Advanced Manufacturing of Silodosin Intermediates via Optimized Seven-Step Synthesis
Advanced Manufacturing of Silodosin Intermediates via Optimized Seven-Step Synthesis
The pharmaceutical landscape for treating benign prostatic hyperplasia (BPH) relies heavily on the efficient production of high-purity active pharmaceutical ingredients (APIs) and their precursors. A pivotal development in this domain is detailed in patent CN102690223A, which outlines a robust preparation method for 1-acetyl-7-cyano-5-(2-aminopropyl)indoline, a critical intermediate in the synthesis of Silodosin. This specific compound serves as the structural backbone for the final drug molecule, necessitating a synthesis pathway that balances high yield with operational simplicity. The disclosed technology addresses long-standing inefficiencies in traditional manufacturing by introducing a streamlined seven-step sequence that begins with 1-acetylindoline. By leveraging common industrial reagents such as Lewis acids and iron powder, this methodology offers a compelling alternative to legacy processes that often suffer from prohibitive costs and complex purification requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the industry relied on synthetic routes that were fraught with economic and technical bottlenecks. One prominent historical approach, documented in European patent application EP0600675A1, depended heavily on the utilization of tribromopyrrolidone for bromination steps and platinum oxide for hydrogenation reductions. These reagents are not only exorbitantly priced but also introduce significant supply chain volatility due to the reliance on precious metals. Furthermore, the cyanidation step in this legacy route typically exhibited low yields, necessitating resource-intensive column chromatography for purification, which is notoriously difficult to scale for industrial production. Another existing method, described in Chinese patent CN101759627A, attempted to circumvent some issues but introduced new problems regarding selectivity.
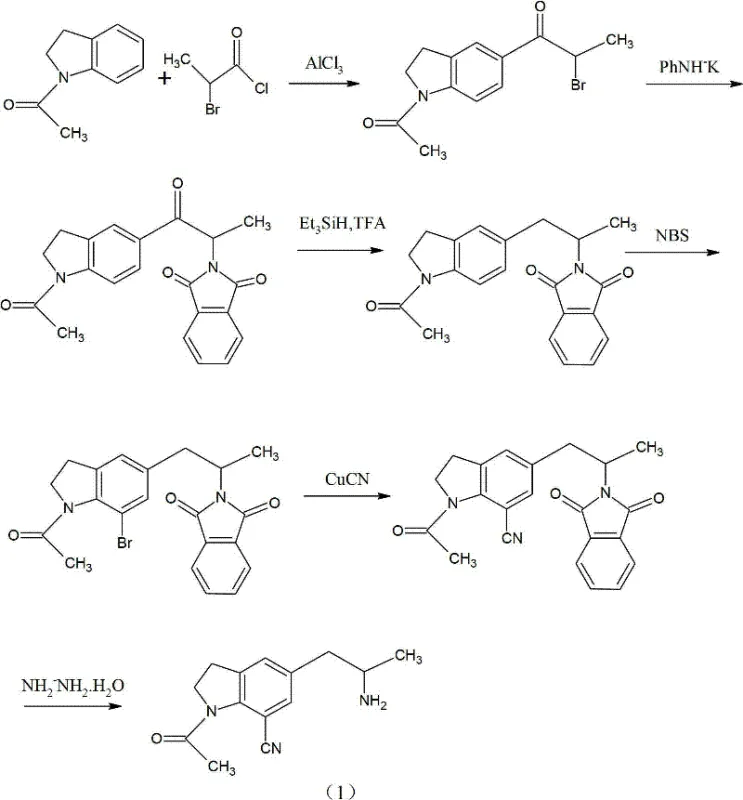
As illustrated in the reaction scheme above, the reliance on N-bromosuccinimide (NBS) for bromination often resulted in poor selectivity, leading to a mixture of isomers that complicated the isolation of the desired intermediate. The lack of specific process conditions in the literature for this route further hindered its industrial viability, leaving manufacturers with a process that was both unpredictable and costly to implement on a commercial scale. These cumulative drawbacks underscored the urgent need for a manufacturing protocol that could deliver high purity without the burden of expensive catalysts or difficult separations.
