Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Manufacturing
Advanced Palladium-Catalyzed Carbonylation for Scalable 2,3-Dihydroquinolone Manufacturing
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and safer synthetic routes to bioactive scaffolds. A pivotal development in this domain is documented in patent CN112239456B, which discloses a novel preparation method for substituted 2,3-dihydroquinolone compounds. This technology represents a significant leap forward for the fine chemical industry, particularly for those focused on the production of complex nitrogen-containing heterocycles. The 2,3-dihydroquinolone skeleton is not merely an academic curiosity; it is a privileged structure found in numerous molecules exhibiting potent biological activities, including significant anti-cancer and analgesic properties. As illustrated in the background art, compounds such as antitumor agent A, antitumor agent B, and analgesic agent C share this core structural motif, underscoring the immense pharmaceutical value of mastering its synthesis.
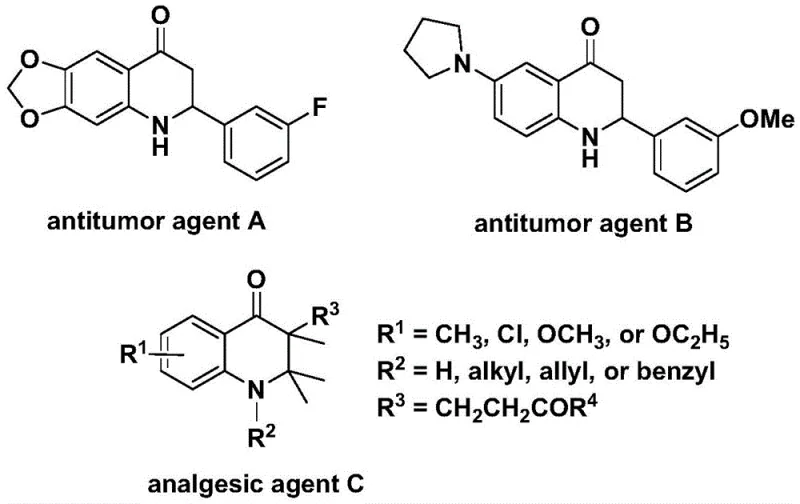
For research and development directors overseeing process chemistry, the ability to access these scaffolds reliably is paramount. The method described in CN112239456B utilizes a transition metal palladium-catalyzed carbonylation reaction, employing N-pyridine sulfonyl-o-iodoaniline and olefins as the primary starting materials. This approach circumvents many of the historical bottlenecks associated with heterocycle formation, offering a pathway that is both operationally simple and chemically robust. By leveraging this patented methodology, manufacturers can secure a competitive edge in the supply of high-purity pharmaceutical intermediates, ensuring that downstream drug development pipelines remain uninterrupted by raw material shortages or quality inconsistencies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of carbonyl-containing heterocycles like 2,3-dihydroquinolones has been fraught with challenges that hinder efficient commercial scale-up. Traditional carbonylation reactions often rely on the direct use of carbon monoxide gas, which poses severe safety hazards due to its high toxicity and flammability. Handling gaseous CO requires specialized high-pressure equipment and rigorous safety protocols, which drastically increases capital expenditure and operational complexity for chemical plants. Furthermore, conventional methods frequently suffer from limited substrate scope, where the presence of sensitive functional groups on the aromatic ring or the olefin partner can lead to catalyst poisoning or side reactions. This lack of functional group tolerance necessitates additional protection and de-protection steps, elongating the synthetic sequence and reducing overall atom economy. Consequently, the cost of goods sold (COGS) for intermediates produced via these legacy routes remains prohibitively high, creating friction in the supply chain for cost-sensitive generic drug manufacturers.
