Advanced Asymmetric Synthesis of Chiral Five-Membered Thiaheterocyclic Nucleoside Analogues for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust methodologies for constructing complex chiral scaffolds, particularly those relevant to antiviral therapeutics. Patent CN112300176B discloses a groundbreaking method for synthesizing chiral five-membered thiaheterocyclic nucleoside analogues via an asymmetric [3+2] cyclization reaction. This technology addresses the critical demand for efficient access to thianucleoside structures, which are pivotal in treating HIV and other viral infections, exemplified by drugs like Lamivudine and Emtricitabine. Unlike conventional approaches that rely on laborious multi-step sequences or expensive chiral starting materials, this invention utilizes readily available achiral substrates—specifically substituted electron-deficient olefins and 2,5-dihydroxy-1,4-dithiane—to directly forge the chiral tetrahydrothiophene core. The process employs a sophisticated catalytic system comprising a Nickel(II) or Iron(II) Lewis acid paired with chiral oxazoline ligands, achieving remarkable stereocontrol and high isolated yields.
![General reaction scheme for asymmetric [3+2] cyclization of electron-deficient olefins and dithiane](/insights/img/chiral-thiaheterocyclic-nucleoside-synthesis-supplier-20260308073435-01.webp)
For R&D directors evaluating process viability, the ability to generate diverse structural analogues from a single platform is invaluable. The reaction tolerates a wide array of functional groups on the olefin substrate, including purines, phthalimides, succinimides, indoles, and benzimidazoles, allowing for rapid library generation for structure-activity relationship (SAR) studies. Furthermore, the mild reaction conditions, typically proceeding at room temperature in solvents like mesitylene or dichloromethane, minimize energy consumption and reduce the risk of thermal degradation of sensitive nucleobase moieties. This technological leap not only accelerates the discovery phase but also lays a solid foundation for process chemistry teams aiming to transition from milligram-scale screening to kilogram-scale production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral five-membered thiaheterocyclic nucleosides has been plagued by significant synthetic inefficiencies that hinder both cost-effectiveness and scalability. Traditional strategies generally fall into two categories: either elaborating a multi-step reaction sequence to build a multifunctional chiral tetrahydrothiophene ring followed by coupling with a base, or introducing an amino group onto a pre-formed chiral ring to construct the base in situ. Both pathways suffer from the inherent drawback of requiring stoichiometric amounts of expensive chiral sources, which drastically inflates the raw material costs. Moreover, these multi-step protocols often result in cumulative yield losses, where the overall efficiency drops precipitously with each additional transformation. The preparation of the necessary chiral substrates themselves is frequently difficult and resource-intensive, creating a bottleneck in the supply chain. Additionally, the reliance on harsh conditions or sensitive reagents in older methods can lead to broad impurity profiles, necessitating complex and costly purification steps to meet the stringent purity specifications required for active pharmaceutical ingredients (APIs).
The Novel Approach
The innovative methodology described in the patent represents a paradigm shift by leveraging asymmetric catalysis to overcome these historical barriers. By employing an asymmetric [3+2] cyclization strategy, the process directly assembles the chiral five-membered heterocyclic core from simple, achiral building blocks. This atom-economical approach eliminates the need for pre-existing chiral centers in the starting materials, thereby significantly reducing the cost of goods sold (COGS). The use of earth-abundant transition metals like Nickel or Iron, rather than precious metals, further enhances the economic profile of the synthesis. Crucially, the reaction demonstrates exceptional stereoselectivity, with diastereomeric ratios (dr) often exceeding 20:1 and enantiomeric excess (ee) values reaching up to 94%. This high level of stereocontrol simplifies downstream processing, as fewer isomers are generated, reducing the burden on chromatographic separation. The versatility of the method is evidenced by its compatibility with various electron-deficient olefins, enabling the synthesis of a broad spectrum of nucleoside analogues with different heterocyclic bases and ester substituents in high yields.
