Revolutionizing Photoinitiator FMT Production: A Green Solid-Acid Catalytic Route for High-Purity Intermediates
Introduction to Advanced Photoinitiator Intermediate Synthesis
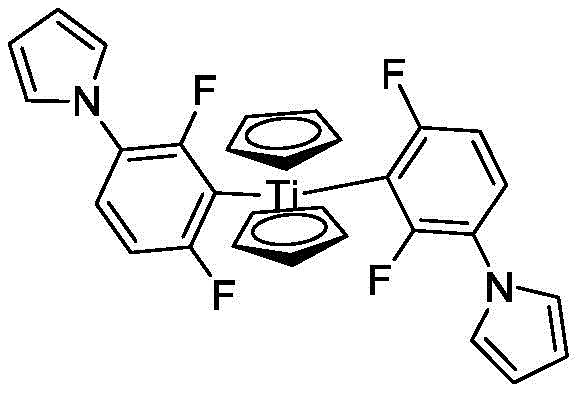
The global demand for high-performance photoinitiators, particularly titanocene-based structures like Photoinitiator FMT (CAS 125051-32-3), has surged due to their critical role in UV curing systems, PCB manufacturing, and advanced dental materials. At the heart of this value chain lies the efficient production of its key precursor, 2,4-difluorophenylpyrrole. Recent intellectual property developments, specifically patent CN111205211B, have unveiled a transformative synthetic methodology that addresses long-standing bottlenecks in yield, purity, and environmental impact. This patent details a novel approach utilizing sulfonated polystyrene microspheres as a heterogeneous catalyst, marking a significant departure from traditional homogeneous transition metal or harsh acid-catalyzed processes. By leveraging this technology, manufacturers can achieve yields exceeding 90% with product purity surpassing 99.0%, ensuring a robust supply of high-quality intermediates for the electronics and pharmaceutical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
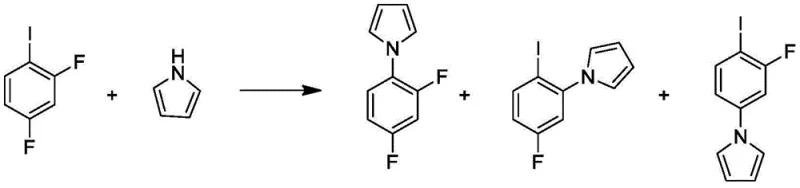
Historically, the synthesis of 2,4-difluorophenylpyrrole has been plagued by significant technical and economic inefficiencies. One prevalent route involves the coupling of 2,4-difluoroiodobenzene with pyrrole using copper catalysts and cesium carbonate. As illustrated in prior art, this method suffers from poor regioselectivity, inevitably generating a complex mixture of isomeric byproducts that possess physical properties strikingly similar to the target molecule. This similarity creates a nightmare for purification teams, often requiring extensive column chromatography which is economically unviable at an industrial scale and leads to substantial material loss. Furthermore, alternative routes utilizing 2,5-dimethoxytetrahydrofuran in acetic acid solvents present their own set of challenges, including insufficient yields ranging typically between 75% and 80%, and the generation of massive volumes of acidic wastewater that complicate environmental compliance.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111205211B introduces a streamlined, green chemistry approach centered on solid-acid catalysis. By employing sulfonated polystyrene microspheres, the reaction proceeds through a clean dehydration pathway between 2,4-difluoroaniline and furan. This heterogeneous system eliminates the need for toxic heavy metals like copper and avoids the corrosive waste streams associated with liquid acids. The solid nature of the catalyst allows for simple filtration and direct reuse, fundamentally altering the cost structure of the manufacturing process. Moreover, the reaction conditions are remarkably mild, operating effectively at temperatures between 60°C and 80°C without the stringent requirement for anhydrous or oxygen-free environments that characterize other high-cost synthetic routes. This robustness translates directly into lower operational expenditures and enhanced process safety for large-scale production facilities.
Mechanistic Insights into Solid-Acid Catalyzed Condensation
The efficacy of this novel synthesis lies in the unique mechanistic behavior of the sulfonated polystyrene microspheres within the polar solvent matrix. These microspheres function as a Brønsted acid, protonating the furan ring to increase its electrophilicity, thereby facilitating a nucleophilic attack by the amino group of the 2,4-difluoroaniline. This interaction promotes a sequential condensation and cyclization sequence that efficiently constructs the pyrrole ring while eliminating water molecules. Unlike homogeneous catalysts that dissolve into the reaction medium and require complex quenching and extraction steps, the polymeric backbone of the catalyst ensures it remains insoluble throughout the process. This phase separation is critical for maintaining high turnover numbers and preventing catalyst degradation, which is a common failure mode in liquid-phase acid catalysis. The result is a highly selective transformation that minimizes side reactions such as polymerization of the furan or over-alkylation of the aniline.

