Advanced Synthesis of 1-Naphthol Phosphine Oxide Ligands for Commercial Scale-up
Advanced Synthesis of 1-Naphthol Phosphine Oxide Ligands for Commercial Scale-up
The landscape of organophosphorus chemistry is continually evolving, driven by the demand for more efficient and versatile ligands for homogeneous catalysis. A significant breakthrough in this domain is documented in patent CN110183488B, which details a novel preparation method for 1-naphthol-based phosphine oxide ligands. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-step annulation process. By leveraging a sophisticated Rhodium(III)-catalyzed system, this method enables the direct construction of the complex naphthol scaffold from readily available sulfur ylide compounds and phenylethynyl phosphine oxide derivatives. For R&D directors and procurement specialists in the fine chemical sector, this innovation offers a compelling value proposition: it drastically simplifies the supply chain for high-value ligands while ensuring exceptional purity and structural diversity. The ability to access these difficult-to-synthesize molecules through a single operational step under mild thermal conditions marks a substantial advancement in synthetic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-naphthol-based phosphine oxide ligands has been plagued by significant synthetic hurdles that impede their widespread adoption in industrial catalysis. Conventional routes often require harsh reaction conditions, involving strong bases, high temperatures, or toxic reagents that pose safety and environmental risks. Furthermore, traditional methodologies typically necessitate multi-step sequences to build the naphthalene core before introducing the phosphine oxide moiety, leading to cumulative yield losses and increased waste generation. The purification of intermediates in these lengthy pathways is often cumbersome, requiring extensive chromatographic separation that drives up production costs and extends lead times. Additionally, the structural rigidity of older methods limits the diversity of substituents that can be introduced, restricting the tunability of the final ligand for specific catalytic applications. These factors collectively create a bottleneck for the reliable supply of high-purity pharmaceutical intermediates and specialty ligands.
The Novel Approach
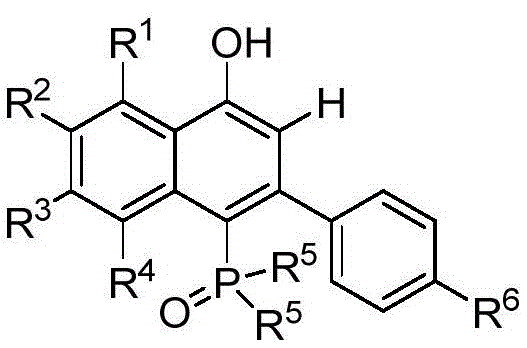
In stark contrast, the methodology disclosed in patent CN110183483488B introduces a transformative one-step annulation strategy that elegantly bypasses these historical limitations. By utilizing a sulfur ylide compound and a phenylethynyl phosphine oxide derivative as the primary building blocks, the reaction constructs the entire 1-naphthol framework in a single operational pot. This approach operates under remarkably mild conditions, typically between 60°C and 100°C in a nitrogen atmosphere, which preserves sensitive functional groups and minimizes side reactions. The use of a specialized Rhodium catalyst combined with a Lewis acid additive facilitates a highly selective cyclization, ensuring that the desired regioisomer is formed with high fidelity. This streamlined process not only accelerates the timeline from raw materials to finished product but also significantly enhances the overall atom economy. The result is a robust synthetic platform capable of generating a wide array of substituted ligands with superior yields compared to legacy methods.
Mechanistic Insights into Rhodium-Catalyzed Annulation
The core of this technological advancement lies in the intricate catalytic cycle mediated by the pentamethylcyclopentadienyl rhodium(III) complex. The reaction initiates with the coordination of the phenylethynyl phosphine oxide derivative to the Rh(III) center, activating the alkyne moiety towards nucleophilic attack. Simultaneously, the sulfur ylide acts as a unique carbon source, undergoing decomposition or transformation to generate a reactive carbene-like species or equivalent intermediate that engages with the activated alkyne. The presence of the Lewis acid, such as zinc acetate, plays a critical role in stabilizing transition states and promoting the subsequent cyclization step that forms the naphthalene ring system. This synergistic interaction between the transition metal catalyst and the Lewis acid ensures that the reaction proceeds with high regioselectivity, favoring the formation of the 4-hydroxy-2-phenyl naphthyl backbone. The mechanistic elegance of this pathway allows for the tolerance of diverse electronic environments on the aromatic rings, enabling the synthesis of ligands with halogen, alkyl, or alkoxy substituents without compromising efficiency.
Understanding the impurity profile is crucial for R&D teams aiming to replicate this process at scale. The high selectivity of the Rh-catalyzed system inherently minimizes the formation of by-products such as polymerized alkynes or unreacted starting materials. However, trace amounts of rhodium residues must be managed, particularly for ligands intended for pharmaceutical applications where heavy metal limits are stringent. The protocol described involves a straightforward workup procedure, typically involving solvent removal and silica gel chromatography, which effectively separates the target ligand from catalyst residues and minor organic impurities. The structural integrity of the phosphine oxide group is maintained throughout the reaction, preventing oxidation to phosphates or reduction to phosphines, which ensures the final product retains its intended coordination properties. This level of control over the杂质谱 (impurity profile) is a key differentiator, providing procurement managers with confidence in the consistency and quality of the supplied material.
