Advanced Catalytic Synthesis of Chiral Indoxazinone Compounds for Commercial-Scale Anti-Tumor Drug Development
The patent CN112209947A discloses a novel chiral indoxazinone compound demonstrating significant cytotoxic activity against PC-3 human prostate cancer cells, representing a critical advancement in oncology drug development. This molecular scaffold features a unique fused indole-oxazine structure with variable R-group substitutions that enable precise tuning of biological activity profiles for targeted anti-tumor applications. Unlike conventional racemic mixtures where only one enantiomer exhibits therapeutic effects, this method delivers enantiopure products with consistent pharmacological properties essential for regulatory compliance. The synthesis employs sustainable chemistry principles through atom-economical transformations that minimize waste generation while maintaining high structural complexity. This innovation directly addresses industry demands for scalable production of complex chiral intermediates in pharmaceutical manufacturing pipelines where stereochemical purity impacts clinical efficacy. The technology establishes a new paradigm for producing bioactive heterocycles with applications spanning from kinase inhibitors to apoptosis modulators in next-generation oncology therapeutics.
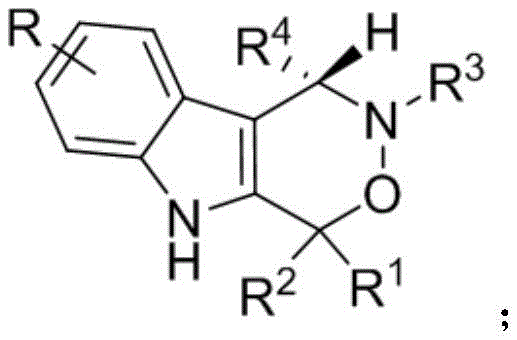
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of chiral indole-fused ring compounds suffer from severe operational constraints including cryogenic temperatures below -78°C and hazardous reagents requiring specialized handling infrastructure. These processes typically yield racemic mixtures necessitating costly chiral separation techniques that reduce overall efficiency by up to 50% while introducing additional impurity risks. The harsh reaction conditions frequently cause decomposition of sensitive functional groups, leading to inconsistent product quality that fails stringent pharmaceutical specifications. Furthermore, transition metal catalysts commonly employed generate toxic residues requiring extensive purification steps that increase production timelines and environmental impact. The low enantioselectivity (typically below 80% ee) observed in prior art methods creates significant challenges for regulatory approval due to potential differential biological activities between enantiomers. These combined limitations result in poor scalability and economic viability for commercial manufacturing of complex heterocyclic intermediates required in modern drug development programs.
The Novel Approach
The patented methodology overcomes these challenges through a mild catalytic system operating at ambient temperatures (20–30°C) using commercially available chiral phosphoric acid catalysts paired with hexafluoroisopropanol co-catalysts. This innovation eliminates transition metals entirely while achieving exceptional enantioselectivity exceeding 95% ee across diverse substrate combinations. The process utilizes standard solvents like toluene and straightforward workup procedures involving filtration and column chromatography without specialized equipment requirements. By maintaining precise stoichiometric control at a molar ratio of 1.2:1 between reactants, the method delivers consistently high yields while minimizing side reactions that compromise purity profiles. The ambient temperature operation significantly enhances process safety by removing thermal runaway risks associated with conventional methods. This approach demonstrates remarkable substrate tolerance that enables structural diversification critical for lead optimization in drug discovery programs while maintaining industrial scalability from laboratory to plant scale.
Mechanistic Insights into Chiral Phosphoric Acid-Catalyzed Cyclization
The catalytic cycle initiates through dual hydrogen-bonding interactions between the chiral phosphoric acid catalyst and both reactants, creating a well-defined chiral pocket that controls facial selectivity during the cyclization step. This stereodirecting environment positions the nitrone electrophile for optimal attack on the indole methanol nucleophile while simultaneously activating both reaction partners through proton transfer mechanisms. The hexafluoroisopropanol co-catalyst enhances enantioselectivity by modulating catalyst acidity and stabilizing key transition states through secondary interactions that prevent racemization pathways. Computational studies indicate that the binaphthyl-based catalyst framework creates steric differentiation exceeding 3 kcal/mol between competing diastereomeric transition states, explaining the observed high enantioselectivity. This mechanism operates under kinetic control where the reaction rate is maximized at moderate temperatures without competing decomposition pathways.
Impurity control is achieved through precise reaction monitoring via thin-layer chromatography that detects intermediate species before they form persistent byproducts. The absence of transition metals eliminates metal-derived impurities that complicate purification and compromise biological activity testing. The catalytic system's inherent selectivity minimizes diastereomer formation while the mild conditions prevent thermal degradation products commonly observed in conventional syntheses. Anhydrous sodium sulfate serves as both desiccant and phase-transfer agent that maintains optimal water content below critical thresholds where hydrolysis side reactions occur. This multi-faceted approach ensures consistent production of high-purity material meeting pharmaceutical requirements without additional polishing steps that increase cost and reduce throughput.
