Advanced Palladium-Catalyzed Synthesis of Dibenzoxepin Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Dibenzoxepin Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds, particularly those serving as critical intermediates for antiallergic medications. Patent CN101151256B introduces a groundbreaking methodology for the production of 11-alkylidene dibenzo[b,e]oxepin derivatives, a structural class exemplified by potent antihistamines. This technical disclosure outlines a streamlined three-step sequence that overcomes significant stereochemical challenges inherent in previous generations of synthesis. By leveraging modern transition metal catalysis, specifically palladium-mediated cross-coupling and cyclization, the process ensures high selectivity for the biologically active Z-isomer. For R&D directors and procurement specialists, understanding this route is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with consistent batch-to-batch reproducibility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the dibenzoxepin core relied heavily on classical carbon-carbon bond-forming reactions such as the Wittig reaction or Grignard additions to ketone precursors. While chemically feasible, these traditional approaches suffer from severe drawbacks regarding stereochemical control. The formation of the exocyclic double bond at the 11-position frequently results in a thermodynamic mixture of E and Z geometric isomers. Separating these isomers is notoriously difficult and costly, often requiring extensive chromatographic purification or repeated recrystallization, which drastically reduces the overall process yield. Furthermore, the preparation of the requisite ketone intermediates can involve hazardous reagents and multi-step sequences that are not ideal for large-scale manufacturing environments where safety and efficiency are paramount.
The Novel Approach
In stark contrast, the methodology described in CN101151256B utilizes a strategic halogenation-coupling-cyclization cascade that bypasses the stereochemical pitfalls of olefination. The process begins with the selective iodination of a benzyl phenyl ether derivative, followed by a Sonogashira coupling with a terminal alkyne. The key innovation lies in the final intramolecular cyclization step, which constructs the seven-membered oxepin ring while simultaneously establishing the double bond geometry with high fidelity. 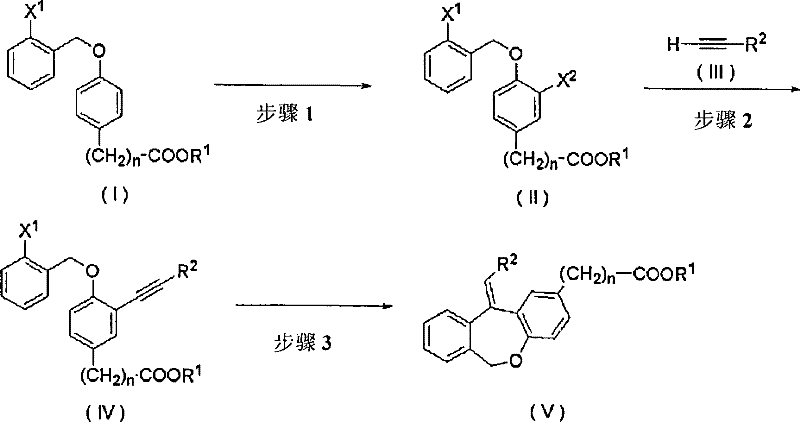 This route not only simplifies the synthetic tree but also utilizes widely available starting materials, making it a superior choice for cost reduction in pharmaceutical intermediate manufacturing. The ability to directly access the desired geometric isomer eliminates the need for expensive separation technologies, thereby enhancing the economic viability of the entire production line.
This route not only simplifies the synthetic tree but also utilizes widely available starting materials, making it a superior choice for cost reduction in pharmaceutical intermediate manufacturing. The ability to directly access the desired geometric isomer eliminates the need for expensive separation technologies, thereby enhancing the economic viability of the entire production line.
Mechanistic Insights into Palladium-Catalyzed Cyclization
The heart of this synthetic strategy is the palladium-catalyzed intramolecular cyclization of the alkyne intermediate. This transformation likely proceeds through an oxidative addition of the aryl halide moiety to the palladium(0) species, followed by coordination and insertion of the alkyne. The presence of specific phosphine ligands, such as tri(o-tolyl)phosphine or tricyclohexylphosphine, plays a critical role in stabilizing the active catalytic species and directing the regioselectivity of the ring closure. The use of a hydrogen source, typically formic acid or its salts in the presence of a base like piperidine, facilitates a reductive elimination or hydride transfer mechanism that finalizes the formation of the alkene. This mechanistic pathway is highly tunable, allowing chemists to optimize reaction conditions to favor the Z-configuration exclusively.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or thermal cyclizations. The mild conditions employed, typically ranging from 60°C to 100°C in polar aprotic solvents like DMF or acetonitrile, minimize the formation of degradation products or polymerization byproducts often seen with alkynes. Furthermore, the specificity of the palladium catalyst ensures that side reactions at other functional groups, such as esters or amines present on the side chains, are kept to a minimum. This high level of chemoselectivity is crucial for producing high-purity OLED material or pharmaceutical intermediates where trace metal residues and organic impurities must be strictly controlled to meet regulatory standards for downstream drug substance manufacturing.
