Scalable Electrocatalytic Production of High-Purity 6-(Sulfonylmethyl)phenanthridines for Pharma
Introduction to Advanced Electrocatalytic Synthesis
The chemical synthesis landscape is undergoing a transformative shift towards sustainable and cost-effective methodologies, particularly in the production of complex heterocyclic intermediates essential for pharmaceutical development. Patent CN110714208B introduces a groundbreaking electrocatalytic protocol for the preparation of 6-(sulfonylmethyl)phenanthridine compounds, addressing critical limitations inherent in traditional synthetic routes. This innovation leverages organic electrochemical catalysis to drive oxidation-reduction reactions directly through electron transfer at the electrode surface, thereby circumventing the need for stoichiometric oxidants or precious metal catalysts. For R&D directors and procurement specialists, this represents a pivotal advancement, as it not only simplifies the reaction setup by removing the requirement for specialized light irradiation equipment but also drastically mitigates the risk of heavy metal contamination in the final active pharmaceutical ingredients. The ability to utilize a mixed solvent system of acetonitrile and water further underscores the environmental and economic benefits, positioning this technology as a superior alternative for the reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of phenanthridine scaffolds has relied heavily on methods such as the Bischler-Napieralski cyclization or, more recently, visible-light-induced radical sulfonylation. While effective on a small laboratory scale, these conventional approaches suffer from significant drawbacks that hinder their commercial viability and supply chain robustness. Specifically, the prior art methods described in the patent background necessitate the use of expensive photosensitizers like tris(bipyridine)ruthenium(II) chloride hexahydrate, which commands a market price as high as 204,000 yuan per kilogram, creating an unsustainable cost structure for large-scale manufacturing. Furthermore, these photoredox reactions require specific LED light sources and strictly anhydrous organic solvents, imposing high equipment requirements and generating substantial organic waste. The reliance on transition metals also introduces the persistent challenge of metal residue removal, necessitating additional purification steps that erode overall yield and extend production lead times, making them less ideal for the commercial scale-up of complex pharmaceutical intermediates.
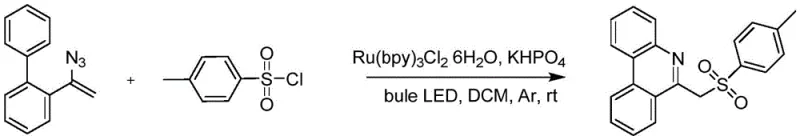
The Novel Approach
In stark contrast to the resource-intensive legacy methods, the novel electrocatalytic approach disclosed in the patent offers a streamlined and economically superior pathway. By utilizing electricity as the primary reagent to generate sulfonyl radicals from sulfonyl hydrazides, this method completely eliminates the dependency on costly ruthenium catalysts and external light sources. The reaction proceeds under mild conditions using a simple constant current power supply, which is far more accessible and easier to maintain than specialized photochemical reactors. Moreover, the introduction of water as a co-solvent alongside acetonitrile not only reduces the consumption of pure organic solvents but also aligns with green chemistry principles by minimizing environmental pollution. This shift from photochemical to electrochemical activation fundamentally alters the cost equation, offering substantial cost savings in API manufacturing while ensuring a cleaner product profile free from toxic metal contaminants, thereby enhancing the overall reliability of the supply chain for critical drug substances.
Mechanistic Insights into Electrochemical Radical Cyclization
The core of this technological breakthrough lies in the precise control of electron transfer at the electrode interface, which facilitates the generation of reactive intermediates without chemical oxidants. In this mechanism, the sulfonyl hydrazide substrate undergoes anodic oxidation at the graphite electrode, losing electrons to form a sulfonyl radical species. This highly reactive radical then attacks the vinyl azide moiety of the biphenyl substrate, initiating a cascade of intramolecular cyclization events. The subsequent loss of nitrogen gas drives the formation of the phenanthridine ring system, resulting in the stable 6-(sulfonylmethyl)phenanthridine product. This direct electrochemical activation ensures high atom economy and selectivity, as the reaction potential can be finely tuned by adjusting the current density, typically optimized around 12mA in the disclosed embodiments. For technical teams, understanding this mechanism is crucial for troubleshooting and optimization, as it highlights the importance of electrolyte conductivity and electrode surface area in maintaining consistent reaction rates and minimizing side reactions.
From an impurity control perspective, the absence of transition metal catalysts is a decisive advantage for pharmaceutical applications. Traditional ruthenium-catalyzed routes often leave trace amounts of heavy metals that are difficult to remove and strictly regulated in final drug products. The electrochemical method inherently avoids this issue, as the only reagents consumed are the organic substrates and the electrolyte, which can be easily separated during workup. Additionally, the use of a mixed aqueous-organic solvent system helps to suppress the formation of certain organic byproducts that might proliferate in purely organic media. The patent data indicates that optimizing the volume ratio of acetonitrile to water to approximately 18:1 maximizes yield while maintaining solubility, suggesting a delicate balance that enhances reaction efficiency. This level of control over the reaction environment ensures that the resulting phenanthridine derivatives meet stringent purity specifications required for downstream medicinal chemistry applications.
