Advanced Copper-Catalyzed Synthesis of 3-Hydroxy-3-Arylindole-2-One Derivatives for Commercial Scale-Up
Introduction to Advanced Indole Scaffold Synthesis
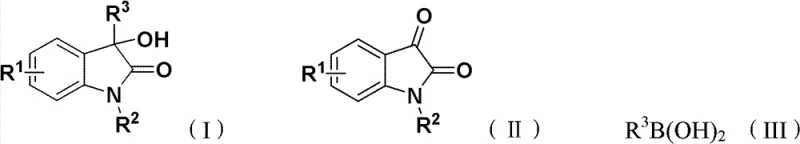
The chemical landscape of pharmaceutical intermediates is constantly evolving, driven by the need for more efficient and sustainable synthetic routes for bioactive scaffolds. Patent CN102432523B introduces a transformative methodology for the synthesis of 3-hydroxy-3-arylindole-2-one derivatives, a class of compounds renowned for their potent biological activities ranging from anticancer to anti-inflammatory properties. This innovation addresses critical bottlenecks in traditional manufacturing by utilizing a robust copper-catalyzed system that operates under remarkably mild conditions. For R&D directors and procurement specialists, this represents a pivotal shift away from expensive precious metal catalysts towards a more economically viable and operationally simple protocol. The strategic implementation of this technology promises to enhance the supply chain stability for high-value API intermediates while maintaining stringent quality standards required by global regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 3-hydroxy-3-arylindole-2-one core has relied heavily on methodologies that present significant economic and operational challenges for industrial scale-up. Traditional approaches often utilize intramolecular cyclization reactions mediated by palladium catalysts, which not only incur substantial material costs due to the price of palladium but also impose strict structural requirements on the starting substrates that limit versatility. Alternatively, the use of highly reactive organometallic reagents such as Grignard or organolithium species introduces severe safety hazards due to their extreme sensitivity to moisture and air, necessitating specialized equipment and rigorous anhydrous conditions that drive up capital expenditure. Furthermore, these aggressive reagents often exhibit poor functional group tolerance, leading to complex impurity profiles that require extensive and costly downstream purification processes to meet pharmaceutical grade specifications.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages a copper-catalyzed cross-coupling strategy between isatin derivatives and arylboronic acids to achieve superior efficiency and selectivity. This methodology employs inexpensive and readily available copper salts, such as copper trifluoromethanesulfonate or copper acetylacetonate, in conjunction with nitrogen-containing bidentate ligands to facilitate the transformation under温和 conditions. The use of arylboronic acids as coupling partners offers a distinct advantage in terms of stability and ease of handling compared to organolithium reagents, thereby simplifying the operational workflow and reducing safety risks in the plant. By shifting to this base-metal catalysis system, manufacturers can significantly lower the cost of goods sold while simultaneously improving the overall robustness of the synthetic route for commercial production.
Mechanistic Insights into Copper-Catalyzed C-H Arylation
The underlying mechanism of this transformation involves a sophisticated catalytic cycle where the copper center facilitates the activation of the isatin substrate and the subsequent transmetallation with the arylboronic acid species. The presence of the nitrogen-containing bidentate ligand, preferably phenanthroline monohydrate, plays a crucial role in stabilizing the active copper species and modulating its electronic properties to enhance reactivity towards the sterically hindered C3 position of the isatin ring. This ligand-accelerated catalysis ensures that the reaction proceeds with high turnover numbers even at relatively low catalyst loadings, typically ranging from 0.01 to 1.0 molar equivalents depending on the specific substrate electronics. The mechanistic pathway avoids the formation of high-energy intermediates that are common in radical-based processes, thereby minimizing side reactions such as homocoupling of the boronic acid or over-oxidation of the indole core which could compromise the final product quality.
From an impurity control perspective, the mild reaction conditions ranging from 30°C to 150°C prevent the thermal degradation of sensitive functional groups that might be present on the aromatic rings of either the isatin or the boronic acid. The selection of dichloromethane or xylene as the inert organic solvent provides an optimal medium for solubilizing the reactants while allowing for easy removal post-reaction, which is critical for minimizing residual solvent impurities in the final API intermediate. Furthermore, the use of inorganic bases like lithium hydroxide or cesium carbonate ensures a controlled pH environment that promotes the formation of the active boronate species without inducing hydrolysis of the lactam ring in the isatin structure. This precise control over the reaction environment results in a clean crude product profile that facilitates straightforward purification via recrystallization, consistently delivering materials with purity levels exceeding 98% as demonstrated in the experimental data.
