Scalable Electrochemical Synthesis of 4-Seleno-1H-Pyrazole Compounds for Commercial Production
Introduction to Advanced Electrochemical Selenylation Technology
The landscape of heterocyclic synthesis is undergoing a paradigm shift driven by the urgent need for sustainable and cost-effective manufacturing processes. Patent CN113089006B introduces a groundbreaking electrochemical methodology for the synthesis of 4-seleno-1H-pyrazole compounds, a class of molecules increasingly recognized for their potent biological and pharmacological activities. This technology leverages electrons as traceless oxidants to facilitate the oxidative dehydrogenation coupling between 1H-pyrazoles and diorganyl diselenides, effectively bypassing the limitations of traditional chemical oxidation. By operating in an undivided cell under mild conditions, this approach not only achieves high selectivity and yield but also aligns perfectly with the principles of green chemistry, offering a compelling value proposition for pharmaceutical and agrochemical manufacturers seeking to optimize their supply chains.
For R&D directors and process chemists, the significance of this patent lies in its ability to access valuable selenium-containing scaffolds without the burden of heavy metal contamination or excessive salt waste. The method utilizes a simple electrolyte system comprising ammonium iodide in acetonitrile, which acts as a redox mediator to facilitate the transformation efficiently. This eliminates the need for stoichiometric amounts of hazardous oxidants such as potassium monopersulfate or molecular iodine, which are common in prior art methods. Consequently, the downstream processing is drastically simplified, reducing the environmental footprint and operational complexity associated with producing high-purity pharmaceutical intermediates on a commercial scale.
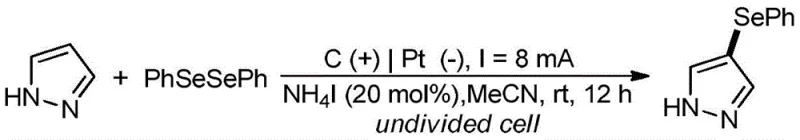
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-selenylpyrazole compounds has relied heavily on chemical oxidants and transition metal catalysts, presenting significant challenges for industrial scalability and cost efficiency. Traditional protocols often require stoichiometric amounts of strong oxidizing agents like Oxone or Selectfluor, which generate substantial quantities of inorganic salt byproducts that complicate product isolation and increase waste disposal costs. Furthermore, methods employing molecular iodine or hydrogen peroxide frequently necessitate the use of excess starting materials to drive the reaction to completion, resulting in poor atom economy and increased raw material consumption. These inefficiencies are compounded by the need for rigorous purification steps to remove residual metals and oxidant derivatives, which can be particularly problematic when targeting high-purity standards required for active pharmaceutical ingredients.
In addition to economic drawbacks, conventional chemical oxidation methods often suffer from limited functional group tolerance and harsh reaction conditions. Many existing procedures are restricted to N1-protected pyrazoles, limiting their applicability to a broader range of substrates. The use of strong acids or bases in some protocols can lead to the degradation of sensitive functional groups, thereby narrowing the scope of accessible chemical space. Moreover, the reliance on expensive reagents and the generation of hazardous waste streams pose significant regulatory and environmental compliance hurdles for manufacturing facilities. These factors collectively hinder the widespread adoption of selenium-functionalized pyrazoles in drug discovery and development pipelines, creating a clear demand for more sustainable and efficient synthetic alternatives.
The Novel Approach
The electrochemical strategy disclosed in CN113089006B represents a transformative solution to these longstanding challenges by replacing chemical oxidants with electricity. This novel approach utilizes an undivided electrochemical cell where the anodic oxidation of iodide ions generates reactive iodine species in situ, which then mediate the activation of the diselenide bond. This catalytic cycle allows for the efficient coupling of 1H-pyrazoles and diselenides under remarkably mild conditions, typically at room temperature and atmospheric pressure. The absence of external chemical oxidants means that the only byproduct of the reaction is hydrogen gas at the cathode, which evolves harmlessly, thereby maximizing atom economy and minimizing waste generation. This "green" profile makes the process inherently safer and more environmentally friendly compared to traditional methods.
From a practical standpoint, this electrochemical method offers exceptional versatility and ease of operation. The reaction proceeds smoothly with a wide variety of substrates, including those bearing electron-donating and electron-withdrawing substituents, demonstrating robust functional group compatibility. The use of inexpensive and readily available electrode materials, such as graphite rods and platinum sheets, further enhances the economic viability of the process. Additionally, the reaction does not require stringent exclusion of air or moisture, simplifying the operational setup and reducing the infrastructure costs associated with inert atmosphere handling. For procurement and supply chain managers, this translates to a more reliable and cost-effective sourcing strategy for complex heterocyclic intermediates, as the process is less susceptible to fluctuations in the price of specialized chemical reagents.
