Advanced Nickel-Catalyzed Synthesis of 2-Iminodihydroquinazolinones for Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access nitrogen-containing heterocycles, particularly the 2-iminodihydroquinazolinone scaffold, which serves as a critical precursor for various bioactive molecules. A significant breakthrough in this domain is detailed in patent CN110511188B, which discloses a novel preparation method utilizing a nickel-catalyzed decarboxylative cyclization strategy. This technology represents a paradigm shift from traditional multi-step syntheses, offering a direct, one-step route from readily available isatoic anhydrides and carbodiimides. By leveraging the unique reactivity of transition metal nickel, which exhibits superior stability towards water and oxygen compared to precious metals, this method achieves high conversion rates under remarkably mild conditions. For R&D directors and process chemists, this innovation opens new avenues for library synthesis, while for supply chain leaders, it promises a drastic simplification of raw material sourcing and inventory management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-imino-substituted dihydroquinazolinones has been plagued by significant operational hazards and procedural complexities. One prevalent method involves the palladium-catalyzed carbonylation of o-iodoanilines with carbodiimides, which necessitates the use of toxic carbon monoxide gas at high pressures, typically around 300 psi. 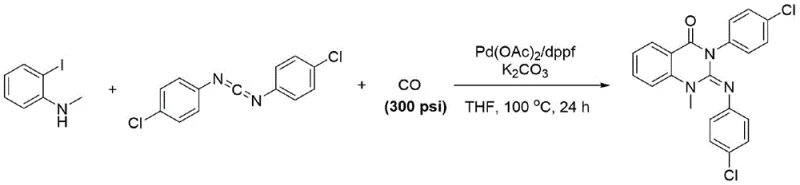 This requirement for high-pressure equipment not only escalates capital expenditure but also introduces severe safety risks regarding gas leakage and containment. Alternatively, another common route proceeds via a two-step sequence where isatoic anhydride is first converted to an anthranilamide, followed by cyclization using cyanogen bromide (CNBr).
This requirement for high-pressure equipment not only escalates capital expenditure but also introduces severe safety risks regarding gas leakage and containment. Alternatively, another common route proceeds via a two-step sequence where isatoic anhydride is first converted to an anthranilamide, followed by cyclization using cyanogen bromide (CNBr). 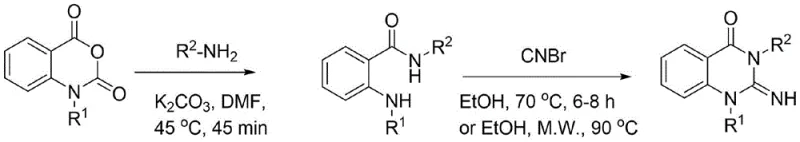 The use of cyanogen bromide is particularly problematic for large-scale manufacturing due to its extreme toxicity and the stringent regulatory controls surrounding its handling, storage, and waste disposal. These legacy methods create bottlenecks in production scalability and impose heavy burdens on environmental health and safety (EHS) compliance teams.
The use of cyanogen bromide is particularly problematic for large-scale manufacturing due to its extreme toxicity and the stringent regulatory controls surrounding its handling, storage, and waste disposal. These legacy methods create bottlenecks in production scalability and impose heavy burdens on environmental health and safety (EHS) compliance teams.