The Novel Approach
In stark contrast, the innovative process detailed in the patent introduces a paradigm shift by utilizing a solid carbon monoxide substitute, specifically 1,3,5-trimesic acid phenol ester (TFBen), instead of gaseous CO. This strategic substitution fundamentally alters the safety profile of the reaction, allowing it to be conducted in standard laboratory glassware or reactors without the need for high-pressure autoclaves designed for toxic gases. The reaction proceeds smoothly in the presence of a palladium catalyst, specifically bis(acetylacetone)palladium, paired with a bidentate phosphine ligand like 1,3-bis(diphenylphosphino)propane (dppp). This catalytic system demonstrates exceptional versatility, capable of synthesizing both 2-aryl and 3-alkyl substituted 2,3-dihydroquinolone compounds simply by varying the olefin substrate. The general reaction scheme highlights the elegance of this transformation, where the iodine atom on the aniline derivative serves as the handle for palladium insertion, initiating a cascade that seamlessly constructs the six-membered lactam ring.
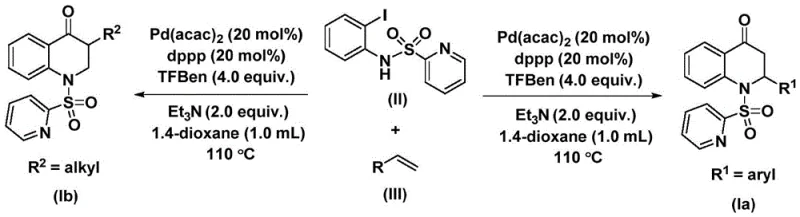
Moreover, the reaction conditions are remarkably mild yet effective, typically operating at temperatures between 100-120 °C in aprotic solvents like 1,4-dioxane. The use of triethylamine as an additive further facilitates the reaction progress by neutralizing acidic byproducts. For procurement managers, this translates to a process that relies on readily available, off-the-shelf reagents rather than bespoke or hazardous specialty gases. The simplicity of the post-treatment procedure, which involves basic filtration and standard column chromatography, ensures that the final product can be isolated with high purity and minimal waste generation. This streamlined workflow not only reduces the time-to-market for new intermediates but also aligns perfectly with modern green chemistry principles, making it an attractive option for environmentally conscious supply chain strategies.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
To fully appreciate the robustness of this synthetic method, one must delve into the mechanistic underpinnings of the palladium-catalyzed cycle. The reaction initiates with the oxidative addition of the palladium(0) species into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline substrate. This critical step generates an aryl-palladium(II) intermediate, which serves as the foundation for the subsequent carbonylation event. Unlike traditional methods where CO gas must diffuse into the solution, the solid CO surrogate (TFBen) decomposes under the reaction conditions to release carbon monoxide in situ. This generated CO then inserts into the aryl-palladium bond to form an acyl-palladium intermediate. This step is crucial as it installs the carbonyl functionality directly onto the growing molecular framework with high regioselectivity.
Following the CO insertion, the olefin substrate coordinates to the palladium center and undergoes migratory insertion into the acyl-palladium bond. This forms a new carbon-carbon bond and generates a palladium-alkyl intermediate. The final step of the catalytic cycle involves an intramolecular nucleophilic attack or reductive elimination, depending on the specific electronic nature of the substrates, which closes the ring to form the 2,3-dihydroquinolone core and regenerates the active palladium(0) catalyst. This mechanism explains the high efficiency and broad substrate compatibility observed in the experimental data. The choice of the dppp ligand is particularly astute, as its bite angle and electronic properties stabilize the palladium intermediates throughout the cycle, preventing premature catalyst decomposition. For R&D teams, understanding this mechanism allows for rational optimization of the process, such as tuning the ligand or solvent to accommodate even more sterically hindered substrates, thereby expanding the chemical space accessible through this reliable pharmaceutical intermediate supplier platform.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis in a practical setting requires adherence to specific protocol parameters to ensure maximum yield and reproducibility. The patent outlines a straightforward procedure where the catalyst, ligand, CO source, and substrates are combined in an organic solvent. The reaction is heated to promote the catalytic turnover, followed by a workup that leverages standard purification techniques familiar to any process chemist. The versatility of the method is evidenced by the successful synthesis of various derivatives, including those with methyl, chloro, and alkyl substituents, all achieving respectable yields ranging from 60% to 88%. Below is the structured guide for executing this transformation effectively.
- Charge a reaction vessel with Pd(acac)2 (20 mol%), dppp (20 mol%), TFBen (4.0 equiv.), N-pyridine sulfonyl-o-iodoaniline, olefin, and Et3N in 1,4-dioxane.
