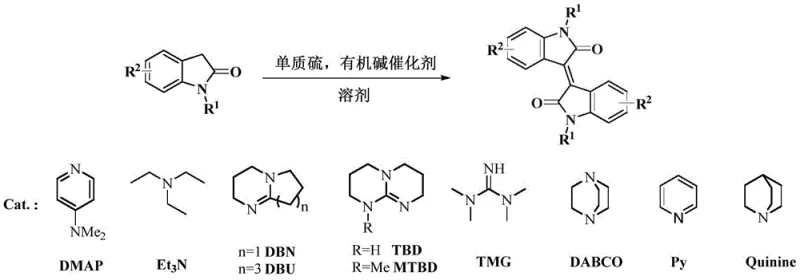
The patented method described in CN113773243B overcomes these limitations through an innovative organic catalytic system that employs elemental sulfur as a safe, inexpensive vulcanizing agent and commercially available organic bases as catalysts operating under mild temperature conditions between 25°C and 100°C without requiring specialized equipment or environmental controls typically needed for gaseous reagents or sensitive catalyst systems. This approach eliminates transition metals entirely from the catalytic cycle while maintaining high reaction efficiency across a broad range of functionalized substrates with documented yields ranging from 65% to 98% across multiple examples demonstrating exceptional functional group tolerance including halogenated derivatives, alkyl-substituted compounds, methoxy-containing structures, acetyl-functionalized products, and silicon-containing variants suitable for diverse electronic material applications requiring specific structural modifications.
Mechanistic Insights into Organic Base-Catalyzed Indole Self-Condensation
The reaction mechanism involves a unique benzylic position-specific sulfidation process where elemental sulfur acts as a vulcanizing agent under the influence of organic base catalysts to facilitate the conversion of oxindole substrates into thioisatin intermediates in situ through a well-defined catalytic cycle that activates the oxindole substrate at the benzylic position while facilitating sulfur incorporation without requiring isolation of unstable intermediates which represents a key innovation enabling efficient large-scale implementation. This green and efficient in-situ synthesis of thioisatin avoids stoichiometric use of expensive or hazardous reagents like Lawson's reagent or carbonyl sulfide that characterized previous approaches by utilizing catalytic amounts of organic bases including DMAP, Et₃N, DBN/DBU series catalysts with varying basicity profiles optimized for different substrate types while maintaining excellent reaction control across diverse functional groups commonly encountered in electronic material synthesis.
The method demonstrates exceptional control over impurity profiles through its selective reaction pathway that minimizes side reactions commonly observed in traditional metal-catalyzed processes by eliminating transition metals entirely from the catalytic system which avoids metal contamination issues that could compromise product performance in sensitive electronic applications where even trace metal residues can significantly degrade device functionality over time through mechanisms such as charge carrier trapping or accelerated degradation pathways under operational conditions. The mild reaction conditions (25-100°C) further contribute to impurity control by preventing thermal degradation pathways that often generate unwanted byproducts in higher temperature processes while enabling simple purification techniques such as organic solvent washing or column chromatography with standard eluents (dichloromethane/ethyl acetate or petroleum ether/ethyl acetate) to efficiently remove any minor impurities while maintaining high product purity suitable for demanding electronic material applications requiring consistent performance characteristics across production batches.
How to Synthesize Isoindigo Compounds Efficiently
This patented synthesis route represents a significant advancement in isoindigo compound manufacturing offering a streamlined process that eliminates traditional bottlenecks while maintaining high product quality and yield consistency across diverse structural variants required for specialized electronic material applications where precise molecular architecture directly impacts device performance metrics such as charge carrier mobility or light absorption characteristics essential for optoelectronic functionality. The method's simplicity combined with its adaptability to existing manufacturing infrastructure makes it particularly suitable for commercial scale-up in electronic materials production where reliability and purity are paramount concerns affecting both product performance and regulatory compliance requirements across global markets serving semiconductor manufacturers and display technology developers requiring consistent high-quality intermediates.
- Combine oxindole substrate with elemental sulfur (molar ratio 1: 1-1:10), organic base catalyst (molar ratio 1:1-1:10), and appropriate solvent in a pressure tube under magnetic stirring.