The Novel Approach
In stark contrast, the methodology described in CN110511188B utilizes a nickel-catalyzed system that merges the reactants in a single pot, effectively bypassing the need for hazardous reagents and extreme conditions. The core transformation involves the reaction of an isatoic anhydride derivative with a carbodiimide in the presence of a nickel catalyst, such as bis(triphenylphosphine)nickel chloride or nickel acetylacetonate. 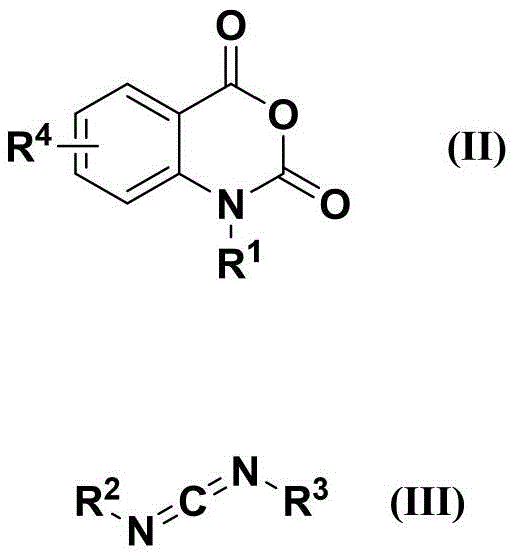 This process operates under a standard nitrogen atmosphere at moderate temperatures ranging from 30°C to 150°C, eliminating the need for high-pressure reactors entirely. The reaction is atom-economical, producing only carbon dioxide as a benign by-product, which aligns perfectly with green chemistry principles. For procurement managers, this translates to a supply chain that is no longer dependent on specialized high-pressure infrastructure or restricted toxic chemicals, thereby enhancing overall supply continuity and reducing regulatory friction.
This process operates under a standard nitrogen atmosphere at moderate temperatures ranging from 30°C to 150°C, eliminating the need for high-pressure reactors entirely. The reaction is atom-economical, producing only carbon dioxide as a benign by-product, which aligns perfectly with green chemistry principles. For procurement managers, this translates to a supply chain that is no longer dependent on specialized high-pressure infrastructure or restricted toxic chemicals, thereby enhancing overall supply continuity and reducing regulatory friction.
Mechanistic Insights into Nickel-Catalyzed Decarboxylative Cyclization
The success of this transformation lies in the unique ability of the nickel catalyst to facilitate a decarboxylative activation of the isatoic anhydride. Mechanistically, the nickel center coordinates with the anhydride, promoting the extrusion of carbon dioxide to generate a highly reactive cyclic nickel-amido intermediate. This organometallic species then undergoes an insertion reaction with the carbodiimide substrate, followed by reductive elimination or protonation to release the final 2-iminodihydroquinazolinone product and regenerate the active catalyst. This pathway is distinct from the nucleophilic attack mechanisms seen in the cyanogen bromide route, offering a different electronic environment that is far more forgiving of sensitive functional groups. Understanding this mechanism is crucial for process optimization, as it highlights the importance of ligand selection and catalyst loading in maintaining the stability of the nickel intermediate throughout the reaction cycle.
Furthermore, the robustness of this catalytic system is evidenced by its exceptional functional group tolerance, a key metric for R&D directors designing complex analogs. The reaction conditions are mild enough to preserve halogen substituents such as fluorine, chlorine, bromine, and iodine, which are often essential for subsequent cross-coupling reactions in drug discovery pipelines. Additionally, alkene and allyl groups remain intact, demonstrating that the nickel catalyst does not promote unwanted side reactions like hydrogenation or isomerization under these specific conditions. This selectivity ensures that the impurity profile of the crude product is significantly cleaner compared to harsher thermal methods, simplifying downstream purification processes like column chromatography or recrystallization. The ability to maintain structural integrity while forming the heterocyclic core adds substantial value to the synthetic utility of this method for generating diverse chemical libraries.
How to Synthesize 2-Iminodihydroquinazolinones Efficiently
Implementing this nickel-catalyzed protocol requires careful attention to the molar ratios of the starting materials and the choice of solvent to maximize yield and purity. The patent specifies that the isatoic anhydride and carbodiimide should be mixed in a molar ratio of approximately 1:1.1 to 1:1.5, ensuring that the carbodiimide is in slight excess to drive the equilibrium forward without generating excessive waste. Common organic solvents such as acetonitrile, toluene, tetrahydrofuran, or ethanol can be employed, providing flexibility based on solubility profiles of specific substrates. The detailed standardized synthesis steps, including specific catalyst loadings and workup procedures, are outlined in the guide below to ensure reproducibility across different laboratory scales.