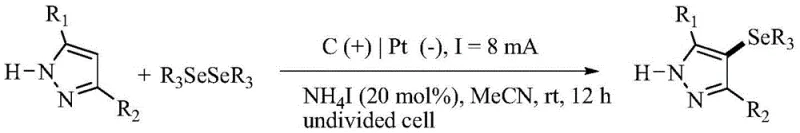
Mechanistic Insights into Electrochemical Oxidative Dehydrogenation Coupling
Understanding the mechanistic underpinnings of this electrochemical transformation is crucial for optimizing reaction parameters and ensuring consistent product quality. The proposed mechanism involves a dual-pathway process mediated by the iodide/ammonium iodide electrolyte system. In the first pathway, iodide anions are oxidized at the anode surface to form iodine cations or reactive iodine species, which subsequently react with the diorganyl diselenide to generate a phenylselenium iodide intermediate. This electrophilic selenium species then attacks the electron-rich C4 position of the 1H-pyrazole ring, forming a cationic intermediate that undergoes deprotonation and rearomatization to yield the final 4-selenylpyrazole product. Simultaneously, protons generated during the aromatization process are reduced at the cathode to form hydrogen gas, completing the electrochemical circuit without the accumulation of acidic byproducts.
Alternatively, a radical pathway may also operate concurrently, particularly depending on the specific substrate and potential applied. In this scenario, the diselenide undergoes single-electron oxidation at the anode to form a radical cation, which fragments to produce a selenyl radical. This radical species adds to the pyrazole ring to form a carbon-centered radical intermediate, which is subsequently oxidized at the anode to a cation before losing a proton to restore aromaticity. Experimental evidence, such as radical trapping studies using TEMPO, supports the involvement of radical intermediates in the reaction manifold. This mechanistic flexibility allows the system to accommodate a diverse range of electronic environments on both the pyrazole and diselenide components, ensuring high yields across a broad substrate scope. The interplay between these ionic and radical pathways underscores the robustness of the electrochemical method in handling complex molecular architectures.
How to Synthesize 4-Seleno-1H-Pyrazole Efficiently
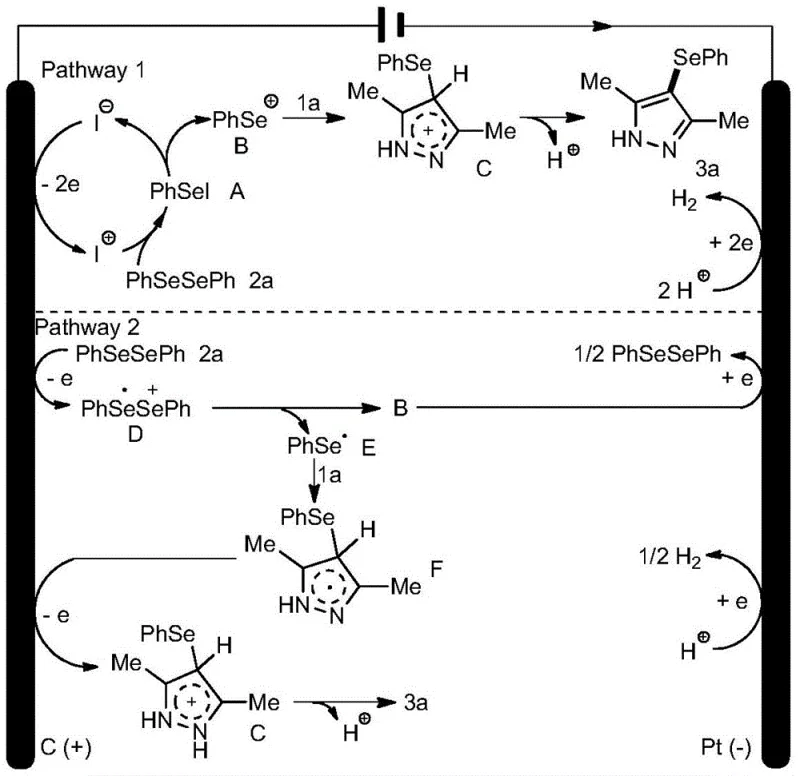
Implementing this electrochemical synthesis protocol requires careful attention to reaction parameters to maximize yield and efficiency. The standard procedure involves dissolving the 1H-pyrazole substrate and the diorganyl diselenide in acetonitrile, which has been identified as the optimal solvent for balancing solubility and conductivity. Ammonium iodide is added as the supporting electrolyte at a concentration of approximately 20 mol% relative to the substrate, serving as the crucial redox mediator. The reaction mixture is then subjected to constant current electrolysis in an undivided cell equipped with a graphite anode and a platinum cathode. Maintaining a current density of around 8 mA at room temperature for 12 hours typically provides the best balance between reaction rate and selectivity, avoiding over-oxidation or side reactions that might occur at higher potentials.