- Maintain reaction temperature between 25°C and 100°C for 8-24 hours depending on substrate reactivity and desired conversion rate.
- After reaction completion, remove solvent under vacuum followed by purification through column chromatography or organic solvent washing to obtain high-purity isoindigo compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process addresses critical pain points in traditional isoindigo compound production that have long challenged procurement and supply chain professionals in the electronic materials industry by eliminating reliance on expensive transition metal catalysts which creates significant cost drivers throughout the production value chain while introducing complex purification requirements that increase both processing time and resource consumption without delivering proportional benefits in terms of product quality or yield consistency required for commercial viability at scale.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes significant cost drivers from both raw material procurement and downstream processing stages by avoiding costly metal removal steps required in traditional methods which typically involve multiple purification cycles using specialized equipment that increases both capital expenditure requirements and operational costs per batch produced without enhancing final product quality characteristics essential for electronic material applications where purity specifications are already stringent enough without additional contamination risks from processing steps themselves.
- Enhanced Supply Chain Reliability: The reliance on commercially available stable reagents that are insensitive to environmental factors like moisture and oxygen significantly improves supply chain resilience by eliminating vulnerabilities associated with specialized or sensitive raw materials which previously created single-source dependencies requiring complex logistics management protocols that increased operational risk exposure during periods of market volatility or geopolitical disruptions affecting specialty chemical supply chains serving the electronics industry globally.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability from laboratory development through pilot scale to full commercial production volumes due to its simple reaction setup using standard manufacturing equipment without requiring specialized infrastructure investments typically needed for handling hazardous reagents or sensitive catalytic systems which simplifies technology transfer between development stages while reducing time-to-market timelines significantly compared to conventional approaches requiring substantial process re-engineering during scale-up phases.
Frequently Asked Questions (FAQ)
The following questions address key technical and commercial considerations regarding the implementation of this patented isoindigo compound manufacturing process based on detailed analysis of the patent documentation technical specifications provided by inventors along with practical insights derived from our extensive experience scaling similar organic synthesis pathways for electronic material applications where precise molecular control directly impacts final device performance metrics requiring consistent high-purity intermediates meeting exacting quality standards demanded by global semiconductor manufacturers.
Q: How does this organic catalytic method eliminate metal residue concerns compared to traditional transition metal-catalyzed approaches?
A: The patented process completely eliminates transition metal catalysts by utilizing commercially available organic bases as catalysts with elemental sulfur as the vulcanizing agent. This fundamental design change removes the need for expensive metal removal steps required in conventional methods where even trace metal residues can compromise performance in sensitive optoelectronic applications.
Q: What specific advantages does elemental sulfur offer as a vulcanizing agent over carbonyl sulfide or Lawson's reagent?
A: Elemental sulfur provides significant advantages including lower cost, greater stability under ambient conditions, reduced environmental impact, and simplified handling compared to gaseous carbonyl sulfide or hazardous Lawson's reagent. Its commercial availability and insensitivity to moisture/oxygen eliminate critical supply chain vulnerabilities associated with previous methods.
Q: How does this process maintain high functional group tolerance while achieving excellent yields across diverse isoindigo derivatives?
A: The mild reaction conditions (25-100°C) combined with the selective benzylic position-specific sulfidation mechanism enable compatibility with various functional groups including halogens, alkyl groups, methoxy groups, and acetyl groups without requiring protective groups or specialized reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isoindigo Compounds Supplier
Our patented manufacturing process represents a transformative advancement in isoindigo compound production delivering exceptional value for electronic materials manufacturers seeking reliable high-purity intermediates essential for next-generation optoelectronic applications where material quality directly determines device performance characteristics such as efficiency lifetime stability under operational conditions requiring consistent molecular architecture across production batches at commercial scale volumes meeting global market demands through our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through rigorous QC labs implementing advanced analytical protocols validated against industry standards.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your production requirements through our Customized Cost-Saving Analysis service which will demonstrate how our patented manufacturing process can optimize your supply chain while delivering high-purity isoindigo compounds meeting exacting specifications required for your advanced electronic material applications.
