Advanced Palladium-Catalyzed Synthesis of 4-Alkoxy Alkenyl Isoxazole Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 4-Alkoxy Alkenyl Isoxazole Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex heterocyclic scaffolds, particularly isoxazole derivatives which serve as critical pharmacophores in numerous bioactive molecules. A significant technological breakthrough in this domain is documented in Chinese Patent CN113185472B, which discloses a highly efficient method for synthesizing 4-alkoxy alkenyl isoxazole derivatives. This patent outlines a novel palladium-catalyzed tandem cyclization strategy that utilizes O-methyl alkynone oxime ethers and alkyl vinyl ethers as primary substrates. Unlike traditional methods that often require harsh conditions or expensive specialized reagents, this invention operates under mild conditions in an air atmosphere, utilizing common oxidants and ammonium salts to drive the reaction forward. The technical significance of this development lies in its ability to introduce a versatile alkoxy alkenyl side chain at the 4-position of the isoxazole ring, a structural motif that enhances metabolic stability and biological activity in potential drug candidates. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing processes for high-value pharmaceutical intermediates.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 4-alkenyl isoxazole derivatives has relied heavily on palladium-catalyzed intermolecular Heck coupling reactions or 1,3-dipolar cycloadditions. Conventional strategies often involve the reaction of O-methyl oxime ethers with phenylacetylene or alpha-halo olefins to introduce the necessary double bond at the 4-position of the isoxazole ring. However, these traditional pathways suffer from several inherent drawbacks that hinder their industrial applicability. Firstly, the use of alpha-halo olefins or enol derivatives often necessitates strict anhydrous and anaerobic conditions, requiring expensive inert gas protection and specialized equipment that increases capital expenditure. Secondly, the substrate scope in older methods is frequently limited, with poor tolerance for sensitive functional groups such as halogens or electron-withdrawing groups, leading to side reactions and reduced yields. Furthermore, the purification of products from these reactions can be cumbersome due to the formation of complex byproduct mixtures, resulting in higher waste generation and increased downstream processing costs. These limitations create significant bottlenecks for supply chain managers aiming to secure reliable sources of high-purity intermediates for API manufacturing.
The Novel Approach
The methodology described in patent CN113185472B offers a transformative solution by employing alkyl vinyl ethers as the coupling partner in a palladium-catalyzed tandem cyclization. This novel approach fundamentally simplifies the synthetic route by eliminating the need for pre-functionalized halo-olefins, thereby reducing the number of synthetic steps and the overall cost of goods. The reaction proceeds efficiently under an air atmosphere, removing the operational burden of maintaining inert conditions and significantly enhancing process safety. By utilizing inexpensive and commercially available oxidants such as 1,2-naphthoquinone or benzoquinone, along with ammonium salts as additives, the process achieves moderate to good yields with exceptional functional group compatibility. This means that sensitive moieties like halogens, trifluoromethyl groups, and alkoxy substituents remain intact during the transformation, preserving the chemical diversity required for medicinal chemistry optimization. The ability to synthesize diversified 4-alkoxy alkenyl isoxazole structures from readily available raw materials positions this technology as a superior alternative for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Tandem Cyclization
The core of this innovation lies in the intricate catalytic cycle mediated by divalent palladium species. The reaction initiates with the coordination of the palladium catalyst to the alkyne moiety of the O-methyl alkynone oxime ether, activating the triple bond for nucleophilic attack. 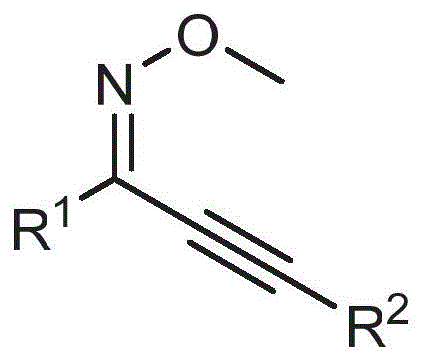 Subsequently, the vinyl ether component undergoes insertion into the palladium-alkyne complex, facilitated by the presence of the ammonium salt which likely acts as a phase transfer catalyst or ligand modifier to stabilize the active catalytic species. This is followed by an intramolecular cyclization where the oxime oxygen attacks the activated carbon center, forming the isoxazole ring skeleton. The final step involves a beta-hydride elimination or oxidative process driven by the quinone oxidant, which regenerates the palladium catalyst and releases the 4-alkoxy alkenyl isoxazole product. Understanding this mechanism is crucial for R&D teams as it highlights the importance of selecting the correct oxidant and solvent system to maintain the catalytic turnover number. The use of ethylene glycol dimethyl ether as a preferred solvent further optimizes the solubility of polar intermediates and stabilizes the transition states, ensuring consistent reaction performance across different substrate batches.
Subsequently, the vinyl ether component undergoes insertion into the palladium-alkyne complex, facilitated by the presence of the ammonium salt which likely acts as a phase transfer catalyst or ligand modifier to stabilize the active catalytic species. This is followed by an intramolecular cyclization where the oxime oxygen attacks the activated carbon center, forming the isoxazole ring skeleton. The final step involves a beta-hydride elimination or oxidative process driven by the quinone oxidant, which regenerates the palladium catalyst and releases the 4-alkoxy alkenyl isoxazole product. Understanding this mechanism is crucial for R&D teams as it highlights the importance of selecting the correct oxidant and solvent system to maintain the catalytic turnover number. The use of ethylene glycol dimethyl ether as a preferred solvent further optimizes the solubility of polar intermediates and stabilizes the transition states, ensuring consistent reaction performance across different substrate batches.
