Advanced Synthesis of Lumefantrine-D9: A Cost-Effective Route for Antimalarial Drug Development
In the rapidly evolving landscape of antimalarial drug development, the demand for deuterated analogues has surged due to their potential for improved metabolic stability and pharmacokinetic profiles. Patent CN111320547B introduces a groundbreaking synthesis method for Lumefantrine-D9, a critical deuterated intermediate for next-generation antimalarial therapies. This technical disclosure addresses a significant bottleneck in the supply chain of deuterated pharmaceuticals by proposing a novel route that circumvents the need for scarce and expensive deuterated diamines. By strategically shifting the introduction of the deuterium label to the final synthetic step, this methodology offers a robust pathway for producing high-purity intermediates essential for clinical research and eventual commercialization. For R&D directors and procurement specialists alike, understanding this shift from early-stage to late-stage deuteration is pivotal for optimizing both budget allocation and project timelines in complex drug synthesis programs.
The traditional approach to synthesizing deuterated lumefantrine analogues often relies on the condensation of a fluorene derivative with di-n-butylamine-D9. However, di-n-butylamine-D9 is not a commercially available commodity; it typically requires custom synthesis, which drives up costs exponentially and introduces significant lead time risks. Furthermore, incorporating such a valuable deuterated building block early in a multi-step synthesis poses a substantial financial risk, as any yield losses in subsequent steps result in the irreversible loss of expensive isotopic material. This conventional limitation has historically hindered the widespread adoption of deuterated lumefantrine in large-scale pharmacological studies, creating a barrier for pharmaceutical companies aiming to explore the full therapeutic potential of this antimalarial agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional synthetic routes for deuterated amines often necessitate the preparation of fully deuterated amine precursors, such as di-n-butylamine-D9, prior to the construction of the core molecular scaffold. This strategy is inherently inefficient because the deuterated amine must undergo multiple chemical transformations, each carrying a risk of yield erosion and isotopic dilution. Moreover, the synthesis of di-n-butylamine-D9 itself is non-trivial, often requiring specialized reagents and rigorous purification protocols to ensure high isotopic enrichment. From a supply chain perspective, relying on a single-source or custom-synthesized precursor creates a fragile dependency that can disrupt production schedules. For procurement managers, the lack of commercial availability translates to unpredictable pricing and extended delivery windows, making budget forecasting for clinical trial materials exceptionally difficult.
The Novel Approach
The innovative method disclosed in patent CN111320547B fundamentally restructures the synthetic logic by employing a "late-stage deuteration" strategy. Instead of starting with a deuterated amine, the process builds the entire lumefantrine scaffold using standard, non-deuterated n-butylamine, which is inexpensive and readily available in bulk quantities. The deuterium label is introduced only in the final step via an alkylation reaction with deuterated n-bromobutane-D9. This approach offers a decisive advantage: the expensive deuterated reagent is used stoichiometrically in the last transformation, minimizing waste and maximizing the economic efficiency of the isotope. Additionally, deuterated alkyl halides like n-bromobutane-D9 are more accessible commercially than their amine counterparts, thereby stabilizing the supply chain and reducing overall raw material costs significantly.
