Advanced Manufacturing of High-Purity Trifluoromethyl Triazoles: Scalable Glucose-Based Synthesis for Pharmaceutical Supply Chains
Patent CN113880781A introduces a transformative methodology for synthesizing biologically significant 3-trifluoromethyl-substituted 1,2,4-triazole compounds through an innovative glucose-based carbon source strategy. This breakthrough directly addresses longstanding challenges in pharmaceutical intermediate manufacturing by eliminating stringent anhydrous and anaerobic conditions that have historically complicated traditional syntheses. The process utilizes readily available biomass-derived glucose and trifluoroethylimine hydrazide under mild thermal parameters of 70–90°C for precisely controlled reaction durations of 2–4 hours with triflic acid catalysis. Notably, this approach achieves exceptional efficiency without requiring expensive transition metal catalysts or specialized equipment, thereby reducing both capital expenditure and operational complexities across production scales. The patent explicitly demonstrates scalability from gram-level laboratory validation to potential industrial implementation, offering pharmaceutical manufacturers a sustainable pathway to high-purity intermediates while enhancing supply chain resilience through simplified logistics and reduced environmental footprint.
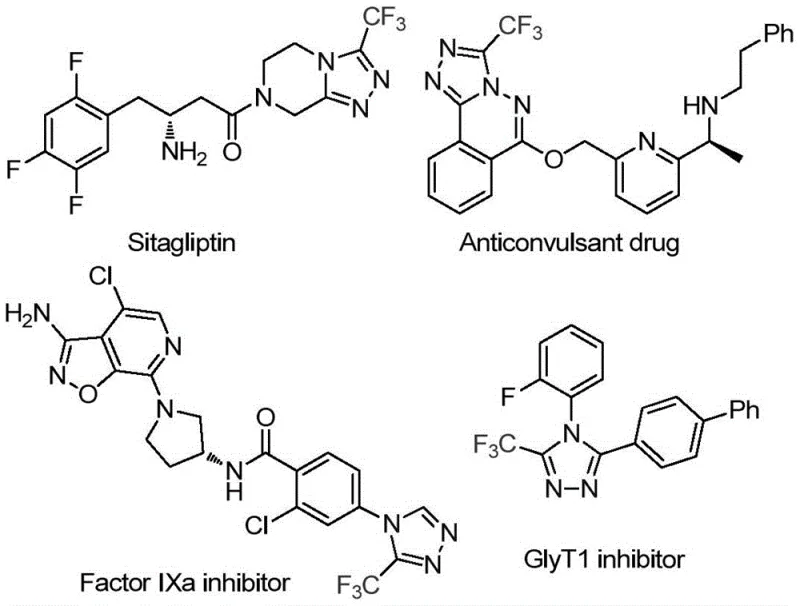
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of trifluoromethyl-substituted triazoles frequently rely on transition metal-catalyzed reactions that necessitate strictly anhydrous and anaerobic environments to prevent catalyst deactivation and unwanted side reactions. These demanding conditions require specialized infrastructure including gloveboxes and solvent purification systems that significantly increase capital investment while complicating process validation under GMP standards. Furthermore, conventional routes often employ expensive fluorinated building blocks with limited structural diversity, restricting the ability to tailor molecular properties for specific pharmacological applications. The necessity for extensive metal removal steps during purification generates hazardous waste streams that require costly treatment protocols while simultaneously reducing overall process efficiency through multiple intermediate isolations. These cumulative limitations create substantial barriers to scalable production and consistent quality control that directly impact both manufacturing costs and supply chain reliability for critical pharmaceutical intermediates.