The Novel Approach
The methodology presented in patent CN102690223A fundamentally reengineers the synthetic pathway to overcome these historical barriers. Instead of relying on precious metal hydrogenation or problematic bromination, the new route employs a strategic sequence of Friedel-Crafts acylation, phthalimide protection, and silane-mediated reduction. This approach ensures that the carbon skeleton is constructed with high fidelity before introducing the critical cyano group via a diazonium salt intermediate. The shift from catalytic hydrogenation to chemical reduction using triethylsilane and trifluoroacetic acid represents a significant operational improvement, allowing for milder reaction conditions that are easier to control in a standard reactor setup. Moreover, the final deprotection step utilizes hydrazine hydrate, a reagent that facilitates clean removal of the phthalimide protecting group through simple filtration and crystallization.
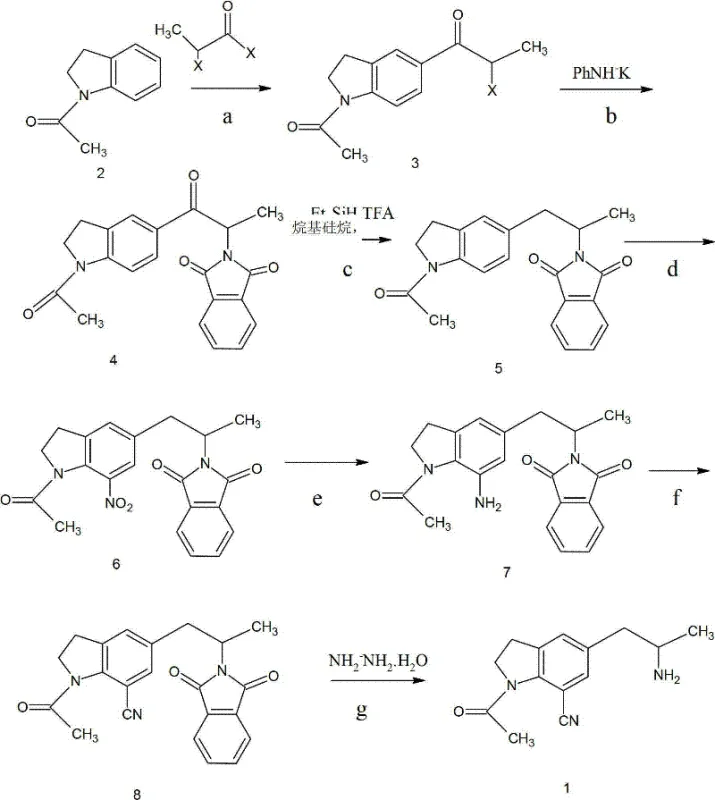
This comprehensive seven-step process, visually summarized in the diagram above, demonstrates a logical progression where each transformation is optimized for yield and ease of workup. By avoiding the formation of difficult-to-separate byproducts early in the synthesis, the overall throughput of the plant is significantly enhanced. The ability to use standard solvents like dichloroethane and ethanol, rather than specialized or hazardous media, further lowers the barrier to entry for contract manufacturing organizations looking to adopt this technology for reliable pharmaceutical intermediate supply.
Mechanistic Insights into the Seven-Step Synthetic Sequence
The core of this technological advancement lies in the precise control of regioselectivity and functional group tolerance throughout the seven distinct stages. The process initiates with a Lewis acid-catalyzed Friedel-Crafts acylation, where 1-acetylindoline reacts with 2-halogenopropionyl halide. The choice of anhydrous zinc chloride or aluminum trichloride as the catalyst is critical, as it activates the acyl halide for electrophilic attack on the indoline ring without causing degradation of the sensitive nitrogen heterocycle. Following this, the introduction of the amine functionality is cleverly managed through a phthalimide substitution. This protects the amine during subsequent harsh conditions, preventing unwanted side reactions that could compromise the integrity of the molecule. The reduction of the carbonyl group to a methylene bridge using triethylsilane and trifluoroacetic acid is a particularly elegant step, utilizing a silane-based mechanism that is highly chemoselective and avoids the safety hazards associated with high-pressure hydrogen gas.
