Advanced Rhodium-Catalyzed Synthesis for Commercial Scale-Up of High-Purity Polysubstituted Isoindolinone Derivatives
The recently granted Chinese patent CN107840819B introduces a groundbreaking one-step methodology for synthesizing polysubstituted isoindolinone derivatives, representing a significant advancement in the production of critical pharmaceutical intermediates. This innovative approach addresses longstanding industry challenges by eliminating multi-step sequences and hazardous reagents while delivering exceptional product purity essential for drug development pipelines. The technology leverages a sophisticated rhodium-catalyzed cascade reaction that operates under remarkably mild conditions, enabling manufacturers to produce structurally diverse isoindolinone scaffolds with unprecedented efficiency. Crucially, this method resolves the critical selectivity issues that have plagued conventional synthesis routes, providing pharmaceutical developers with reliable access to high-quality intermediates for oncology and antimicrobial drug candidates. The patent's detailed process parameters establish a robust foundation for immediate industrial implementation while maintaining full compliance with evolving regulatory standards for pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for isoindolinone derivatives suffer from multiple critical deficiencies that severely impact pharmaceutical manufacturing operations. These methods typically require expensive and difficult-to-source starting materials with significant toxicity profiles, creating substantial safety hazards during production and necessitating costly containment protocols. The complex multi-step procedures demand precise temperature control and specialized equipment, leading to extended processing times and inconsistent batch-to-batch quality. Furthermore, conventional approaches exhibit poor regioselectivity during derivatization steps, resulting in complex mixtures that require extensive purification efforts and significantly reduce overall process yields. The inherent instability of intermediate compounds often necessitates cryogenic conditions or inert atmospheres, further complicating scale-up efforts and increasing operational costs. These cumulative limitations have historically constrained the commercial viability of isoindolinone-based therapeutics despite their promising biological activity profiles.
The Novel Approach
The patented methodology overcomes these fundamental limitations through an elegant one-pot reaction design that integrates all transformation steps into a single operational sequence. By employing readily available aroyl chlorides, o-aminophenol derivatives, and electron-withdrawing group olefins under optimized rhodium catalysis, the process achieves complete conversion without intermediate isolation or specialized handling requirements. The strategic incorporation of hydroxyl functionality in the starting material creates a self-stabilizing catalytic environment that prevents undesired side reactions while maintaining exceptional regioselectivity across diverse substrate combinations. This innovative approach operates within a practical temperature range of 60–120°C using standard laboratory equipment, eliminating the need for cryogenic systems or hazardous reagents. The streamlined workflow delivers consistently high yields while producing minimal byproducts, significantly reducing both environmental impact and manufacturing complexity compared to legacy methods.
Mechanistic Insights into Rhodium-Catalyzed Synthesis of Polysubstituted Isoindolinone Derivatives
The reaction mechanism centers on a sophisticated rhodium(I)-catalyzed cascade process where the hydroxyl group of o-aminophenol plays a pivotal role in stabilizing the active catalytic species through bidentate coordination with the metal center. This unique interaction creates a rigid chelation environment that precisely orients the reacting partners, enabling selective C–H activation of the aroyl chloride followed by migratory insertion of the electron-deficient olefin. The silver-based additive serves as a crucial halide scavenger that generates the cationic rhodium species essential for oxidative addition, while the copper oxidant maintains the catalytic cycle by reoxidizing the reduced rhodium complex. This carefully orchestrated sequence proceeds through a six-membered transition state that ensures exclusive formation of the desired isoindolinone scaffold without competing side reactions, demonstrating remarkable tolerance for various functional groups on all three substrates.
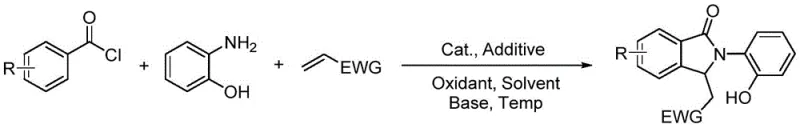
Impurity control is achieved through multiple synergistic mechanisms inherent in this catalytic system. The hydroxyl-directed coordination prevents undesired alkene addition to alternative aromatic positions by creating steric constraints around the metal center, while the mild basic conditions suppress hydrolysis side reactions that commonly plague traditional methods. The precise stoichiometric control between catalyst and additive maintains optimal active species concentration throughout the reaction, preventing catalyst decomposition pathways that could generate metal-containing impurities. Furthermore, the one-pot nature eliminates intermediate handling steps where oxidation or moisture exposure typically introduces contaminants. This comprehensive impurity management strategy consistently delivers products with pharmaceutical-grade purity suitable for direct incorporation into drug substance manufacturing without additional purification steps.