The Novel Approach
The patented methodology overcomes these constraints through a fundamentally redesigned reaction pathway that leverages glucose as a sustainable carbon source under ambient atmospheric conditions. By utilizing triflic acid as a dual-function catalyst that promotes both glucose cleavage and subsequent cyclization without sensitivity to moisture or oxygen, the process eliminates all requirements for specialized handling equipment while maintaining high reaction efficiency. The strategic selection of commercially available trifluoroethylimine hydrazide enables precise structural modification through simple aryl group substitutions without necessitating process reoptimization. This innovative approach achieves comparable or superior yields to conventional methods while operating at significantly lower temperatures (70–90°C versus typical >150°C) with reduced reaction times (2–4 hours versus multi-step sequences). Crucially, the absence of transition metals removes entire purification stages related to metal residue removal, thereby streamlining production workflows and enhancing both cost-effectiveness and environmental sustainability across the manufacturing lifecycle.
Mechanistic Insights into Glucose-Mediated Triflic Acid Catalysis
The reaction mechanism initiates with acid-catalyzed cleavage of glucose under triflic acid promotion to generate reactive aldehyde intermediates that readily undergo condensation with trifluoroethylimine hydrazide. This forms hydrazone species that subsequently undergo intramolecular nucleophilic addition to establish the triazole ring framework through a cascade cyclization process. The critical role of tert-butyl hydroperoxide as a mild oxidant facilitates final aromatization without generating hazardous byproducts or requiring elevated temperatures beyond the optimal reaction window of 70–90°C. This carefully orchestrated sequence avoids unstable intermediates common in alternative pathways while maintaining excellent functional group tolerance across diverse aryl substituents. The solvent system comprising dioxane with controlled water addition creates an ideal microenvironment that balances reactant solubility with catalytic activity while preventing unwanted hydrolysis side reactions that could compromise yield or purity.
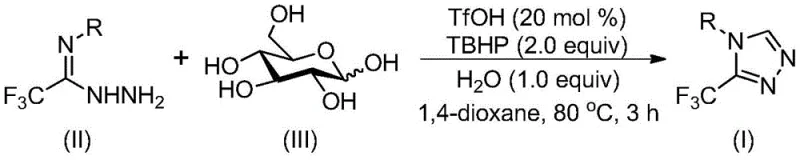
Impurity control is inherently achieved through the reaction's self-regulating nature where water acts as both reactant and moderator to prevent overoxidation or decomposition pathways. The absence of transition metals eliminates metal-derived impurities that typically require complex chelation or chromatographic removal steps in conventional processes. Careful optimization of reactant stoichiometry (trifluoroethylimine hydrazide : glucose at 2:1 ratio) ensures complete conversion while minimizing dimerization or oligomerization byproducts. The patent demonstrates how precise temperature control within the narrow window of 80±5°C prevents thermal degradation while maintaining sufficient kinetic energy for cyclization completion. This integrated approach to impurity management results in consistently high-purity products requiring only straightforward silica gel filtration followed by standard column chromatography purification without specialized equipment or additional processing stages.
How to Synthesize Trifluoromethyl Triazoles Efficiently
This patented synthesis route represents a significant advancement in manufacturing efficiency for critical pharmaceutical intermediates through its innovative use of renewable biomass feedstocks under operationally simple conditions. The methodology eliminates multiple pain points associated with traditional approaches including hazardous reagent handling requirements and complex purification protocols while maintaining excellent structural flexibility for diverse applications. Detailed standard operating procedures have been developed based on the patent specifications to ensure consistent quality across production scales from laboratory validation through commercial manufacturing phases. The following standardized synthesis protocol provides step-by-step guidance for implementing this breakthrough technology within existing manufacturing frameworks while optimizing both quality control parameters and resource utilization metrics.
- Combine trifluoroethylimine hydrazide (2.0 mmol), glucose (1.0 mmol), triflic acid (0.2 mmol), TBHP aqueous solution (2.0 mmol), water (1.0 mmol), and dioxane solvent (5 mL per mmol glucose) in a reaction vessel under ambient conditions.
- Heat the mixture to 80°C with continuous stirring for three hours while monitoring reaction progression through standard analytical techniques to ensure complete conversion.