- Heat the reaction mixture to 110 °C and stir continuously for a duration of 24 to 48 hours under inert atmosphere.
- Upon completion, filter the mixture, adsorb onto silica gel, and purify via column chromatography to isolate the target 2,3-dihydroquinolone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for procurement and supply chain stakeholders. The shift away from gaseous carbon monoxide to a solid surrogate eliminates a major logistical and safety bottleneck. Transporting and storing toxic gases requires specialized infrastructure and regulatory compliance that can delay project timelines. By contrast, solid reagents like TFBen are stable, easy to ship, and can be stocked in standard warehouses, significantly enhancing supply chain reliability. Furthermore, the use of common solvents like dioxane and commercially available palladium catalysts ensures that the raw material supply is robust and less susceptible to market volatility compared to exotic reagents. This stability is crucial for maintaining continuous production schedules in a high-demand pharmaceutical environment.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the operational setup. Eliminating the need for high-pressure reactors designed for toxic gases reduces capital investment and maintenance costs. Additionally, the high reaction efficiency and broad substrate tolerance mean fewer batches are rejected due to impurities or low conversion, leading to substantial cost savings in raw material consumption. The simplified post-treatment process, which avoids complex extraction or distillation steps, further lowers energy consumption and labor costs, contributing to a leaner manufacturing model that enhances overall profitability.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials, such as o-iodoaniline derivatives and simple olefins, ensures a steady flow of inputs. These precursors are produced by multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in temperature or reagent quality, resulting in consistent batch-to-bystock quality. This predictability allows supply chain managers to forecast inventory needs more accurately and reduce safety stock levels, freeing up working capital while ensuring that downstream API manufacturing lines never face interruptions due to intermediate shortages.
- Scalability and Environmental Compliance: Scaling this reaction from gram to kilogram or ton scale is facilitated by the homogeneous nature of the catalytic system and the absence of hazardous gas handling. The reaction can be performed in standard stirred-tank reactors, making the transition from pilot plant to full commercial production seamless. Moreover, the use of a solid CO source minimizes the release of volatile organic compounds and toxic emissions, aligning the process with increasingly stringent environmental regulations. This eco-friendly profile not only reduces waste disposal costs but also enhances the corporate sustainability credentials of the manufacturer, a factor that is becoming increasingly important for multinational clients evaluating their supplier base.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring that potential partners have a clear understanding of the process capabilities and limitations. Understanding these details is essential for making informed decisions about integrating this technology into existing production portfolios.
Q: What is the primary advantage of using TFBen in this synthesis?
A: TFBen (1,3,5-trimethoxybenzene phenol ester) serves as a solid carbon monoxide substitute, eliminating the need for handling toxic and hazardous gaseous CO, thereby significantly enhancing operational safety and simplifying reactor requirements.
Q: What types of substrates are compatible with this palladium catalytic system?
A: The method exhibits excellent substrate compatibility, successfully accommodating both aryl olefins (yielding 2-aryl substituted products) and alkyl or silyl olefins (yielding 3-alkyl substituted products) with high functional group tolerance.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is scalable to gram levels and potentially industrial scales due to the use of commercially available reagents, simple post-treatment procedures, and robust reaction conditions that do not require extreme pressures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
The technological breakthroughs encapsulated in patent CN112239456B represent more than just a chemical curiosity; they are a blueprint for the future of efficient heterocycle manufacturing. At NINGBO INNO PHARMCHEM, we recognize the strategic value of such innovations and have integrated similar advanced catalytic methodologies into our CDMO operations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need grams for early-stage discovery or tons for commercial API launch, we can deliver. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of 2,3-dihydroquinolone intermediate meets the exacting standards required by the global pharmaceutical industry.
We invite you to leverage our expertise to optimize your supply chain and reduce your time-to-market. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific project needs, identifying opportunities to streamline your synthesis and lower overall production costs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us be your trusted partner in navigating the complexities of fine chemical synthesis and delivering high-quality intermediates that drive your success.
