Scalable Synthesis of Gamma-Trifluoromethyl Amino Acids via Catalytic Tandem Reaction
Scalable Synthesis of Gamma-Trifluoromethyl Amino Acids via Catalytic Tandem Reaction
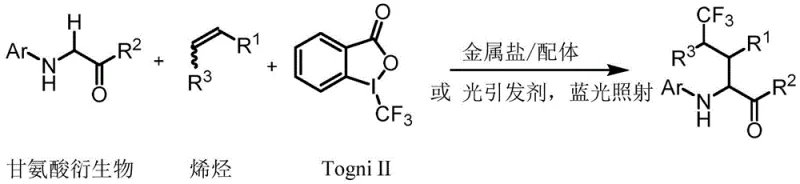
The pharmaceutical and fine chemical industries are constantly seeking efficient routes to fluorinated building blocks, particularly non-natural amino acids that enhance metabolic stability and bioavailability. Patent CN114890908B introduces a groundbreaking methodology for the preparation of gamma-trifluoromethyl amino acid derivatives, addressing critical bottlenecks in traditional fluorination chemistry. This technology utilizes a tandem reaction strategy involving N-aryl glycine derivatives, olefins, and hypervalent iodine trifluoromethyl sources, specifically Togni reagents. By leveraging either transition metal catalysis or visible-light photoredox catalysis, the process achieves direct alpha-C-H alkylation of glycine derivatives without the need for stoichiometric oxidants. This represents a significant leap forward for reliable pharmaceutical intermediate supplier networks, as it simplifies the synthetic workflow while maintaining high chemical selectivity and yield across a diverse range of substrates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorine-containing amino acids has been plagued by inefficient methodologies that hinder cost reduction in API manufacturing. Traditional routes often rely on the assembly of functional groups prior to cross-coupling via transition metal catalysis, which necessitates harsh reaction conditions, frequently exceeding 100°C. Furthermore, existing methods typically require stoichiometric amounts of strong oxidants to drive the reaction, leading to poor atom utilization and the generation of substantial chemical waste. These factors not only increase the environmental footprint but also complicate the purification process, resulting in lower overall yields and limited substrate universality. For supply chain heads, these inefficiencies translate into higher raw material costs and longer lead times for high-purity pharmaceutical intermediates, making the adoption of greener alternatives an economic imperative.
The Novel Approach
The innovative process described in the patent overcomes these hurdles by employing a catalytic tandem reaction that directly converts glycine derivatives into gamma-trifluoromethyl amino acids. As illustrated in the reaction scheme below, the method combines a trifluoromethyl source, an N-aryl glycine derivative, and a non-conjugated olefin in a single pot. The use of Togni reagents allows the reaction to proceed under mild conditions, typically between 0°C and 60°C, or even at room temperature under blue light irradiation. This approach eliminates the need for equivalent oxidants, as the hypervalent iodine reagent serves a dual purpose, thereby streamlining the post-reaction workup. The broad compatibility with various olefins and glycine derivatives ensures that this route is robust enough for the commercial scale-up of complex polymer additives and specialty chemicals.
Mechanistic Insights into Fe/Cu-Catalyzed Radical Alkylation
The mechanistic elegance of this transformation lies in its radical-mediated pathway, which facilitates the formation of carbon-carbon bonds under exceptionally mild conditions. The catalyst, whether a transition metal salt like Cu(I) or Fe(II) or a photoinitiator like Ir(ppy)3, initiates the cycle by reducing the high-valence iodine reagent to generate a trifluoromethyl radical. This reactive species adds selectively to the carbon-carbon double bond of the olefin, creating a new alkyl radical intermediate. Simultaneously, the high-valence catalyst species oxidizes the glycine ester substrate twice to form an electrophilic imine intermediate. The final C-C bond formation occurs through the addition of the alkyl radical to this imine, yielding the desired gamma-trifluoromethyl amino acid derivative. This intricate balance of redox potentials ensures high chemoselectivity, preventing side reactions that often plague radical chemistry.