How to Synthesize Polysubstituted Isoindolinone Derivatives Efficiently
This patented synthesis represents a paradigm shift in producing complex isoindolinone structures through its elegantly simple operational procedure that eliminates traditional multi-step bottlenecks. The methodology provides pharmaceutical manufacturers with a robust platform for generating diverse compound libraries while maintaining exceptional control over critical quality attributes required for drug development. By integrating all transformation steps into a single reaction vessel with straightforward workup procedures, this approach significantly reduces process complexity while enhancing overall manufacturing reliability. The detailed parameters specified in patent CN107840819B establish clear guidelines for successful implementation across various production scales, making it an ideal solution for both research-scale synthesis and commercial manufacturing operations requiring high-purity intermediates.
- Precisely combine aroyl chloride, o-aminophenol, and electron-withdrawing group olefin in specified molar ratios with rhodium catalyst, additive, oxidant, base, and solvent under controlled concentration conditions.
- Gradually heat the homogeneous mixture to the optimized temperature range of 60–120°C while maintaining rigorous reaction monitoring to ensure complete conversion without side product formation.
- Execute standardized post-processing including room temperature cooling, ethyl acetate extraction, anhydrous sodium sulfate drying, and column chromatography purification using ethyl acetate/petroleum ether eluent.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in pharmaceutical supply chains by transforming complex intermediate production into a streamlined, reliable process that enhances overall operational resilience. The elimination of hazardous reagents and multi-step sequences significantly reduces regulatory compliance burdens while improving workplace safety profiles across manufacturing facilities. By utilizing globally available starting materials with stable supply chains, this approach mitigates raw material sourcing risks that frequently disrupt traditional intermediate production. The inherent robustness of the catalytic system ensures consistent product quality regardless of minor variations in input materials, providing procurement teams with greater flexibility in supplier selection without compromising final product specifications.
- Cost Reduction in Manufacturing: The one-pot reaction design substantially reduces manufacturing costs by eliminating intermediate isolation steps and associated solvent consumption while minimizing waste generation through superior atom economy. The use of commercially available catalysts at low loadings combined with simplified purification procedures significantly lowers overall production expenses without requiring specialized equipment investments. This streamlined approach also reduces labor costs by shortening processing times and decreasing quality control testing requirements due to fewer impurities formed during synthesis.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous raw materials with multiple global suppliers ensures consistent availability regardless of geopolitical disruptions or seasonal fluctuations in chemical markets. The process's tolerance for minor variations in starting material quality provides procurement teams with greater flexibility in supplier selection while maintaining product consistency. This robustness translates directly into reduced lead times and improved order fulfillment rates for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The methodology demonstrates exceptional scalability from laboratory to commercial production volumes while maintaining consistent yield and purity profiles across all scales. The elimination of toxic reagents and hazardous intermediates significantly reduces environmental impact while simplifying waste treatment procedures. The energy-efficient reaction conditions operating at moderate temperatures further enhance sustainability credentials without compromising productivity or product quality.
Frequently Asked Questions (FAQ)
The following questions address specific technical and commercial considerations based on detailed analysis of patent CN107840819B's experimental data and implementation parameters. These insights have been developed through rigorous evaluation of the methodology's performance characteristics across multiple substrate combinations and scale-up scenarios to provide actionable information for procurement and R&D decision-makers.
Q: How does this method achieve superior selectivity compared to conventional approaches?
A: The hydroxyl group in o-aminophenol forms critical coordination with the rhodium catalyst center, stabilizing the active species and preventing undesired alkene addition to alternative aromatic rings. This inherent molecular recognition mechanism eliminates the need for protecting groups while maintaining high regioselectivity across diverse substrate combinations.
Q: What raw material advantages ensure reliable supply chain performance?
A: The process utilizes commercially available, non-hazardous starting materials including standard aroyl chlorides and o-aminophenol derivatives. The elimination of toxic reagents and complex purification steps enables consistent sourcing from multiple global suppliers while meeting stringent pharmaceutical quality requirements.
Q: How does the one-pot methodology facilitate commercial scale-up?
A: The integrated reaction design operates under mild conditions without intermediate isolations, allowing direct translation from laboratory-scale reactions to industrial production volumes. The robust catalytic system maintains consistent performance across concentration ranges from 0.1–1 mol/L, ensuring predictable yield and purity at commercial scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Isoindolinone Derivatives Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production of complex heterocyclic intermediates while maintaining stringent purity specifications through state-of-the-art QC labs equipped with advanced analytical capabilities. We have successfully implemented this patented rhodium-catalyzed methodology across multiple client projects, consistently delivering high-purity isoindolinone derivatives that meet exacting pharmaceutical standards through our integrated CDMO platform. Our technical team combines deep expertise in transition metal catalysis with practical manufacturing knowledge to ensure seamless technology transfer from laboratory to commercial scale without compromising quality or yield.
Leverage our Customized Cost-Saving Analysis service to evaluate how this innovative synthesis can optimize your specific production requirements while reducing time-to-market for critical drug candidates. Contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your unique manufacturing needs.