- Perform post-reaction processing by filtration through silica gel followed by column chromatography purification using standard elution protocols to isolate high-purity trifluoromethyl triazole compounds.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing approach directly addresses critical pain points in pharmaceutical supply chains by transforming traditionally complex intermediate production into a streamlined process with enhanced reliability and cost-effectiveness. The elimination of specialized infrastructure requirements significantly reduces capital expenditure barriers while improving facility utilization rates across existing manufacturing networks. By leveraging globally available biomass feedstocks instead of scarce fluorinated precursors, the process creates more resilient supply chains less vulnerable to geopolitical disruptions or single-source dependencies that frequently impact traditional chemical manufacturing routes.
- Cost Reduction in Manufacturing: The complete removal of transition metal catalysts inherently eliminates multiple costly processing stages including metal removal protocols and associated waste treatment procedures that typically consume significant operational resources. This fundamental process simplification delivers substantial cost savings through reduced raw material consumption, lower energy requirements from milder reaction conditions (70–90°C versus >150°C), and minimized solvent usage from fewer purification steps while maintaining high product quality standards required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Utilizing glucose as a globally available biomass feedstock creates more robust supply chains compared to routes dependent on specialized fluorinated building blocks with limited suppliers. The simplified process design reduces vulnerability to equipment failures since standard reactors can be used without specialized moisture control systems while maintaining consistent output quality through fewer critical control points during manufacturing operations.
- Scalability and Environmental Compliance: The demonstrated scalability from gram-scale laboratory validation to potential multi-ton production capacity provides immediate pathways for commercial implementation without requiring major process reengineering. The elimination of hazardous metal residues significantly reduces environmental impact while simplifying waste stream management through fewer processing stages and inherently safer reaction conditions that align with green chemistry principles increasingly mandated by regulatory frameworks worldwide.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement specialists and R&D teams regarding implementation of this patented manufacturing technology within existing pharmaceutical supply chains. Each response is derived directly from experimental data documented in patent CN113880781A to ensure technical accuracy while addressing practical commercial concerns related to quality assurance and process integration requirements.
Q: How does this glucose-based method eliminate anhydrous/anaerobic requirements compared to traditional syntheses?
A: The patent demonstrates that glucose's inherent stability under aqueous conditions and triflic acid's dual role as catalyst and water-tolerant promoter enable reactions without moisture-sensitive intermediates. This eliminates costly glovebox operations and specialized drying equipment required in conventional metal-catalyzed routes.
Q: What cost advantages arise from using biomass-derived glucose as carbon source?
A: Glucose's commercial availability as a renewable feedstock significantly reduces raw material costs while avoiding expensive fluorinated precursors. The simplified purification process from eliminating transition metal residues further lowers operational expenses through reduced waste treatment requirements.
Q: How does substrate design flexibility enhance pharmaceutical intermediate applicability?
A: The patent documents demonstrate how varying aryl substituents on trifluoroethylimine hydrazide allows precise tuning of molecular properties while maintaining high yields. This modular approach enables rapid development of custom intermediates meeting specific pharmacological requirements without process revalidation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our patented technology represents a significant advancement in sustainable pharmaceutical intermediate manufacturing that aligns perfectly with evolving industry demands for both high-quality products and environmentally responsible production practices. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications through our state-of-the-art QC labs equipped with advanced analytical capabilities specifically validated for triazole compound characterization. Our dedicated technical teams have successfully implemented similar glucose-based catalytic processes across multiple therapeutic categories, ensuring seamless integration into your existing supply chain infrastructure while meeting all regulatory requirements for pharmaceutical intermediates.
We invite you to initiate a Customized Cost-Saving Analysis tailored to your specific production needs by contacting our technical procurement team today. Request detailed COA data and route feasibility assessments to evaluate how this innovative synthesis method can enhance your manufacturing efficiency while ensuring reliable supply of high-purity trifluoromethyl triazole intermediates for your critical drug development programs.