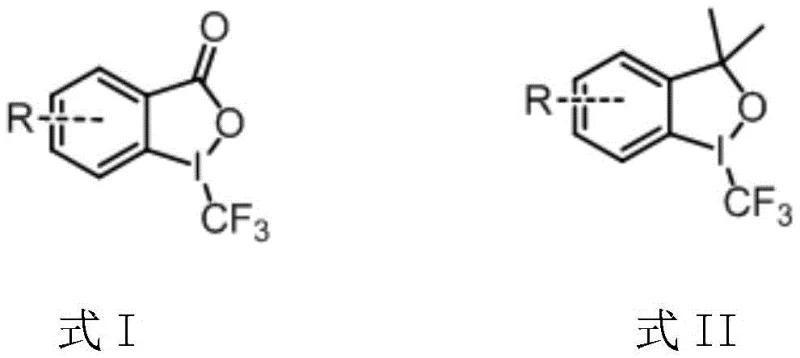
Understanding the role of the trifluoromethyl source is crucial for optimizing impurity profiles and ensuring batch-to-batch consistency. The patent specifies the use of Formula I or Formula II reagents, commonly known as Togni reagents, which are stable solids that release the CF3 group upon reduction. The structure of these reagents, shown below, highlights the hypervalent iodine center that drives the oxidative capacity of the system. By avoiding external oxidants, the process minimizes the formation of over-oxidized byproducts and metal residues, which is a key concern for R&D directors focused on purity specifications. The ability to tune the catalyst system between thermal metal catalysis and photocatalysis offers further flexibility, allowing manufacturers to select the most energy-efficient mode for their specific production infrastructure.
How to Synthesize Gamma-Trifluoromethyl Amino Acid Efficiently
Implementing this synthesis requires careful control of reaction parameters to maximize yield and minimize impurities. The process begins with the selection of appropriate starting materials, ensuring the olefin is non-conjugated to prevent competing polymerization or alternative addition pathways. The reaction is typically conducted in solvents such as dichloroethane, acetonitrile, or toluene under an inert atmosphere to protect the sensitive radical intermediates from oxygen quenching. Detailed standardized synthesis steps see the guide below.
- Combine trifluoromethyl source (Togni reagent), N-aryl glycine derivative, and non-conjugated olefin in an organic solvent such as dichloroethane or acetonitrile.
- Add a catalytic system comprising either a transition metal salt with a ligand (e.g., Cu/Pybox) or a photoinitiator (e.g., Ir(ppy)3) under inert atmosphere.
- Stir the mixture at 0-60°C or under blue light irradiation (420-500 nm) until completion, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this catalytic methodology offers tangible benefits that extend beyond simple yield improvements. The elimination of stoichiometric oxidants significantly reduces the volume of hazardous waste generated per kilogram of product, aligning with increasingly stringent environmental regulations and lowering disposal costs. Moreover, the use of commercially available and stable Togni reagents ensures a consistent supply of the trifluoromethyl source, mitigating risks associated with specialized or unstable reagents. The mild reaction conditions also imply lower energy consumption for heating or cooling, contributing to substantial cost savings in large-scale manufacturing operations. These factors collectively enhance the reliability of the supply chain for high-purity fluorinated intermediates.
- Cost Reduction in Manufacturing: The catalytic nature of the process means that expensive metal salts or photoinitiators are used in minute quantities, drastically reducing the raw material cost per unit. By removing the requirement for stoichiometric oxidants, the process simplifies the bill of materials and reduces the complexity of the downstream purification train. This efficiency translates directly into a more competitive pricing structure for the final amino acid derivatives, allowing partners to optimize their margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: The robustness of the reaction across a wide substrate scope means that a single production line can be adapted to manufacture various derivatives with minimal changeover time. The starting materials, including simple olefins and glycine esters, are commodity chemicals with established global supply chains, reducing the risk of shortages. This flexibility ensures continuous production capability, which is critical for meeting the demanding delivery schedules of multinational pharmaceutical clients.
- Scalability and Environmental Compliance: Operating at near-ambient temperatures and pressures makes this process inherently safer and easier to scale from laboratory to pilot and commercial plants. The reduced generation of toxic byproducts simplifies wastewater treatment and aligns with green chemistry principles, facilitating regulatory approval in key markets. This environmental compliance is a strategic asset, future-proofing the manufacturing process against tightening global sustainability mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing portfolios.
Q: What are the advantages of using Togni reagents in this synthesis?
A: Togni reagents serve as both the trifluoromethyl source and the oxidant, eliminating the need for stoichiometric external oxidants and improving atom economy.
Q: Can this method be scaled for industrial production?
A: Yes, the reaction operates under mild conditions (room temperature to 60°C) without harsh reagents, making it highly suitable for commercial scale-up.
Q: What types of olefins are compatible with this protocol?
A: The protocol accepts a wide range of non-conjugated olefins, including terminal alkenes, cyclic alkenes, and functionalized alkenes, providing broad substrate scope.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gamma-Trifluoromethyl Amino Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic technology in the production of advanced pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art reactors capable of handling both thermal and photochemical processes, supported by stringent purity specifications and rigorous QC labs that guarantee the highest quality standards for every batch delivered.
We invite you to collaborate with us to leverage this efficient synthesis route for your next-generation drug candidates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with our reliable supply of high-performance fluorinated building blocks.