Mechanistic Insights into Ni(II)-Catalyzed Asymmetric [3+2] Cyclization
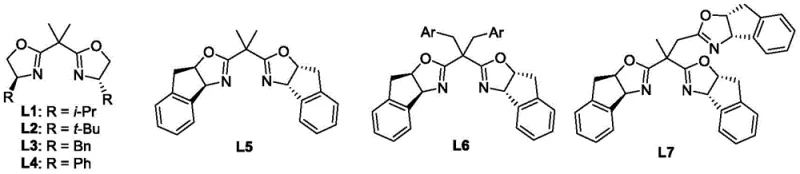
The success of this transformation hinges on the precise interplay between the Lewis acid catalyst and the chiral ligand environment. The reaction mechanism involves the activation of the electron-deficient olefin by the Nickel(II) species, which coordinates with the carbonyl oxygen to increase the electrophilicity of the beta-carbon. Simultaneously, the chiral bis-oxazoline ligand creates a rigid, sterically defined pocket around the metal center. This chiral environment dictates the facial selectivity of the nucleophilic attack by the sulfur atom of the 2,5-dihydroxy-1,4-dithiane. Among the library of ligands screened, the bulky bis-oxazoline ligand L7, featuring fused aromatic systems, proved to be superior in inducing asymmetry. The steric bulk of L7 effectively blocks one face of the coordinated olefin, forcing the dithiane to approach from the less hindered trajectory, thus establishing the absolute configuration of the newly formed stereocenters with high fidelity.
From an impurity control perspective, the robustness of this catalytic cycle is paramount for commercial manufacturing. The high diastereoselectivity observed (>20:1 dr in many cases) indicates that the transition state leading to the minor diastereomer is significantly higher in energy, effectively suppressing its formation. This minimizes the generation of hard-to-remove diastereomeric impurities that often co-elute during purification. Furthermore, the use of Ni(OTf)2 as the Lewis acid ensures rapid turnover and complete conversion of the starting materials, reducing the presence of unreacted olefins or dithiane in the crude mixture. The reaction proceeds smoothly in solvents like mesitylene or cyclopentyl methyl ether (CPME), which are known for their stability and ease of removal. The mechanistic understanding allows process chemists to fine-tune parameters such as temperature and concentration to maximize the ratio of the desired enantiomer, ensuring that the final product meets the rigorous quality standards demanded by regulatory bodies for antiviral drug intermediates.
How to Synthesize Chiral Five-Membered Thiaheterocyclic Nucleoside Analogues Efficiently
The practical execution of this synthesis is designed for operational simplicity while maintaining high performance. The general procedure involves charging a reaction vessel with the substituted electron-deficient olefin and the dithiane precursor under an inert atmosphere to prevent oxidation or moisture interference. The catalyst system, consisting of Ni(OTf)2 and ligand L7, is introduced in specific molar ratios optimized for maximum turnover. The reaction mixture is then stirred at ambient temperature, allowing the cyclization to proceed over a period of approximately three days. Upon completion, monitored by thin-layer chromatography (TLC), the reaction is quenched and subjected to a standard aqueous workup. The organic layer is separated, dried over anhydrous sodium sulfate, and concentrated. Final purification is achieved via column chromatography, yielding the target chiral nucleoside analogue as a white solid or oil with high optical purity. For detailed operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining substituted electron-deficient olefin and 2,5-dihydroxy-1,4-dithiane in an organic solvent such as mesitylene or dichloromethane under a nitrogen atmosphere.
- Add the Lewis acid catalyst, preferably Ni(OTf)2, and the chiral oxazoline ligand L7 to the reaction vessel, ensuring precise molar ratios for optimal stereocontrol.
