Advanced Palladium-Catalyzed Synthesis of Chiral Disubstituted Indolinone Intermediates for Commercial Drug Development
Introduction to Next-Generation Indolinone Synthesis
The pharmaceutical industry continuously seeks efficient pathways to construct complex nitrogen-containing heterocyclic frameworks, which serve as critical scaffolds in numerous bioactive natural alkaloids and drug candidates. Patent CN115197199A introduces a groundbreaking methodology for synthesizing aromatic amine compounds containing a disubstituted indolinone skeleton, addressing long-standing challenges in stereoselective organic synthesis. This innovation leverages a sophisticated palladium-catalyzed asymmetric allylic alkylation strategy, utilizing vinylbenzoxazinones as versatile building blocks and 3-pyrrolyl oxindoles as nucleophiles. The significance of this technology lies in its ability to generate quaternary carbon chiral centers with exceptional precision under remarkably mild conditions. For research and development teams focused on accelerating drug discovery pipelines, this protocol offers a reliable route to access structurally diverse libraries of high-purity intermediates that were previously difficult to obtain with such high levels of stereocontrol.
Furthermore, the operational simplicity of this method represents a paradigm shift from traditional multi-step syntheses that often suffer from low atom economy and cumbersome purification processes. By employing a chiral oxamido phosphine ligand in conjunction with a tris(dibenzylideneacetone)dipalladium-chloroform adduct, the reaction achieves outstanding chemical selectivity and enantioselectivity without the need for extreme temperatures or pressures. This technological advancement not only streamlines the laboratory workflow but also lays a solid foundation for industrial translation, making it an attractive option for organizations aiming to secure a competitive edge in the synthesis of complex pharmaceutical intermediates. The robustness of this catalytic system ensures consistent quality, which is paramount for meeting the stringent regulatory standards required in active pharmaceutical ingredient manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of disubstituted indolinone skeletons has relied on cycloaddition reactions or less selective alkylation protocols that often impose significant limitations on process efficiency and product purity. Prior art, such as the iridium/Brønsted acid synergistic catalysis reported in 2019 or the palladium/chiral phosphonamide systems from 2021, while effective, frequently necessitate rigorous reaction conditions that can degrade sensitive functional groups or require extensive optimization for different substrates. These conventional approaches often struggle to maintain high linear selectivity during the allylic alkylation process, leading to mixtures of regioisomers that complicate downstream purification and reduce overall yield. Additionally, many existing methods involve the use of protected substrates, such as N-Ts protected vinylbenzoxazinones, which introduce extra synthetic steps for protection and deprotection, thereby increasing material costs and waste generation. The inability to consistently achieve high enantiomeric excess across a broad range of substrates further restricts the utility of these older methodologies in the rapid development of chiral drug candidates.
The Novel Approach
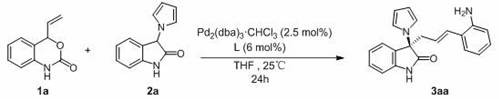
In stark contrast, the novel approach detailed in the patent utilizes a highly efficient palladium catalytic system that enables linear selective allylic alkylation directly between vinylbenzoxazinone and 3-pyrrolyl oxindole. This method eliminates the need for protecting groups on the nitrogen atom, significantly simplifying the synthetic route and reducing the number of unit operations required. The reaction proceeds smoothly at room temperature (25°C) in common organic solvents like tetrahydrofuran, demonstrating remarkable tolerance to various substituents including halogens and electron-withdrawing groups. As illustrated in the optimized reaction scheme, the use of a specific chiral oxamido phosphine ligand ensures that the formation of the quaternary carbon center occurs with superior stereocontrol, routinely achieving enantiomeric excess values exceeding 99%. This level of precision minimizes the formation of unwanted stereoisomers, thereby enhancing the overall purity of the final product and reducing the burden on purification resources. The simplicity of the post-reaction workup, which involves standard concentration and column chromatography, further underscores the practical advantages of this new methodology for both laboratory and pilot-scale applications.