How to Synthesize 6-(Sulfonylmethyl)phenanthridines Efficiently
Implementing this electrocatalytic protocol requires careful attention to reaction parameters to replicate the high yields reported in the patent literature. The process begins with the preparation of a homogeneous reaction mixture containing the olefin azide and sulfonyl hydrazide substrates, along with potassium carbonate as a base and tetrabutylammonium tetrafluoroborate as the supporting electrolyte. The choice of solvent is critical, with a specific volume ratio of acetonitrile to water proven to be optimal for balancing substrate solubility and electrochemical conductivity. Once the mixture is prepared under an inert nitrogen atmosphere to prevent unwanted oxidation by air, the electrochemical cell is assembled using a graphite working electrode and a platinum counter electrode. The detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining olefin azide, sulfonyl hydrazide, and potassium carbonate in an acetonitrile-water solvent system with tetrabutylammonium tetrafluoroborate electrolyte.
- Insert graphite and platinum electrodes into the solution and apply a constant current of 10mA to 16mA for 3 to 5 hours under nitrogen atmosphere.
- Upon completion, extract the product with dichloromethane, dry the organic phase, and purify via column chromatography to isolate the target phenanthridine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrocatalytic technology translates into tangible strategic benefits that extend beyond simple unit cost reductions. The elimination of the ruthenium photocatalyst, which is not only exorbitantly priced but also subject to supply volatility due to its status as a precious metal, removes a significant bottleneck from the raw material sourcing process. This shift ensures enhanced supply chain reliability, as the requisite starting materials—vinyl azides and sulfonyl hydrazides—are commodity chemicals with robust global availability. Furthermore, the simplification of the reaction apparatus from complex photochemical setups to standard electrochemical cells reduces capital expenditure on specialized equipment and lowers maintenance overheads. These factors collectively contribute to a more resilient manufacturing infrastructure capable of sustaining continuous production schedules without the risk of catalyst shortages or equipment failure.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of the expensive ruthenium catalyst and the associated initiator reagents from the bill of materials. By replacing these high-cost inputs with electricity and inexpensive electrolytes, the variable cost per kilogram of the product is drastically lowered. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces labor and consumable costs associated with purification. This structural change in the cost base allows for more competitive pricing strategies and improved margin protection, especially when scaling production volumes where catalyst costs would otherwise become prohibitive.
- Enhanced Supply Chain Reliability: Relying on a synthesis route that depends on rare earth or precious metal catalysts introduces inherent supply chain risks, including price spikes and geopolitical sourcing issues. This electrochemical method mitigates these risks by utilizing universally available electrical energy and common organic reagents. The robustness of the reaction conditions, which tolerate a variety of functional groups and do not require stringent exclusion of moisture beyond standard inert atmosphere practices, further ensures consistent batch-to-batch quality. This reliability is paramount for maintaining uninterrupted supply to downstream pharmaceutical customers who demand strict adherence to delivery timelines.
- Scalability and Environmental Compliance: Scaling electrochemical reactions is inherently more straightforward than scaling photochemical ones, as light penetration depth is not a limiting factor in larger reactors. This facilitates the commercial scale-up of complex phenanthridine derivatives from pilot batches to multi-ton production without significant re-engineering of the process. Moreover, the reduced usage of pure organic solvents through the introduction of water aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. This compliance advantage reduces the burden on waste treatment facilities and minimizes the environmental footprint of the manufacturing process, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic synthesis method. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: What are the primary advantages of this electrochemical method over traditional photoredox catalysis?
A: The electrochemical method eliminates the need for expensive ruthenium-based photocatalysts and specialized light sources, significantly reducing raw material costs and equipment complexity while avoiding toxic metal residues in the final product.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the use of standard electrochemical cells and readily available reagents like sulfonyl hydrazides makes this process highly amenable to scale-up, offering better supply chain reliability compared to light-dependent methods.
Q: What is the typical yield range for these phenanthridine derivatives?
A: According to the patent data, optimized conditions using a constant current of 12mA and an acetonitrile-water ratio of 18:1 can achieve yields exceeding 86%, demonstrating high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-(Sulfonylmethyl)phenanthridines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this electrocatalytic technology in delivering high-value pharmaceutical intermediates with superior efficiency and purity. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and robust. Our state-of-the-art facilities are equipped to handle electrochemical processes with precision, supported by rigorous QC labs that enforce stringent purity specifications to meet the exacting standards of the global pharmaceutical industry. We are committed to leveraging such innovative synthetic routes to provide our clients with a competitive edge in their drug development pipelines.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume needs. We encourage you to contact us today to obtain specific COA data for our phenanthridine derivatives and to receive comprehensive route feasibility assessments that will accelerate your path to market.