How to Synthesize 3-Hydroxy-3-Arylindole-2-One Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for generating high-quality indole derivatives suitable for downstream drug development applications. The process begins with the precise charging of stoichiometric amounts of the isatin derivative and arylboronic acid into a reaction vessel, followed by the addition of the copper catalyst system and base in an inert solvent.
- Charge isatin derivatives, arylboronic acids, copper catalyst (e.g., Cu(OTf)2), nitrogen ligand, and base into a reactor with inert solvent.
- Heat the reaction mixture to 45-100°C and maintain stirring for 24-48 hours until conversion is complete.
- Remove solvent, extract with saturated bicarbonate, purify the organic layer, and recrystallize the crude product from ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed synthesis route offers profound strategic advantages that directly impact the bottom line and operational resilience. The substitution of expensive palladium catalysts with abundant copper salts results in a drastic reduction in raw material costs, which is particularly significant when scaling production to multi-ton quantities required for commercial API manufacturing. Additionally, the enhanced stability of arylboronic acids compared to pyrophoric organometallic reagents simplifies logistics and storage requirements, reducing the risk of supply disruptions caused by the specialized handling needs of hazardous chemicals. This robustness translates into a more reliable supply chain capable of meeting tight delivery schedules without the frequent delays associated with managing sensitive reagents or complex purification workflows.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the need for expensive metal scavenging steps and reduces the overall catalyst cost per kilogram of product significantly. By utilizing base metal catalysis, the process avoids the regulatory and environmental costs associated with heavy metal residue limits in pharmaceutical products, streamlining the quality control process. The simplified workup procedure involving solvent evaporation and recrystallization further reduces energy consumption and labor hours compared to chromatographic purification methods. These cumulative efficiencies lead to a substantially lower cost of goods sold, enhancing the competitiveness of the final pharmaceutical intermediate in the global market.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials such as substituted isatins and arylboronic acids ensures a consistent and secure supply of raw materials from multiple vendors. Unlike methods requiring custom-synthesized precursors or air-sensitive reagents, this protocol allows for flexible sourcing strategies that mitigate the risk of single-supplier dependency. The mild reaction conditions also reduce wear and tear on reactor equipment, extending asset life and minimizing unplanned maintenance downtime that could otherwise disrupt production schedules. This operational stability is crucial for maintaining long-term contracts with pharmaceutical clients who demand unwavering supply continuity.
- Scalability and Environmental Compliance: The process is inherently scalable due to the absence of exothermic hazards associated with organometallic additions, allowing for safe expansion from pilot plant to full commercial scale. The use of recyclable solvents and the generation of less hazardous waste streams align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The high atom economy of the coupling reaction minimizes waste generation, reducing disposal costs and the environmental footprint of the manufacturing facility. These factors collectively position the technology as a sustainable solution for the long-term production of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology in an industrial setting.
Q: Why is copper catalysis preferred over palladium for this synthesis?
A: Copper catalysts are significantly more cost-effective than palladium systems and avoid the strict substrate requirements associated with intramolecular cyclization methods, making the process more economically viable for large-scale production.
Q: What are the purity levels achievable with this method?
A: The patented method demonstrates the ability to achieve high purity levels, often exceeding 98%, through simple recrystallization steps, minimizing the need for complex chromatographic purification.
Q: Is this method suitable for sensitive functional groups?
A: Yes, unlike organometallic reagents like Grignard reagents which are highly sensitive to moisture and air, the boronic acid coupling tolerated in this protocol allows for better functional group compatibility and operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Hydroxy-3-Arylindole-2-One Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity 3-hydroxy-3-arylindole-2-one derivatives that meet stringent purity specifications through our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging the copper-catalyzed methodology described in CN102432523B, we offer our partners a competitive edge through cost-effective and reliable manufacturing solutions.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis and obtain specific COA data along with comprehensive route feasibility assessments. Let us collaborate to optimize your supply chain and accelerate the development of your next-generation therapeutic agents with our premium quality intermediates.