Subsequent functionalization of the aromatic ring is achieved through a classic nitration followed by reduction, setting the stage for the installation of the nitrile group. The conversion of the resulting aniline derivative into the cyano compound via a diazonium salt intermediate is a masterstroke of process chemistry. By generating the diazonium salt in situ at low temperatures (between -5°C and 50°C) and reacting it immediately with a copper cyanide complex, the process minimizes the accumulation of unstable intermediates. This Sandmeyer-type reaction is far superior to direct nucleophilic substitution on a halogenated precursor, which often requires extreme temperatures and pressures. The final step involves the removal of the phthalimide group using hydrazine hydrate, which cleaves the imide bonds to release the free primary amine, yielding the target 1-acetyl-7-cyano-5-(2-aminopropyl)indoline with high purity.
How to Synthesize 1-acetyl-7-cyano-5-(2-aminopropyl)indoline Efficiently
Implementing this synthesis requires strict adherence to the temperature profiles and molar ratios defined in the patent to ensure optimal yield and safety. The process is designed to be telescoped where possible, minimizing the number of isolation steps and reducing solvent consumption. For R&D teams looking to replicate this success, the key lies in the careful control of the diazotization temperature and the stoichiometry of the Lewis acid in the initial acylation. The detailed standardized synthesis steps, including specific reagent quantities and workup procedures for each of the seven stages, are outlined in the guide below.
- Perform Friedel-Crafts acylation on 1-acetylindoline using 2-halogenopropionyl halide and Lewis acid catalyst.
- Execute nucleophilic substitution with potassium phthalimide followed by triethylsilane reduction.
- Conduct nitration, followed by iron powder reduction, diazotization-cyanation, and final hydrazine deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift away from precious metal catalysts and exotic reagents translates directly into a more resilient and cost-effective supply chain. The elimination of platinum oxide and tribromopyrrolidone removes exposure to the volatile pricing of precious metals and specialized fine chemicals, stabilizing the cost of goods sold (COGS). Furthermore, the reliance on commodity chemicals such as iron powder, sodium nitrite, and common organic solvents ensures that raw material availability is rarely a bottleneck, even during periods of global supply chain disruption. This accessibility allows for more accurate forecasting and long-term contracting with suppliers, securing a steady flow of inputs for continuous manufacturing campaigns.
- Cost Reduction in Manufacturing: The replacement of expensive catalytic hydrogenation with chemical reduction using triethylsilane and the substitution of column chromatography with crystallization drastically reduces operational expenditures. By removing the need for precious metal recovery systems and complex purification columns, the capital expenditure (CAPEX) required for plant setup is significantly lowered, while the operating expenditure (OPEX) benefits from reduced energy consumption and shorter cycle times.
- Enhanced Supply Chain Reliability: The use of widely available starting materials like 1-acetylindoline and 2-chloropropionyl chloride mitigates the risk of single-source dependency. Since the reagents are standard industrial chemicals rather than custom-synthesized intermediates, lead times for raw material procurement are substantially shortened, enabling faster response to market demand fluctuations and ensuring consistent production schedules for downstream API synthesis.
- Scalability and Environmental Compliance: The process generates fewer hazardous waste streams compared to prior art methods that utilize heavy metal catalysts and brominating agents. The aqueous workups and simple extractions facilitate easier waste treatment and compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces disposal costs but also enhances the sustainability credentials of the final pharmaceutical product, aligning with the corporate social responsibility goals of major healthcare companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations and process robustness.
Q: What are the primary cost drivers eliminated in this new synthesis route?
A: The process eliminates the need for expensive platinum oxide catalysts and tribromopyrrolidone reagents found in prior art, replacing them with cost-effective Lewis acids and iron powder.
Q: How does this method improve purification compared to previous techniques?
A: Unlike prior routes requiring complex column chromatography, this method utilizes crystallization and standard extraction techniques, significantly simplifying downstream processing.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of readily available raw materials, mild reaction conditions, and high yields at each step makes it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-acetyl-7-cyano-5-(2-aminopropyl)indoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory scale to commercial production requires a partner with deep technical expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate seven-step synthesis of Silodosin intermediates is executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for pharmaceutical applications, providing our clients with peace of mind regarding regulatory compliance and product consistency.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your API manufacturing operations.