Mechanistic Insights into Pd-Catalyzed Asymmetric Allylic Alkylation
The core of this synthetic breakthrough lies in the intricate interplay between the palladium catalyst and the chiral oxamido phosphine ligand, which creates a highly organized transition state favoring the formation of a single enantiomer. The mechanism initiates with the oxidative addition of the palladium(0) species to the vinylbenzoxazinone, generating a pi-allyl palladium intermediate that is stabilized by the chiral environment provided by the ligand. This chiral pocket effectively discriminates between the two faces of the allyl system, directing the nucleophilic attack of the 3-pyrrolyl oxindole to occur exclusively at the linear position with high facial selectivity. The unique electronic and steric properties of the oxamido phosphine ligand are crucial in suppressing branched alkylation pathways, ensuring that the reaction proceeds with excellent linear selectivity. This mechanistic control is vital for constructing the specific disubstituted indolinone architecture required for biological activity, as even minor deviations in regiochemistry can render the molecule inactive or toxic.
From an impurity control perspective, the high stereoselectivity of this catalytic system inherently limits the generation of diastereomeric and enantiomeric impurities, which are often the most challenging contaminants to remove in chiral synthesis. The mild reaction conditions prevent thermal degradation of the sensitive indolinone and pyrrole moieties, preserving the integrity of the molecular scaffold throughout the transformation. Furthermore, the compatibility of the catalyst system with a wide array of functional groups means that diverse analogs can be synthesized without the need for orthogonal protection strategies, which often introduce additional impurity profiles. The robustness of the catalytic cycle allows for low catalyst loadings while maintaining high turnover numbers, which not only reduces the residual metal content in the final product but also aligns with green chemistry principles by minimizing heavy metal waste. This deep understanding of the reaction mechanism allows process chemists to confidently predict outcomes and troubleshoot potential issues during scale-up, ensuring a reliable supply of high-quality intermediates.
How to Synthesize Chiral Disubstituted Indolinone Efficiently
To implement this advanced synthesis in a laboratory or pilot plant setting, operators must adhere to strict protocols regarding reagent quality and atmospheric control to maximize yield and enantioselectivity. The process begins with the preparation of the active catalytic species under an inert nitrogen atmosphere to prevent oxidation of the palladium center, followed by the sequential addition of substrates to maintain optimal concentration gradients. Detailed standardized operating procedures are essential to replicate the high performance observed in the patent examples, particularly concerning the stoichiometry of the vinylbenzoxazinone which is used in slight excess to drive the reaction to completion. The following guide outlines the critical operational parameters derived from the patented methodology, ensuring that technical teams can reproduce the exceptional results reported in the literature.
- Prepare the catalytic system by mixing a chiral oxamido phosphine ligand (6 mol%) and tris(dibenzylideneacetone)dipalladium-chloroform adduct (2.5 mol%) in anhydrous THF under nitrogen atmosphere.
- Add vinylbenzoxazinone (1.5 equivalents) and 3-pyrrolyl oxindole (1.0 equivalent) sequentially to the reaction mixture at room temperature (25°C).