- Mix isatoic anhydride and carbodiimide in a solvent such as acetonitrile or toluene with a molar ratio of 1: 1.1 to 1.5.
- Add a nickel catalyst (1-15 mol%) such as Ni(PPh3)2Cl2 or nickel acetylacetonate to the reaction mixture.
- Heat the mixture to 30-150°C under nitrogen protection for 2-24 hours, then purify via column chromatography or distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this nickel-catalyzed technology offers profound advantages in terms of cost structure and operational reliability. By shifting away from palladium-based systems and toxic cyanating agents, manufacturers can achieve significant cost reductions in pharmaceutical intermediate manufacturing. The elimination of high-pressure carbon monoxide removes the need for specialized autoclaves and rigorous gas monitoring systems, drastically lowering capital investment and maintenance overheads. Furthermore, the avoidance of cyanogen bromide mitigates the high costs associated with hazardous waste treatment and regulatory compliance, allowing for a leaner and more agile production model. These factors combined result in a more competitive pricing structure for the final intermediates, benefiting the entire value chain from raw material suppliers to end-product formulators.
- Cost Reduction in Manufacturing: The substitution of expensive palladium catalysts with inexpensive and earth-abundant nickel salts directly lowers the bill of materials for each batch. Additionally, the one-pot nature of the reaction reduces labor costs and solvent consumption by eliminating isolation steps required in multi-step sequences. The simplified workflow means less energy is consumed for heating and cooling cycles, and the reduced reaction time compared to sluggish thermal methods enhances overall plant throughput. These cumulative efficiencies translate into substantial economic savings without compromising the quality or purity of the output, making it an ideal candidate for cost-sensitive generic drug production.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like isatoic anhydrides and carbodiimides ensures a stable and resilient supply chain, as these materials are widely produced and less susceptible to geopolitical shortages than specialized reagents. The robustness of the nickel catalyst against moisture and oxygen simplifies logistics, as strict anhydrous conditions are not mandatory, reducing the risk of batch failures due to environmental exposure during transport or storage. This reliability allows supply chain heads to maintain lower safety stocks and respond more quickly to fluctuating market demands, ensuring continuous availability of critical intermediates for downstream API synthesis.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of toxic by-products make this process inherently scalable from gram-scale R&D to multi-ton commercial production. The generation of carbon dioxide as the sole by-product aligns with increasingly stringent environmental regulations, facilitating easier permitting and reducing the carbon footprint of the manufacturing facility. This environmental compatibility is a strategic asset for companies aiming to meet sustainability goals, as it minimizes the need for complex effluent treatment plants and reduces the liability associated with handling dangerous substances.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this nickel-catalyzed synthesis route. These insights are derived directly from the experimental data and scope defined in the patent documentation, providing clarity on substrate compatibility and process parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production lines or research workflows.
Q: What are the safety advantages of this nickel-catalyzed method over traditional synthesis?
A: This method eliminates the need for highly toxic cyanogen bromide (CNBr) and avoids the use of high-pressure carbon monoxide (CO) gas required in palladium-catalyzed routes, significantly improving operational safety and reducing hazardous waste disposal costs.
Q: Does this reaction tolerate sensitive functional groups like halogens?
A: Yes, the nickel-catalyzed decarboxylative cyclization demonstrates excellent functional group tolerance. Experimental data confirms that substituents such as fluoro, chloro, bromo, and iodo groups remain intact during the reaction, allowing for diverse downstream derivatization.
Q: What is the typical yield range for this one-step transformation?
A: Under optimized conditions using nickel catalysts like Ni(PPh3)2Cl2 at temperatures between 50°C and 120°C, the process consistently achieves high yields, often exceeding 80%, with some substrates reaching up to 88% isolated yield after purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Iminodihydroquinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this nickel-catalyzed technology in streamlining the production of high-value heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-iminodihydroquinazolinone meets the exacting standards required by global pharmaceutical clients. We are committed to leveraging advanced catalytic methods to deliver superior quality intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this nickel-catalyzed process can improve your margins. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, and let us help you build a more efficient and sustainable supply chain.