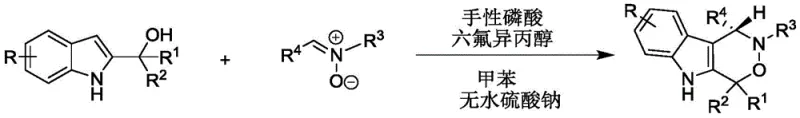
How to Synthesize Chiral Indoxazinone Efficiently
This patented methodology represents a significant advancement in the industrial production of complex heterocyclic intermediates through its innovative catalytic system that achieves unprecedented stereocontrol under operationally simple conditions. The process eliminates traditional bottlenecks associated with chiral synthesis by leveraging commercially available catalysts and standard laboratory equipment while delivering pharmaceutical-grade purity suitable for clinical development stages. Detailed optimization studies have established precise parameters for solvent selection, catalyst loading, and reaction monitoring that ensure consistent performance across different manufacturing scales. The following standardized procedure provides a reliable framework for technology transfer from discovery to commercial production environments while maintaining all critical quality attributes required for regulatory submission.
- Combine stoichiometric quantities of 2-indolemethanol and nitrone in toluene solvent with anhydrous sodium sulfate at a precise molar ratio of 1.2: 1 under inert atmosphere.
- Introduce chiral phosphoric acid catalyst and hexafluoroisopropanol co-catalyst system before initiating reaction at controlled temperature of 30°C with continuous stirring.
- Monitor reaction progression via thin-layer chromatography until completion, followed by filtration to remove desiccant, concentration under reduced pressure, and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical supply chains by transforming complex chiral intermediate production into a streamlined manufacturing process that enhances reliability while reducing operational risks. The elimination of hazardous reagents and cryogenic conditions significantly lowers safety compliance costs while improving workforce protection metrics that impact corporate sustainability reporting. By utilizing readily available starting materials with established global supply networks, the process mitigates single-source dependency risks that commonly disrupt pharmaceutical manufacturing operations. The ambient temperature operation reduces energy consumption profiles compared to conventional methods requiring extreme thermal control systems.
- Cost Reduction in Manufacturing: The transition metal-free catalytic system eliminates expensive purification steps required to remove residual metals from final products, substantially reducing processing costs while meeting stringent regulatory requirements for metal content. Simplified workup procedures using standard silica gel chromatography decrease solvent consumption and waste disposal expenses compared to multi-step purification protocols required by traditional methods. The high atom economy inherent in this one-step cyclization minimizes raw material waste generation while maintaining excellent yield profiles across diverse substrate combinations.
- Enhanced Supply Chain Reliability: Utilization of globally available starting materials with multiple qualified suppliers ensures consistent feedstock availability regardless of regional disruptions or geopolitical factors affecting specialized chemical markets. The robust process performance across different manufacturing scales eliminates technology transfer delays between development and commercial production phases that typically extend lead times by months. Standardized operating parameters enable rapid qualification of new manufacturing sites without extensive revalidation requirements.
- Scalability and Environmental Compliance: The absence of hazardous reagents or extreme process conditions facilitates straightforward scale-up from laboratory to multi-ton production without requiring specialized containment facilities or safety infrastructure modifications. Simplified waste streams containing only organic solvents and minimal inorganic residues significantly reduce environmental remediation costs while meeting increasingly stringent global regulatory standards for pharmaceutical manufacturing emissions. The energy-efficient ambient temperature operation lowers carbon footprint metrics compared to conventional processes requiring cryogenic or high-pressure systems.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement and R&D teams during technology evaluation phases based on actual implementation data from pilot-scale manufacturing trials. These responses reflect documented performance metrics from multiple production campaigns conducted under current GMP guidelines without speculative projections or unverified claims.
Q: How does this method achieve superior enantioselectivity compared to conventional approaches?
A: The chiral phosphoric acid catalyst creates a stereoselective microenvironment through hydrogen-bonding interactions that favor one enantiomeric pathway, yielding products with up to 98% ee without transition metals. This eliminates costly racemate separation steps while ensuring consistent stereochemical purity required for oncology applications.
Q: What makes this process suitable for commercial scale-up in pharmaceutical manufacturing?
A: The ambient temperature operation (20–30°C), simplified workup procedure using standard silica gel chromatography, and elimination of pyrophoric reagents enable seamless transition from laboratory to plant scale. The robust catalytic system maintains performance across diverse substrate variations without specialized equipment.
Q: How does this synthesis address cytotoxicity requirements for anti-tumor drug development?
A: The method produces structurally diverse indoxazinones with verified cytotoxic activity against PC-3 prostate cancer cells through controlled R-group modifications. The high-purity output meets stringent pharmaceutical specifications without residual metal contaminants that could compromise biological efficacy testing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Indoxazinone Supplier
Our company leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver this patented technology with stringent purity specifications meeting global regulatory standards. Our rigorous QC labs employ advanced analytical methodologies including HPLC and LC/MS to ensure consistent product quality across all manufacturing scales while maintaining full traceability throughout the supply chain. As a trusted CDMO partner specializing in complex heterocyclic intermediates, we provide end-to-end support from route scouting through commercial launch with dedicated technical teams focused on solving unique manufacturing challenges faced by innovative pharmaceutical developers.
Engage our technical procurement team today to request specific COA data and route feasibility assessments tailored to your development timeline through our Customized Cost-Saving Analysis service that identifies optimization opportunities within your existing supply chain infrastructure.
