Advanced Pyranose Ring Opening Technology for Scalable Pharmaceutical Intermediate Manufacturing
Introduction to Advanced Pyranose Ring-Opening Technology
The landscape of carbohydrate chemistry is undergoing a significant transformation driven by the need for more sustainable and efficient synthetic routes for complex sugar derivatives. Patent CN114671849B introduces a groundbreaking methodology for the ring-opening of pyranose rings, specifically targeting methyl-α-D-glucopyranoside to generate valuable open-chain derivatives. This technology addresses critical bottlenecks in the synthesis of enoses, which are pivotal synthons for glycosides, oligosaccharides, and biomacromolecules. By leveraging a unique sulfonation strategy to induce ring strain, this invention enables reactions under remarkably mild conditions, bypassing the severe limitations of legacy protocols. For R&D directors and procurement specialists alike, this represents a paradigm shift towards greener, higher-yielding processes that align with modern regulatory and economic demands.
The core innovation lies in the strategic manipulation of the pyranose ring's conformational energy. Traditional approaches often struggle with the inherent stability of the six-membered ring, necessitating aggressive reagents that compromise atom economy and safety. In contrast, the disclosed method transforms the stable glucopyranoside into a highly reactive intermediate through precise 2,3-position sulfonation. This structural modification creates a twisted ring geometry that stores potential energy, effectively priming the molecule for cleavage. The result is a robust pathway to open-chain structures that serves as a versatile platform for downstream applications, ranging from fine chemical intermediates to advanced tumor diagnostic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the opening of oxygen-containing pyranose rings has relied heavily on reactions with thiols and thiophenols or catalysis by strong Lewis acids. These conventional pathways are fraught with significant operational and environmental drawbacks that hinder their viability in large-scale manufacturing. The use of sulfur-containing reagents invariably leads to the generation of large quantities of pungent, toxic gases, posing severe challenges for waste management and worker safety. Furthermore, these reactions typically demand harsh conditions, including elevated temperatures or strictly anhydrous environments, which increase energy consumption and equipment costs. The complexity of purifying products from sulfur-laden byproducts further erodes the overall process efficiency, making these methods less attractive for cost-sensitive pharmaceutical supply chains.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by employing a sulfonate esterification strategy that fundamentally alters the reactivity profile of the substrate. By converting the 2,3-hydroxyl groups into sulfonate esters, the method induces a twisted ring structure that dramatically enhances internal tension within the pyranose ring. This stored strain energy lowers the activation barrier for ring opening, allowing the reaction to proceed smoothly under mild acidic conditions at room temperature (15-25°C). 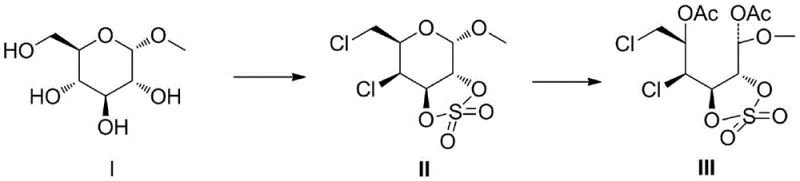 This gentle protocol not only eliminates the release of noxious gases but also achieves exceptional yields, as demonstrated by the 93.3% yield reported in specific embodiments. The simplicity of the workup, involving standard extraction and chromatography, underscores the practical superiority of this method for industrial adoption.
This gentle protocol not only eliminates the release of noxious gases but also achieves exceptional yields, as demonstrated by the 93.3% yield reported in specific embodiments. The simplicity of the workup, involving standard extraction and chromatography, underscores the practical superiority of this method for industrial adoption.
Mechanistic Insights into Sulfonate-Induced Ring Strain and Cleavage
The mechanistic elegance of this process centers on the concept of strain-promoted reactivity. When the 2,3-positions of the glucopyranoside are sulfonated, the resulting cyclic sulfonate adopts a distorted conformation that deviates from the ideal chair form. This distortion creates significant steric hindrance and torsional stress, destabilizing the ring system. Upon exposure to an electrophilic acid catalyst, such as concentrated sulfuric acid, the anomeric carbon becomes highly susceptible to nucleophilic attack or ionization. The release of ring tension acts as a powerful thermodynamic driving force, facilitating the formation of an open-chain carbocation intermediate that is rapidly trapped by acylating agents like acetic anhydride. This sequence ensures high regioselectivity and prevents the formation of unwanted side products often seen in non-strained systems.
