Optimized Industrial Synthesis of Bambuterol Hydrochloride via Epoxide Ring-Opening Strategy
The Limitations of Conventional Methods vs. The Novel Approach
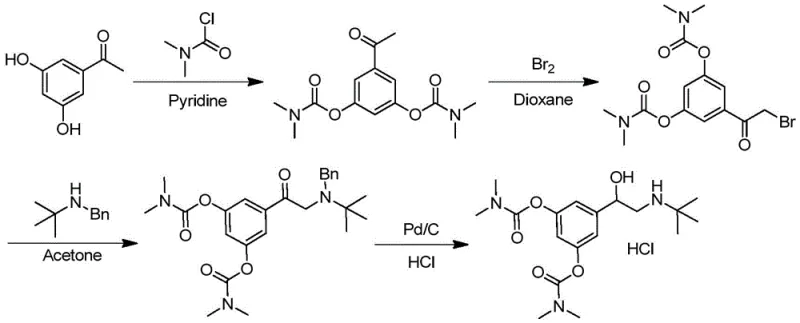
The Limitations of Conventional Methods
Historically, the industrial production of Bambuterol Hydrochloride has been plagued by significant safety and efficiency bottlenecks inherent in legacy synthetic routes. Traditional methodologies, such as those disclosed in patent EP43807, typically rely on a starting material of 3,5-dihydroxyacetophenone, necessitating a hazardous bromination step to introduce the requisite side-chain functionality. This reliance on elemental bromine introduces severe operational challenges, including high corrosivity, toxic vapor emissions, and the potential formation of genotoxic impurities that are difficult to purge from the final API. Furthermore, these conventional pathways invariably require a palladium-on-carbon (Pd/C) catalyzed hydrogenolysis step to remove benzyl protecting groups, which mandates the use of high-pressure reactors and introduces the risk of heavy metal contamination. The cumulative effect of these complex transformations results in a protracted four-to-five-step sequence with a dismal overall yield of approximately 20%, rendering the process economically inefficient and environmentally burdensome for large-scale manufacturing.
The Novel Approach
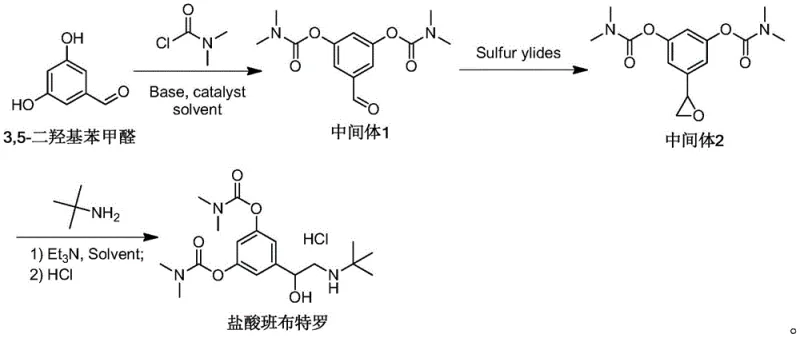
In stark contrast to these archaic methods, the innovative process detailed in patent CN109942462B presents a paradigm shift by utilizing a concise three-step synthesis originating from 3,5-dihydroxybenzaldehyde. This modern approach strategically bypasses the need for halogenation entirely, instead employing a sulfur ylide-mediated epoxidation to construct the critical ethanolamine backbone. By shifting the synthetic logic from a substitution-based strategy to an addition-based strategy via an epoxide intermediate, the process achieves remarkable atom economy and operational simplicity. The elimination of high-pressure hydrogenation and toxic bromine reagents not only drastically reduces the environmental footprint but also simplifies the equipment requirements, allowing for production in standard glass-lined or stainless steel reactors without specialized high-pressure capabilities. This streamlined architecture facilitates a substantial improvement in total yield, reaching nearly 60% in optimized examples, while simultaneously ensuring a finished product purity exceeding 99.5% through fewer purification cycles.
Mechanistic Insights into Sulfur Ylide Epoxidation and Ring Opening
The cornerstone of this advanced synthetic route is the utilization of the Corey-Chaykovsky reaction, where a sulfur ylide reagent acts as a nucleophile to convert the aldehyde functionality of Intermediate 1 into an oxirane (epoxide) ring in Intermediate 2. Mechanistically, this transformation involves the generation of a reactive sulfonium or sulfoxonium ylide species in situ using a strong base such as potassium hydroxide or sodium hydride in a polar aprotic solvent like acetonitrile. The nucleophilic carbon of the ylide attacks the electrophilic carbonyl carbon of the bis-carbamate aldehyde, forming a betaine intermediate which subsequently undergoes intramolecular nucleophilic displacement to expel the sulfide leaving group and close the three-membered epoxide ring. This specific mechanistic pathway is highly advantageous because it proceeds under mild thermal conditions (50-60°C) and exhibits excellent chemoselectivity, leaving the sensitive carbamate ester groups intact while efficiently constructing the sterically congested carbon framework required for the beta-agonist activity.