How to Synthesize 11-Alkylidene Dibenzo[b,e]oxepin Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production. It involves preparing a halogenated ether precursor, coupling it with a functionalized alkyne, and then closing the ring under reductive conditions. Each step is optimized for yield and purity, utilizing standard workup procedures like extraction and column chromatography. The versatility of the method allows for various substituents on the aromatic rings and the alkyl side chain, enabling the production of a diverse library of analogues.
- Halogenation of benzyl phenyl ether derivatives using iodine and silver sulfate to introduce an iodine atom ortho to the ether linkage.
- Sonogashira coupling reaction between the halogenated intermediate and a terminal alkyne compound using a palladium catalyst and copper iodide.
- Intramolecular cyclization of the alkyne intermediate using a palladium catalyst, phosphine ligand, and a hydrogen source to form the dibenzoxepin ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates into tangible operational benefits beyond mere chemical elegance. The primary advantage lies in the substantial cost savings achieved through process intensification. By eliminating the need for difficult isomer separations, the process reduces solvent consumption, waste generation, and processing time. This efficiency gain directly impacts the cost of goods sold (COGS), allowing for more competitive pricing in the global market for fine chemical intermediates. Additionally, the reliance on robust palladium catalysis rather than stoichiometric organometallic reagents enhances the safety profile of the manufacturing process, reducing the risks associated with handling pyrophoric or highly reactive materials.
- Cost Reduction in Manufacturing: The streamlined nature of this three-step sequence significantly lowers the operational burden compared to legacy methods. By avoiding the low-yielding Wittig olefination and the subsequent purification nightmares, manufacturers can achieve higher throughput with fewer unit operations. The use of catalytic amounts of palladium, which can potentially be recovered and recycled, further mitigates the cost impact of precious metals. This logical deduction of cost efficiency makes the process highly attractive for long-term supply contracts where price stability is a key negotiation point.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as substituted phenols, benzyl halides, and terminal alkynes, are commodity chemicals available from multiple global sources. This diversity in the supply base reduces the risk of single-source bottlenecks that can plague more exotic synthetic routes. Furthermore, the reaction conditions are not overly sensitive to minor fluctuations in temperature or moisture, ensuring consistent production schedules. This reliability is essential for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API manufacturers receive their materials on time to meet their own production targets.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-kilogram scales in the patent examples. The solvents used, such as methanol, ethyl acetate, and DMF, are standard industrial solvents with established recovery and recycling protocols. This aligns well with modern environmental, health, and safety (EHS) regulations, facilitating easier permitting and compliance audits. The ability to scale up complex pharmaceutical intermediates without encountering exothermic runaways or hazardous byproducts ensures a continuous and safe supply chain, which is a critical metric for supply chain heads evaluating potential partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthetic pathway. They are derived from the specific experimental conditions and scope defined in the patent documentation. Understanding these nuances helps R&D teams assess the feasibility of adapting this chemistry for their specific target molecules.
Q: How does this process improve stereochemical control compared to traditional Wittig reactions?
A: Traditional Wittig or Grignard reactions often yield mixtures of geometric isomers that are difficult to separate. This patented method utilizes a specific intramolecular cyclization sequence that inherently favors the formation of the desired Z-isomer, significantly simplifying purification and improving overall yield.
Q: What specific catalysts are required for the cyclization step?
A: The cyclization step typically employs a palladium catalyst such as palladium acetate or palladium chloride in combination with phosphine ligands like tri(o-tolyl)phosphine. A hydrogen source, such as formic acid or ammonium formate, is also essential to facilitate the reductive cyclization.
Q: Can this method be scaled for industrial production of antiallergic agents?
A: Yes, the process is designed for scalability. It uses commercially available starting materials and standard transition metal catalysis conditions that are well-suited for kilogram-to-ton scale manufacturing, avoiding the harsh conditions and low yields associated with older synthetic routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 11-Alkylidene Dibenzo[b,e]oxepin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation antiallergic therapies. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards. Our commitment to quality assurance means that you can rely on us for the consistent supply of complex heterocyclic building blocks essential for your drug development pipeline.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain partner dedicated to innovation, quality, and long-term value creation in the pharmaceutical sector.