Beyond the primary ring-opening event, this chemistry offers profound implications for the synthesis of complex bioactive molecules. The resulting open-chain derivatives possess multiple functional handles that can be selectively manipulated. For instance, the patent illustrates the conversion of these intermediates into precursors for tumor diagnostic reagents, such as 18F-FDG. 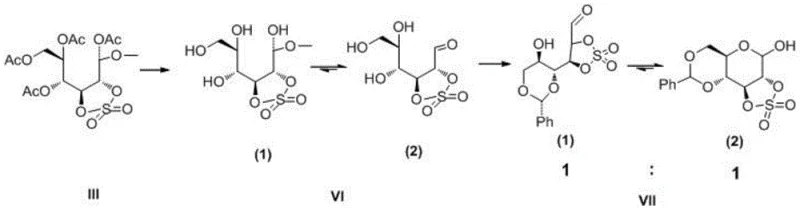 The ability to protect specific hydroxyl groups while leaving others reactive allows for the precise construction of radiolabeled sugars used in PET imaging. This level of control is essential for producing high-purity pharmaceutical intermediates where isotopic labeling efficiency and specific activity are critical parameters for clinical efficacy.
The ability to protect specific hydroxyl groups while leaving others reactive allows for the precise construction of radiolabeled sugars used in PET imaging. This level of control is essential for producing high-purity pharmaceutical intermediates where isotopic labeling efficiency and specific activity are critical parameters for clinical efficacy.
How to Synthesize Ring-Opened Glucopyranoside Derivatives Efficiently
Implementing this synthesis requires careful attention to stoichiometry and reaction monitoring to maximize the benefits of the strained intermediate. The process begins with the preparation of the sulfonated cyclic intermediate, followed by the acid-catalyzed ring opening in the presence of an anhydride. Detailed operational parameters, including solvent ratios and temperature controls, are critical for reproducibility. The following guide outlines the standardized procedure derived from the patent examples, ensuring that technical teams can replicate the high yields and purity profiles described.
- Sulfonate the 2,3-positions of methyl-α-D-glucopyranoside using sulfuryl chloride and pyridine to form the twisted cyclic sulfonate intermediate (Compound II).
- Subject Compound II to ring-opening esterification using acetic anhydride and concentrated sulfuric acid at room temperature to generate the open-chain derivative (Compound III).
- Purify the final product via silica gel column chromatography to achieve high purity suitable for downstream pharmaceutical applications.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial advantages that directly impact the bottom line and supply chain resilience. The elimination of hazardous thiol reagents simplifies regulatory compliance and reduces the costs associated with specialized waste disposal and ventilation systems. Operating at ambient temperatures significantly lowers energy expenditures compared to thermal processes, contributing to a smaller carbon footprint and reduced utility costs. These factors combine to create a more economically viable manufacturing process that is less susceptible to fluctuations in energy markets and environmental regulations.
- Cost Reduction in Manufacturing: The substitution of expensive or hazardous reagents with commodity chemicals like sulfuryl chloride and acetic anhydride drives down raw material costs. Additionally, the high yields achieved (over 90% in key steps) minimize material loss and reduce the burden on purification units. The absence of pungent gas byproducts eliminates the need for complex scrubbing systems, leading to significant capital expenditure savings and lower operational overheads for facility maintenance.
- Enhanced Supply Chain Reliability: The reliance on widely available, stable reagents ensures a consistent supply of inputs, mitigating the risk of production delays caused by specialty chemical shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in environmental parameters, enhancing batch-to-b consistency. This reliability is crucial for maintaining continuous production schedules and meeting the stringent delivery timelines required by global pharmaceutical partners.
- Scalability and Environmental Compliance: The mild nature of the reaction facilitates straightforward scale-up from laboratory to commercial production without the need for exotic high-pressure reactors. The environmentally friendly profile, characterized by the absence of toxic gas emissions, aligns perfectly with increasingly strict global environmental standards. This compliance advantage future-proofs the manufacturing asset against tightening regulations, ensuring long-term operational continuity and reducing the risk of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ring-opening technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the method for potential partners and licensees.
Q: What are the advantages of this ring-opening method over traditional thiol-based methods?
A: Unlike traditional methods that use thiols or thiophenols which produce pungent gases and require harsh conditions, this patented method utilizes 2,3-sulfonation to create ring strain, allowing for mild, room-temperature acidic conditions with high yields and no offensive odors.
Q: Can this method be applied to the synthesis of tumor diagnostic reagents?
A: Yes, the resulting open-chain derivatives serve as excellent precursors for tumor diagnostic reagents like 18F-FDG. The method allows for high-selectivity functionalization at the 2-position, facilitating the production of targeted imaging agents.
Q: Is this process scalable for commercial production?
A: The process operates at ambient temperatures (15-25°C) and uses common reagents like acetic anhydride and sulfuric acid, eliminating the need for specialized high-pressure or cryogenic equipment, which significantly enhances scalability and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl-α-D-glucopyranoside Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced ring-opening technology for the production of high-value carbohydrate intermediates. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are seamlessly translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of methyl-α-D-glucopyranoside derivatives meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline while optimizing your manufacturing costs.