Following the formation of the epoxide, the final step involves a regioselective ring-opening reaction with tert-butylamine, which serves as both the nitrogen source and the nucleophile. In this acid-base mediated process, the lone pair of the tert-butylamine attacks the less hindered benzylic carbon of the epoxide ring, driven by the electronic activation provided by the adjacent aromatic system and the basic conditions maintained by triethylamine. This nucleophilic attack cleaves the strained C-O bond of the epoxide, establishing the critical beta-amino alcohol motif characteristic of adrenergic agonists. The reaction is thermodynamically favorable due to the relief of ring strain in the epoxide, and the use of excess amine ensures complete conversion while suppressing potential polymerization side reactions. The subsequent treatment with hydrochloric acid allows for the precise isolation of the hydrochloride salt, leveraging the differential solubility of the ionic species in organic solvents to achieve the high crystalline purity observed in the patent data.
How to Synthesize Bambuterol Hydrochloride Efficiently
The execution of this synthesis requires precise control over stoichiometry and reaction parameters to maximize the yield of the key epoxide intermediate. The process begins with the protection of the phenolic hydroxyl groups using N,N-dimethylcarbamoyl chloride, followed by the critical epoxidation step where moisture control is paramount to prevent ylide decomposition. Finally, the ring-opening step demands careful temperature management during reflux to ensure complete amination without degrading the carbamate moieties. While the general chemical principles are outlined above, the specific operational details regarding solvent volumes, addition rates, and work-up procedures are critical for reproducibility.
- Carbamoylation of 3,5-dihydroxybenzaldehyde with N,N-dimethylcarbamoyl chloride in the presence of a base like triethylamine to form the bis-carbamate aldehyde intermediate.
- Epoxidation of the aldehyde intermediate using a sulfur ylide reagent (e.g., trimethylsulfonium iodide) and a strong base to generate the oxirane ring.
- Nucleophilic ring-opening of the epoxide with tert-butylamine followed by salt formation with hydrochloric acid to yield the final Bambuterol Hydrochloride API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits regarding cost structure and logistical reliability. By fundamentally redesigning the molecular construction to avoid precious metal catalysts and hazardous halogens, the process decouples production costs from the volatile market prices of palladium and the stringent regulatory costs associated with handling controlled toxic substances. The removal of the hydrogenation step eliminates the need for specialized high-pressure vessels, thereby expanding the pool of eligible contract manufacturing organizations (CMOs) capable of producing this API and enhancing supply chain resilience against facility-specific bottlenecks. Furthermore, the reduction in reaction steps from five to three inherently shortens the manufacturing cycle time, allowing for faster inventory turnover and more responsive fulfillment of market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the elimination of expensive input materials and downstream processing costs. By removing the requirement for palladium on carbon, manufacturers avoid the significant capital expenditure associated with purchasing noble metals and the operational expense of recovering or disposing of spent catalyst. Additionally, the avoidance of bromine removes the need for specialized corrosion-resistant equipment and extensive scrubbing systems for toxic off-gases, leading to lower CAPEX and OPEX. The higher overall yield of approximately 60% compared to the historical 20% means that significantly less raw material is required to produce the same amount of finished API, directly improving the gross margin profile for the final product.
- Enhanced Supply Chain Reliability: From a logistics perspective, this route utilizes commodity chemicals such as 3,5-dihydroxybenzaldehyde, ethyl acetate, and tert-butylamine, which are readily available from multiple global suppliers, mitigating the risk of single-source dependency. The absence of high-pressure hydrogenation steps means that production is not limited to facilities with specific safety certifications for high-pressure gas handling, thereby broadening the geographical options for manufacturing sites. This flexibility is crucial for maintaining business continuity in the face of regional disruptions or regulatory changes, ensuring a steady flow of high-purity intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently green and scalable, aligning with modern ESG (Environmental, Social, and Governance) mandates that are increasingly influencing procurement decisions. The replacement of toxic reagents with safer alternatives reduces the volume of hazardous waste generated per kilogram of product, simplifying waste disposal compliance and lowering environmental fees. The mild reaction conditions and use of standard solvents facilitate straightforward scale-up from pilot plants to multi-ton commercial reactors without the need for complex engineering modifications, ensuring that supply can be rapidly ramped up to meet commercial launch timelines or unexpected demand surges.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering a clear view of the process capabilities.
Q: How does the new epoxide route improve safety compared to traditional bromination methods?
A: The novel process eliminates the use of elemental bromine, which is highly corrosive and toxic, and avoids the generation of genotoxic impurities often associated with alpha-bromination. Furthermore, it removes the requirement for high-pressure hydrogenation with Pd/C, significantly reducing operational safety risks and explosion hazards in the manufacturing plant.
Q: What is the impact of removing Palladium catalysts on the cost of goods?
A: By replacing the palladium-catalyzed hydrogenolysis step with a direct nucleophilic substitution, the process eliminates the cost of expensive noble metal catalysts and the subsequent rigorous purification steps required to remove trace heavy metals to ppm levels. This simplification directly lowers raw material costs and reduces waste treatment expenses related to heavy metal disposal.
Q: Can this synthesis route be scaled for commercial production?
A: Yes, the route is specifically designed for industrial scalability. It utilizes common organic solvents like ethyl acetate and acetonitrile, operates at atmospheric pressure, and avoids cryogenic conditions. The high yields reported in the patent examples (up to 90% for intermediates) demonstrate robust process control suitable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bambuterol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a more efficient synthetic route requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this epoxide-based synthesis, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest pharmacopeial standards for respiratory therapeutics.
We invite pharmaceutical developers and procurement leaders to engage with our technical team to explore how this optimized route can enhance your supply chain economics. By requesting a Customized Cost-Saving Analysis, you can quantify the potential reductions in COGS specific to your volume requirements. We encourage you to contact our technical procurement team today to obtain specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your project timelines.