Impurity control is another critical aspect addressed by this mechanistic pathway. The high functional group tolerance observed in this reaction suggests that the catalytic cycle is highly selective for the desired tandem cyclization over competing side reactions such as homocoupling of the alkyne or polymerization of the vinyl ether. The presence of the methoxy group on the oxime ether serves as a directing group that guides the regioselectivity of the cyclization, ensuring that the alkoxy alkenyl chain is installed specifically at the 4-position rather than the 3- or 5-positions. This regiocontrol is vital for maintaining the purity profile of the final intermediate, as positional isomers can be difficult to separate and may exhibit different toxicological properties. By minimizing the formation of regioisomeric impurities, this process reduces the burden on downstream purification units, aligning with the stringent quality standards required for reliable pharmaceutical intermediate suppliers.
How to Synthesize 4-Alkoxy Alkenyl Isoxazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The general procedure involves charging a reaction vessel with the O-methyl alkynone oxime ether and the alkyl vinyl ether in a molar ratio ranging from 1:1 to 1:3. To this mixture, a palladium catalyst such as palladium acetate or palladium bromide is added, typically at a loading of 10 mol%, along with an ammonium salt like tetrabutylammonium bromide. The choice of oxidant is flexible, with 1,2-naphthoquinone showing particularly favorable results in optimizing the reaction kinetics. The reaction is conducted in a solvent such as ethylene glycol dimethyl ether or DMF, often in the presence of 4Å molecular sieves to scavenge trace moisture that could deactivate the catalyst.
- Combine O-methyl alkynone oxime ether and alkyl vinyl ether in a solvent such as ethylene glycol dimethyl ether.
- Add palladium catalyst (e.g., palladium acetate), ammonium salt, oxidant (e.g., 1,2-naphthoquinone), and 4Å molecular sieves.
- Stir the mixture at 60-120°C under air atmosphere for 4-16 hours, then purify via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers substantial strategic benefits for procurement managers and supply chain heads focused on cost reduction in API manufacturing. The primary advantage stems from the use of commodity chemicals as starting materials; alkyl vinyl ethers and O-methyl oxime ethers are widely available in the global chemical market, reducing the risk of supply chain disruptions associated with specialized reagents. Furthermore, the elimination of inert gas protection and the ability to run the reaction under air atmosphere drastically simplifies the engineering requirements for production reactors, allowing for the utilization of standard glass-lined or stainless steel equipment without the need for complex nitrogen blanketing systems. This operational simplicity translates directly into lower capital investment and reduced utility costs, making the process economically attractive for large-scale production.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the avoidance of expensive transition metal catalysts beyond standard palladium salts and the use of cheap organic oxidants. By eliminating the need for cryogenic conditions or high-pressure equipment, the energy consumption per kilogram of product is substantially lowered. Additionally, the high functional group tolerance means that protecting group strategies can often be bypassed, shortening the overall synthetic sequence and reducing the consumption of auxiliary chemicals. These factors combine to deliver a manufacturing process with a markedly improved cost structure compared to legacy methods, providing a competitive edge in pricing for high-purity isoxazole derivatives.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the broad availability of raw materials. Since the process tolerates a wide variety of substituents on the aromatic rings, manufacturers can source diverse starting materials from multiple vendors without compromising reaction performance. The moderate reaction temperatures (60-120°C) and ambient pressure operation reduce the likelihood of thermal runaway incidents or equipment failures, ensuring consistent batch-to-batch output. This reliability is critical for maintaining continuous supply lines to downstream API manufacturers, minimizing the risk of stockouts that could delay drug development timelines or commercial launches.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is facilitated by the homogeneous nature of the catalytic system and the straightforward workup procedure involving extraction and chromatography. The use of recyclable solvents and the potential to recover palladium residues align with modern green chemistry principles, helping companies meet increasingly stringent environmental regulations. The reduction in hazardous waste generation, particularly through the avoidance of halogenated coupling partners, simplifies waste disposal protocols and lowers environmental compliance costs. This sustainability profile makes the technology not only commercially sound but also environmentally responsible, appealing to stakeholders focused on corporate social responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making.
Q: What are the key advantages of this synthesis method over traditional Heck coupling?
A: This method utilizes readily available alkyl vinyl ethers instead of complex alpha-halo olefins, operates under mild air atmosphere without inert gas protection, and demonstrates superior functional group tolerance for halogens and trifluoromethyl groups.
Q: Is this process suitable for large-scale manufacturing of API intermediates?
A: Yes, the process uses inexpensive catalysts and oxidants, avoids cryogenic conditions, and employs simple workup procedures, making it highly scalable for commercial production of pharmaceutical intermediates.
Q: What is the substrate scope regarding functional group compatibility?
A: The reaction tolerates a wide range of substituents including halogens (Cl, Br, F), alkoxy groups, trifluoromethyl groups, and heterocycles, allowing for diverse structural modifications essential for drug discovery.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Alkoxy Alkenyl Isoxazole Supplier
As the demand for complex heterocyclic intermediates continues to grow in the pharmaceutical sector, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from process development to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 4-alkoxy alkenyl isoxazole meets the highest industry standards. We understand the critical nature of timeline adherence in drug development and are committed to delivering high-purity intermediates with the reliability and consistency required by global innovator companies.
We invite you to engage with our technical team to explore how this patented synthesis route can be optimized for your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume requirements. We encourage potential partners to contact our technical procurement team to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this advanced technology into your supply chain. Let us collaborate to accelerate your drug discovery programs with efficient, scalable, and cost-effective chemical solutions.