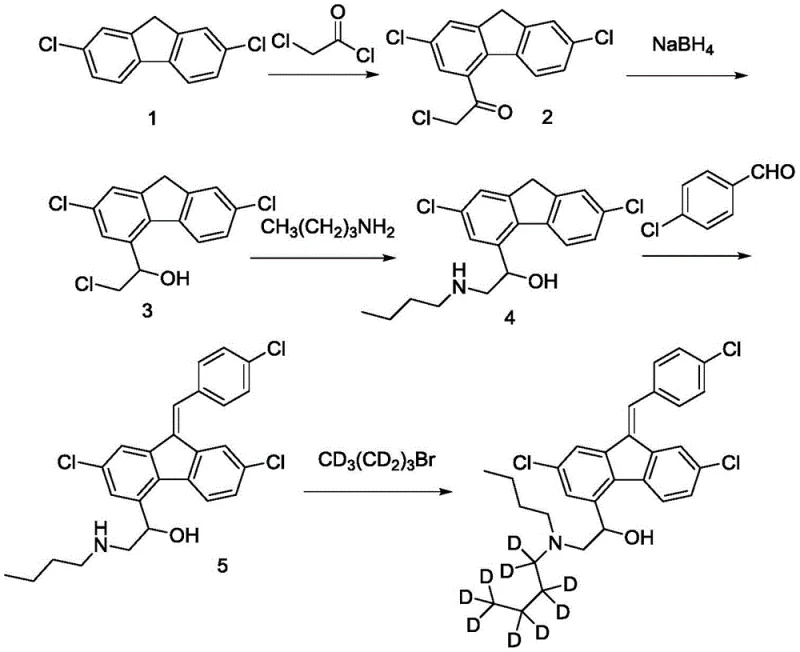
Mechanistic Insights into the Five-Step Synthetic Sequence
The synthesis begins with a classic Friedel-Crafts acylation, where 2,7-dichlorofluorene reacts with chloroacetyl chloride in the presence of anhydrous aluminum chloride. This electrophilic aromatic substitution selectively installs a chloroacetyl group onto the fluorene ring system, generating a key ketone intermediate. Following this, a chemoselective reduction using sodium borohydride converts the ketone into a secondary alcohol without affecting the aryl chlorides or the alkyl chloride side chain. The subsequent step involves a nucleophilic substitution where n-butylamine displaces the terminal chloride on the side chain, forming the secondary amine linkage. This sequence effectively constructs the core amine functionality of lumefantrine using standard, robust chemistry that is easily scalable in industrial reactors.
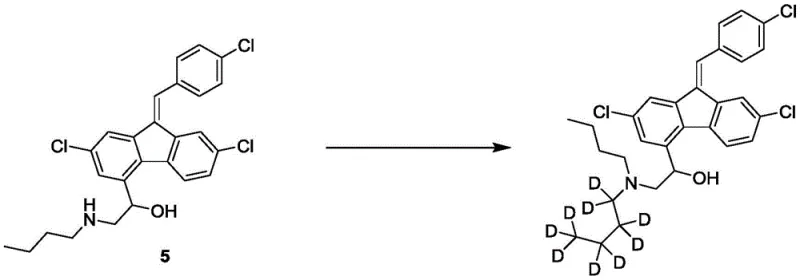
The final two steps are critical for establishing the pharmacophore and the isotopic label. First, the amine intermediate undergoes a condensation reaction with p-chlorobenzaldehyde under basic conditions to form the characteristic stilbene double bond, completing the lumefantrine carbon skeleton. The culmination of the process is the N-alkylation of the secondary amine with deuterated n-bromobutane-D9 in the presence of potassium carbonate. This SN2 reaction cleanly replaces the remaining proton on the nitrogen with the deuterated butyl chain. By reserving this specific transformation for the end of the sequence, the process ensures that the costly deuterated moiety is not subjected to harsh acidic or oxidative conditions found in earlier steps, thereby preserving isotopic integrity and ensuring high radiochemical purity suitable for ADME studies.
How to Synthesize Lumefantrine-D9 Efficiently
The detailed protocol for synthesizing Lumefantrine-D9 involves precise control of reaction parameters across five distinct stages, ranging from acylation to final alkylation. Each step has been optimized in the patent to maximize yield and purity, utilizing common solvents like dichloromethane, ethanol, and acetonitrile to facilitate easy workup and solvent recovery. The following guide outlines the standardized operational procedure derived from the patent examples, providing a clear roadmap for laboratory and pilot-scale execution.
- Perform Friedel-Crafts acylation on 2,7-dichlorofluorene with chloroacetyl chloride using AlCl3 to form the chloro-ketone intermediate.
- Reduce the ketone intermediate to the corresponding alcohol using sodium borohydride in ethanol.