From an impurity control perspective, this mechanism offers superior predictability and manageability. The steric environment provided by the cross-linked polystyrene matrix can subtly influence the transition state, favoring the formation of the desired 2,4-substituted product over potential regioisomers. In traditional copper-catalyzed couplings, the lack of such steric control often leads to a statistical distribution of products. Here, the specific activation of the furan substrate ensures that the reaction pathway is kinetically driven towards the target architecture. Furthermore, the ability to recycle the solvent and excess furan via reduced pressure distillation means that any unreacted starting materials are not lost but rather reintroduced into the cycle. This closed-loop material flow not only boosts the overall atom economy but also ensures that the final crude product entering the crystallization stage is already of high quality, reducing the burden on downstream purification units.
How to Synthesize 2,4-Difluorophenylpyrrole Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the solid-acid catalyst. The process begins with the preparation of the reaction mixture, where 2,4-difluoroaniline and furan are combined in a polar solvent such as toluene or N,N-dimethylformamide. The molar ratio is carefully adjusted, typically maintaining a slight excess of furan (1:1.5 to 1:3) to drive the equilibrium forward. Once the sulfonated polystyrene microspheres are added, the system is sealed and heated. The detailed standardized synthesis steps, including specific workup procedures and crystallization protocols, are outlined in the guide below.
- Prepare the reaction system by mixing 2,4-difluoroaniline and furan in a polar solvent like toluene or DMF with sulfonated polystyrene microspheres.
- Heat the sealed mixture to 60-80°C for 12-18 hours to facilitate the catalytic dehydration reaction.
- Filter to recover the solid catalyst, concentrate the filtrate to recover solvent/furan, and crystallize the residue with non-polar solvents to obtain the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this solid-acid catalyzed route represents a strategic opportunity to optimize both cost structures and supply reliability. The elimination of expensive transition metal catalysts like copper or specialized magnetic nanospheres removes a significant variable cost component from the bill of materials. Additionally, the ability to recover and reuse the catalyst multiple times without significant loss of activity means that the effective cost per kilogram of catalyst consumed is drastically reduced. This efficiency is compounded by the recovery of solvents and excess raw materials, which transforms what would traditionally be waste disposal costs into recovered asset value. Consequently, the overall manufacturing cost for high-purity 2,4-difluorophenylpyrrole is significantly lowered, providing a competitive edge in pricing negotiations with downstream formulators.
- Cost Reduction in Manufacturing: The transition from homogeneous to heterogeneous catalysis eliminates the need for complex aqueous workups and heavy metal scavenging steps, which are both time-consuming and costly. By simply filtering the solid catalyst, the process streamlines the isolation of the product, reducing labor hours and utility consumption associated with distillation and extraction. Furthermore, the high yield of over 90% ensures that raw material utilization is maximized, minimizing the cost of goods sold (COGS) related to starting materials like 2,4-difluoroaniline.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as furan and 2,4-difluoroaniline, which are widely available industrial products, mitigates the risk of raw material shortages. Unlike specialized reagents that may have single-source suppliers, these feedstocks can be sourced from multiple vendors globally, ensuring continuity of supply. The robustness of the reaction conditions, which do not require stringent anhydrous or inert atmospheres, also reduces the risk of batch failures due to environmental fluctuations, leading to more predictable production schedules and shorter lead times for customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the simplicity of the unit operations involved—mixing, heating, filtration, and distillation. The absence of hazardous heavy metals and corrosive liquid acids simplifies waste treatment protocols, making it easier for manufacturing sites to comply with increasingly strict environmental regulations. This "green" profile not only reduces regulatory risk but also aligns with the sustainability goals of major multinational corporations, enhancing the marketability of the final photoinitiator products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a factual basis for evaluating the technology's viability for your specific application needs.
Q: What are the advantages of using sulfonated polystyrene microspheres over traditional copper catalysts?
A: Unlike copper catalysts which suffer from poor regioselectivity and generate difficult-to-separate byproducts, sulfonated polystyrene microspheres act as a heterogeneous solid acid. This allows for easy filtration and recycling, eliminates heavy metal contamination, and significantly simplifies the purification process while maintaining high yields above 90%.
Q: How does this new method improve environmental compliance compared to acetic acid routes?
A: Traditional acetic acid routes generate large amounts of waste acid and water that are difficult to treat. The new method utilizes recyclable polar solvents like toluene or DMF and a solid catalyst that can be reused multiple times, drastically reducing liquid waste discharge and aligning with stricter environmental regulations.
Q: Is the 2,4-difluorophenylpyrrole produced suitable for high-end electronic applications?
A: Yes, the process consistently achieves a purity of greater than or equal to 99.0% as detected by HPLC. This high level of purity is critical for downstream applications in photoinitiator FMT production, which is used in sensitive areas like PCB resists, holographic imaging, and dental materials where impurity profiles must be tightly controlled.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Difluorophenylpyrrole Supplier
As the demand for advanced photoinitiators continues to grow across the electronics and healthcare sectors, securing a stable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of 2,4-difluorophenylpyrrole meets the exacting standards required for high-end UV curing applications. We understand the critical nature of supply chain continuity and are committed to delivering consistent quality that enables your downstream processes to run smoothly.
We invite you to engage with our technical team to discuss how this innovative synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener, more efficient manufacturing method. Please contact our technical procurement team today to obtain specific COA data and route feasibility assessments, and let us partner with you to drive value and efficiency in your supply chain.