- Stir the reaction at room temperature for approximately 3 days, then proceed with standard workup procedures including extraction, drying, and column chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this asymmetric [3+2] cyclization technology offers transformative benefits in terms of cost structure and supply reliability. The most significant advantage lies in the drastic reduction of raw material costs. By shifting from expensive, multi-step chiral pool synthesis to a direct catalytic assembly using commodity chemicals, the overall cost of manufacturing is significantly lowered. The elimination of precious metal catalysts in favor of nickel or iron salts further contributes to substantial cost savings, as these base metals are far more abundant and affordable. Additionally, the high yields and selectivity reduce the amount of waste generated per kilogram of product, aligning with green chemistry principles and potentially lowering waste disposal fees. This efficiency translates directly into a more competitive pricing model for the final pharmaceutical intermediate, providing a strategic edge in a cost-sensitive market.
- Cost Reduction in Manufacturing: The process eliminates the need for stoichiometric chiral auxiliaries and reduces the number of synthetic steps, which inherently lowers labor, energy, and material costs. The use of non-precious metal catalysts avoids the volatility associated with rhodium or palladium pricing. Furthermore, the high atom economy of the [3+2] cyclization means less raw material is wasted as byproducts, optimizing the input-output ratio. These factors combined create a leaner manufacturing process that is resilient to raw material price fluctuations.
- Enhanced Supply Chain Reliability: The starting materials, such as substituted acrylates and dithiane derivatives, are commercially available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate room temperature and common organic solvents, simplifies the engineering requirements for production facilities. This ease of execution ensures consistent batch-to-batch quality and reliable delivery schedules, crucial for maintaining uninterrupted API production lines. The scalability of the method from gram to ton scale has been demonstrated, ensuring that supply can ramp up quickly to meet market demand without extensive process re-engineering.
- Scalability and Environmental Compliance: The reaction operates under mild conditions without the need for cryogenic temperatures or high-pressure equipment, reducing the capital expenditure required for scale-up. The solvents used, such as mesitylene and CPME, have favorable environmental profiles and can be efficiently recovered and recycled, minimizing the environmental footprint. The high selectivity reduces the load on purification units, leading to lower solvent consumption during chromatography. This alignment with sustainable manufacturing practices not only meets regulatory compliance but also enhances the corporate sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature, providing clarity on the method's capabilities and limitations. Understanding these details is essential for project managers assessing the feasibility of integrating this route into their existing manufacturing portfolios. The high degree of stereocontrol and substrate tolerance makes this method a versatile tool for producing a wide range of bioactive molecules.
Q: What are the primary advantages of this asymmetric [3+2] cyclization method over traditional synthesis routes?
A: This method utilizes cheap, achiral raw materials to directly construct the chiral center, eliminating the need for expensive chiral pools and multi-step sequences. It achieves high yields (up to 95%) and excellent stereoselectivity (>20:1 dr, up to 94% ee) under mild conditions.
Q: Which catalyst system provides the best enantioselectivity for this transformation?
A: The combination of Nickel(II) triflate (Ni(OTf)2) as the Lewis acid and the bis-oxazoline ligand L7 has been identified as the optimal catalytic system, delivering superior yields and enantiomeric excess compared to other metal salts or ligand variants.
Q: Can the resulting chiral tetrahydrothiophene skeletons be further functionalized?
A: Yes, the products serve as versatile intermediates that can undergo various derivatizations, including reduction to dihydroxy compounds, fluorination to introduce fluoro-substituents, and elimination reactions to form dihydrothiophenes or sulfolanes, expanding their utility in drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Five-Membered Thiaheterocyclic Nucleoside Analogue Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development and commercialization. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle the specific requirements of asymmetric catalysis, including strict moisture control and specialized filtration systems for catalyst removal. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral thiaheterocyclic nucleoside analogue meets the highest international standards for pharmaceutical intermediates. Our commitment to quality and consistency makes us the preferred partner for global pharmaceutical companies seeking reliable supply chains.
We invite you to leverage our technical expertise to optimize your production costs and timelines. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this novel cyclization route can enhance your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data for our catalog compounds or to discuss route feasibility assessments for custom synthesis projects. By collaborating with us, you gain access to a wealth of chemical knowledge and industrial capacity dedicated to bringing life-saving antiviral therapies to market faster and more efficiently.