- Prepare the electrolyte solution by dissolving the 1H-pyrazole substrate and diorganyl diselenide in acetonitrile with ammonium iodide as the supporting electrolyte.
- Set up an undivided electrochemical cell equipped with a graphite rod anode and a platinum sheet cathode, ensuring proper electrode spacing.
- Apply a constant direct current of 8 mA at room temperature for 12 hours under air or nitrogen atmosphere to drive the oxidative dehydrogenation coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology offers tangible strategic advantages that extend beyond mere technical feasibility. The elimination of stoichiometric chemical oxidants and transition metal catalysts directly addresses two major cost drivers in fine chemical manufacturing: raw material expenses and waste treatment costs. By removing the need for expensive reagents like Selectfluor or large quantities of iodine, the variable cost per kilogram of the product is significantly reduced. Furthermore, the simplified workup procedure, which avoids complex extraction and purification steps to remove metal residues, leads to shorter production cycles and higher overall throughput. This efficiency gain allows manufacturers to respond more agilely to market demands and reduce the lead time for delivering high-purity intermediates to downstream customers.
- Cost Reduction in Manufacturing: The primary economic benefit stems from the substitution of costly chemical oxidants with electricity, which is generally cheaper and more consistent in price. Since the reaction does not require transition metal catalysts, there is no need for expensive metal recovery processes or rigorous testing for residual metal limits, which are critical in pharmaceutical production. The high atom economy of the reaction ensures that a larger proportion of the starting materials ends up in the final product, minimizing raw material waste. Additionally, the use of simple, durable electrode materials like graphite reduces capital expenditure on specialized reactor hardware, making the technology accessible for both pilot-scale and full-scale production facilities.
- Enhanced Supply Chain Reliability: Relying on electricity as the primary "reagent" decouples the production process from the volatility of the global chemical reagent market. Unlike specialized oxidants that may face supply disruptions or price spikes, electrical power is a stable and ubiquitous utility. The robustness of the reaction conditions, which tolerate air and moisture, further reduces the risk of batch failures due to environmental factors. This reliability ensures a consistent supply of critical intermediates, mitigating the risk of production delays for downstream API manufacturing. The ability to source simple starting materials like pyrazoles and diselenides from multiple vendors also diversifies the supply base, enhancing overall supply chain resilience against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The mild operating conditions of this electrochemical process make it inherently scalable. Unlike exothermic chemical oxidations that require careful thermal management to prevent runaway reactions, electrochemical reactions can be precisely controlled by adjusting the current. This controllability facilitates the transition from laboratory benchtop to industrial flow reactors or large batch tanks with minimal re-optimization. From an environmental perspective, the generation of hydrogen gas as the sole byproduct aligns with increasingly stringent environmental regulations regarding waste discharge. The absence of heavy metal waste and inorganic salt sludge simplifies compliance reporting and reduces the liability associated with hazardous waste disposal, positioning the manufacturer as a leader in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 4-seleno-1H-pyrazole compounds. These insights are derived directly from the experimental data and optimization studies detailed in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these aspects is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows and for assessing its potential impact on product quality and cost structures.
Q: What are the primary advantages of this electrochemical method over traditional oxidation?
A: This method eliminates the need for stoichiometric chemical oxidants like Oxone or molecular iodine and avoids expensive transition metal catalysts, significantly reducing chemical waste and purification costs while maintaining high atom economy.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under mild conditions at room temperature with simple electrode materials (graphite and platinum), making it highly amenable to scale-up in flow electrochemical reactors or larger batch cells without complex safety measures.
Q: What is the functional group tolerance of this selenylation reaction?
A: The protocol demonstrates excellent compatibility with various substituents on both the pyrazole ring and the diselenide, including electron-donating and electron-withdrawing groups like halogens, methoxy, nitro, and cyano groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Seleno-1H-Pyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex heterocyclic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113089006B can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global pharmaceutical clients. We are committed to leveraging our technical expertise to deliver high-quality 4-seleno-1H-pyrazole derivatives that meet the exacting standards of the life sciences industry.
We invite you to collaborate with us to explore how this sustainable synthesis route can enhance your supply chain efficiency and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data for our available selenium-containing intermediates and to discuss route feasibility assessments for your custom synthesis projects. Together, we can drive innovation and sustainability in the production of next-generation pharmaceutical building blocks.