How to Synthesize 1-Naphthol Phosphine Oxide Ligand Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the rigorous drying of glassware and solvents to prevent catalyst deactivation, followed by the sequential addition of the phenylethynyl phosphine oxide, sulfur ylide, catalyst, and Lewis acid under an inert atmosphere. The reaction mixture is then heated to the optimal temperature range, typically around 80°C, and stirred for a duration of 6 to 16 hours to ensure complete conversion. Upon completion, the solvent is removed under reduced pressure, and the crude residue is purified using standard chromatographic techniques to isolate the pure ligand. This standardized approach allows for the reproducible manufacture of complex ligands, making it an ideal candidate for technology transfer to pilot and production scales. For a detailed visualization of the target molecular architecture achieved through this method, refer to the specific product structure below.
- Mix sulfur ylide compound, phenylethynyl phosphine oxide derivative, Rh catalyst, Lewis acid, and organic solvent under nitrogen.
- Heat the reaction mixture to 60-100°C and maintain for 6-16 hours to facilitate cyclization.
- Purify the crude product via silica gel column chromatography to obtain the high-purity ligand.
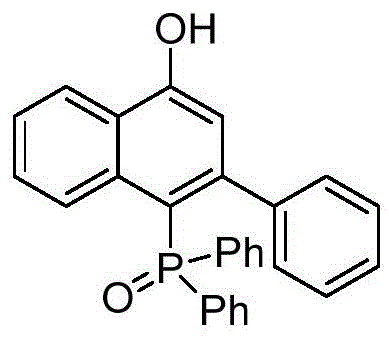
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound advantages for supply chain stability and cost management. The elimination of multiple synthetic steps translates directly into reduced operational expenditures, as fewer unit operations mean lower labor, energy, and equipment utilization costs. Furthermore, the use of commercially available starting materials mitigates the risk of raw material shortages, ensuring a continuous and reliable flow of production. The mild reaction conditions also reduce the burden on safety infrastructure, lowering the overhead associated with handling hazardous reagents or extreme temperatures. For procurement managers, this means a more predictable pricing model and the ability to secure long-term contracts with greater confidence. The scalability of the process ensures that demand surges can be met without the need for extensive process re-engineering, providing a resilient supply chain for critical chemical intermediates.
- Cost Reduction in Manufacturing: The one-step nature of this synthesis fundamentally alters the cost structure of ligand production. By consolidating what was previously a multi-stage process into a single reaction vessel, manufacturers can achieve substantial savings in solvent consumption, waste disposal, and processing time. The high yields reported in the patent data indicate that raw material utilization is optimized, minimizing the financial loss associated with low-conversion steps. Additionally, the avoidance of expensive protecting group strategies further reduces the bill of materials. These efficiencies accumulate to offer a significantly lower cost of goods sold (COGS), allowing downstream users to benefit from more competitive pricing without sacrificing quality. The economic viability of this route makes it an attractive option for large-volume manufacturing of specialty chemicals.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the accessibility of the reagents. Unlike processes that rely on exotic or unstable intermediates, this method utilizes stable sulfur ylides and alkynes that are readily sourced from established chemical suppliers. The tolerance of the reaction to various substituents means that supply disruptions for one specific derivative can often be mitigated by switching to an alternative analog without halting production entirely. This flexibility is invaluable for maintaining continuity in the face of global logistical challenges. Moreover, the simplified purification process reduces the dependency on specialized separation technologies, further decentralizing the risk of production bottlenecks and ensuring a steady delivery schedule for clients.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton quantities is facilitated by the homogeneous nature of the catalytic system and the moderate thermal requirements. The reaction does not generate excessive heat or pressure, simplifying the engineering controls needed for larger reactors. From an environmental standpoint, the improved atom economy and reduced solvent usage align with green chemistry principles, helping companies meet increasingly stringent regulatory standards. The minimized waste stream lowers the environmental footprint of the manufacturing process, reducing disposal costs and enhancing the sustainability profile of the final product. This alignment with environmental, social, and governance (ESG) goals is a critical factor for modern supply chain decision-makers seeking responsible partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these advanced ligands. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows. The insights provided here aim to clarify the operational benefits and technical capabilities associated with this novel synthetic route.
Q: What are the key advantages of this Rh-catalyzed method over traditional synthesis?
A: This method allows for a one-step construction of the naphthol core under mild conditions (60-100°C), avoiding the harsh reagents and multi-step sequences typical of conventional routes, resulting in significantly higher yields and simpler purification.
Q: Is this process suitable for large-scale manufacturing of ligands?
A: Yes, the process utilizes commercially available starting materials and standard solvents like DCE or toluene, with robust reaction conditions that are highly amenable to scale-up for industrial production of complex ligands.
Q: What is the substrate scope for this phosphine oxide ligand synthesis?
A: The method demonstrates excellent substrate adaptability, tolerating various substituents such as halogens, alkyl groups, and methoxy groups on both the sulfur ylide and the alkyne components, allowing for diverse ligand libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Naphthol Phosphine Oxide Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ligands play in driving innovation in catalysis and material science. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of specific derivatives or bulk supply of standard ligands, our infrastructure is designed to support your most demanding projects with precision and reliability. We understand the nuances of organophosphorus chemistry and are prepared to tackle the complexities of scaling sensitive catalytic processes.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner dedicated to excellence, innovation, and long-term value creation in the fine chemical industry.