- Stir the reaction mixture for 24 hours to ensure complete conversion, then concentrate under reduced pressure and purify the crude product via flash column chromatography using petroleum ether and ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers tangible strategic benefits that extend beyond mere technical performance, directly impacting the bottom line and operational resilience. The reliance on industrially available and inexpensive raw materials, such as unsubstituted or simply substituted vinylbenzoxazinones and 3-pyrrolyl oxindoles, mitigates the risk of supply chain disruptions associated with exotic or custom-synthesized starting materials. This accessibility ensures a stable flow of inputs, allowing for consistent production scheduling and reducing the lead time for high-purity pharmaceutical intermediates. Moreover, the elimination of protection and deprotection steps significantly shortens the overall manufacturing cycle, enabling faster time-to-market for new drug candidates and providing a competitive advantage in the fast-paced pharmaceutical landscape. The simplified workflow also reduces the consumption of solvents and reagents, contributing to substantial cost savings in raw material procurement and waste disposal.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of low catalyst loadings, specifically 2.5 mol% for the palladium source and 6 mol% for the chiral ligand, which drastically reduces the cost per kilogram of the final product compared to methods requiring stoichiometric chiral auxiliaries. The mild reaction conditions eliminate the need for energy-intensive heating or cooling systems, resulting in lower utility costs and a smaller carbon footprint for the manufacturing facility. Additionally, the high yield and selectivity minimize the loss of valuable starting materials to side reactions, maximizing the return on investment for every batch produced. By streamlining the purification process to a single column chromatography step using common solvent systems, the method avoids the need for expensive preparative HPLC or complex crystallization sequences, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: The robustness of the reaction against variations in substrate electronics ensures that suppliers can source a wide range of commercially available derivatives without fearing batch-to-batch failures. This flexibility allows procurement teams to diversify their supplier base for raw materials, reducing dependency on single-source vendors and enhancing overall supply security. The stability of the catalytic system under ambient conditions simplifies logistics and storage requirements, as there is no need for specialized cold chain transport or handling of hazardous pyrophoric reagents. Consequently, this reliability translates into more predictable delivery schedules for downstream customers, fostering stronger long-term partnerships and trust within the global pharmaceutical supply network.
- Scalability and Environmental Compliance: The patent explicitly validates the scalability of this method through successful ten-fold scale-up experiments that maintained high yield and enantiomeric excess, demonstrating its readiness for commercial production. The use of tetrahydrofuran, a solvent with well-established recovery and recycling protocols, facilitates compliance with environmental regulations regarding volatile organic compound emissions. The absence of toxic by-products and the generation of minimal waste streams align with increasingly stringent global environmental standards, reducing the regulatory burden on manufacturing sites. This eco-friendly profile not only safeguards the company's reputation but also future-proofs the production process against evolving sustainability mandates, ensuring long-term operational continuity.

Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this palladium-catalyzed synthesis, providing clarity for stakeholders evaluating its integration into their existing workflows. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is critical for assessing the feasibility of adopting this technology for specific project requirements and for anticipating potential challenges during technology transfer.
Q: What are the key advantages of this synthesis method compared to existing technologies?
A: This method utilizes a novel palladium/chiral oxamido phosphine ligand system that operates under mild conditions (25°C) with exceptional enantioselectivity (>99% ee). Unlike prior art which often requires harsh conditions or complex protection groups, this approach offers a linear selective allylic alkylation with simple post-reaction treatment and no by-product generation.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the patent explicitly demonstrates successful scale-up experiments where raw material usage was increased tenfold while maintaining high yield and excellent enantiomeric excess. The use of industrially available, inexpensive raw materials and standard purification techniques like column chromatography supports robust commercial scalability.
Q: What is the substrate scope for this reaction?
A: The method exhibits broad substrate compatibility, tolerating various functional groups on both the vinylbenzoxazinone and the 3-pyrrolyl oxindole scaffolds. Substituents such as halogens (F, Cl, Br), alkyl groups (methyl), and electron-withdrawing groups (trifluoromethyl, nitro) are well-tolerated without compromising yield or stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Disubstituted Indolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing high-quality chiral intermediates to drive innovation in drug development, and we are uniquely positioned to support your needs with this cutting-edge synthesis technology. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of disubstituted indolinone intermediate meets the highest industry standards. Our commitment to excellence extends beyond mere compliance; we actively collaborate with our partners to optimize processes for maximum yield and minimal environmental impact, delivering value at every stage of the supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced palladium-catalyzed method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this more efficient synthetic route for your target molecules. We encourage you to contact us today to obtain specific COA data for our available intermediates and to receive comprehensive route feasibility assessments that will empower your R&D and supply chain strategies. Let us be your trusted partner in navigating the complexities of modern pharmaceutical synthesis and securing a reliable supply of critical building blocks for your next breakthrough therapy.