- React the chloro-alcohol with n-butylamine at elevated temperatures to substitute the chloride and form the secondary amine.
- Condense the amine intermediate with p-chlorobenzaldehyde under basic conditions to establish the stilbene double bond.
- Execute the final alkylation using deuterated n-bromobutane-D9 and potassium carbonate to introduce the deuterated butyl chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route presents a compelling value proposition centered on cost stability and logistical reliability. By decoupling the synthesis of the complex molecular scaffold from the sourcing of the deuterated label, manufacturers can leverage established supply chains for the bulk of the raw materials. This separation of concerns means that fluctuations in the market for specialized deuterated chemicals have a minimized impact on the overall production cost, as the deuterated reagent constitutes a smaller fraction of the total mass balance compared to a fully deuterated amine approach. Furthermore, the use of commodity chemicals for the initial four steps allows for bulk purchasing power, driving down the baseline cost of goods sold.
- Cost Reduction in Manufacturing: The primary economic driver of this process is the elimination of the need for custom-synthesized di-n-butylamine-D9. By substituting this rare reagent with commercially available deuterated n-bromobutane-D9, the route drastically simplifies the sourcing strategy. The late-stage introduction of the isotope ensures that the expensive deuterated material is not lost to inefficiencies in earlier synthetic steps, effectively improving the atom economy of the label. This strategic material usage translates directly into lower variable costs per kilogram of the final API intermediate, making the project more financially viable for long-term development.
- Enhanced Supply Chain Reliability: Reliance on custom-synthesized precursors often introduces single points of failure in the supply chain. In contrast, deuterated alkyl halides are standard catalog items available from multiple global suppliers, ensuring continuity of supply even if one vendor faces production issues. The robustness of the chemical steps, which avoid exotic catalysts or extreme conditions, further reduces the risk of batch failures. This reliability is crucial for maintaining the strict timelines required for clinical trial material production, where delays can have cascading effects on regulatory submissions and market entry strategies.
- Scalability and Environmental Compliance: The synthetic route employs well-understood unit operations such as filtration, extraction, and distillation, which are easily transferred from the laboratory to multi-ton production facilities. The solvents used, including dichloromethane and acetonitrile, have established recovery and recycling protocols, allowing manufacturers to implement closed-loop systems that minimize waste generation. This alignment with green chemistry principles not only reduces disposal costs but also ensures compliance with increasingly stringent environmental regulations, safeguarding the manufacturing license and corporate reputation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Lumefantrine-D9. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and advantages of this specific synthetic pathway for stakeholders evaluating potential manufacturing partners.
Q: Why is this synthesis route for Lumefantrine-D9 considered more cost-effective?
A: This route avoids the use of di-n-butylamine-D9, which is not commercially available and requires complex custom synthesis. Instead, it utilizes readily available deuterated n-bromobutane-D9 in the final step, significantly reducing raw material costs and supply chain complexity.
Q: What is the key chemical transformation in the final step?
A: The final step involves a nucleophilic substitution reaction where the secondary amine nitrogen of the lumefantrine precursor attacks the deuterated n-bromobutane-D9. This late-stage functionalization ensures high isotopic purity and minimizes the loss of expensive deuterated materials during earlier synthetic stages.
Q: Can this process be scaled for commercial production?
A: Yes, the process utilizes standard organic transformations such as Friedel-Crafts acylation, borohydride reduction, and SN2 alkylation, all of which are well-established and scalable in industrial reactors. The use of common solvents like dichloromethane, ethanol, and acetonitrile further facilitates easy scale-up and solvent recovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lumefantrine-D9 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality deuterated intermediates in advancing antimalarial drug candidates through the clinical pipeline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee the isotopic enrichment and chemical purity of every batch of Lumefantrine-D9 we produce.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By leveraging our expertise in process chemistry, we can provide a Customized Cost-Saving Analysis tailored to your volume needs. Please contact us today to request specific COA data and route feasibility assessments, and let us support your journey toward delivering next-generation antimalarial therapies to the global market.
