Revolutionizing Amide Synthesis: Direct C-H Alkynylation for Scalable Pharmaceutical Manufacturing
Introduction to Breakthrough Amide Functionalization Technology
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more efficient and sustainable methodologies. Patent CN108640945B introduces a significant advancement in the field of amide compound preparation, specifically addressing the challenges associated with introducing alkynyl groups onto aromatic rings. This technology leverages a sophisticated transition metal-catalyzed strategy to achieve direct carbon-hydrogen (C-H) bond activation. Unlike traditional methods that often rely on pre-functionalized starting materials, this approach utilizes simple, readily available primary and secondary amides as direct substrates. The innovation lies in its ability to bypass the tedious installation and removal of auxiliary directing groups, which has historically been a bottleneck in the synthesis of complex bioactive molecules. By streamlining the synthetic route, this method offers a compelling value proposition for industrial applications where step economy and waste reduction are paramount.
For research and development teams focused on drug discovery, the ability to rapidly diversify amide scaffolds is crucial. The disclosed method provides a robust platform for generating novel chemical space around the amide core, a ubiquitous motif in medicinal chemistry. The process operates under relatively mild conditions compared to earlier generations of C-H activation technologies, utilizing a dual catalytic system that ensures high efficiency and selectivity. This patent represents a shift towards more atom-economical processes, aligning with the green chemistry principles that modern chemical manufacturing increasingly demands. The resulting ortho-alkynylated amides serve as versatile building blocks for further downstream transformations, including cyclization reactions to form complex heterocycles, thereby expanding the utility of this technology beyond simple functionalization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-alkynylated aryl amides has been fraught with synthetic inefficiencies. Traditional approaches, such as the Sonogashira coupling, typically require pre-halogenated substrates, necessitating additional synthetic steps to introduce the halogen handle prior to the coupling event. Furthermore, direct C-H activation strategies developed in the past often relied heavily on the presence of strong coordinating directing groups, such as pyridine or quinoline moieties, installed on the amide nitrogen. While effective for lab-scale synthesis, these groups are synthetically useless in the final target molecule and must be removed in a subsequent step. This install-remove sequence not only increases the overall step count and production time but also generates significant chemical waste, negatively impacting the environmental factor (E-factor) of the process. Additionally, many existing methods utilize expensive hypervalent iodine reagents or harsh reaction conditions that limit their scalability and economic viability for large-scale manufacturing.
The Novel Approach
The methodology described in CN108640945B overcomes these hurdles by enabling the direct alkynylation of ordinary amides without any extra guiding assistance. This represents a paradigm shift in how chemists approach late-stage functionalization of amide-containing drug candidates. The core of this innovation is a carefully optimized catalytic system that activates the ortho-C-H bond of the amide substrate directly. 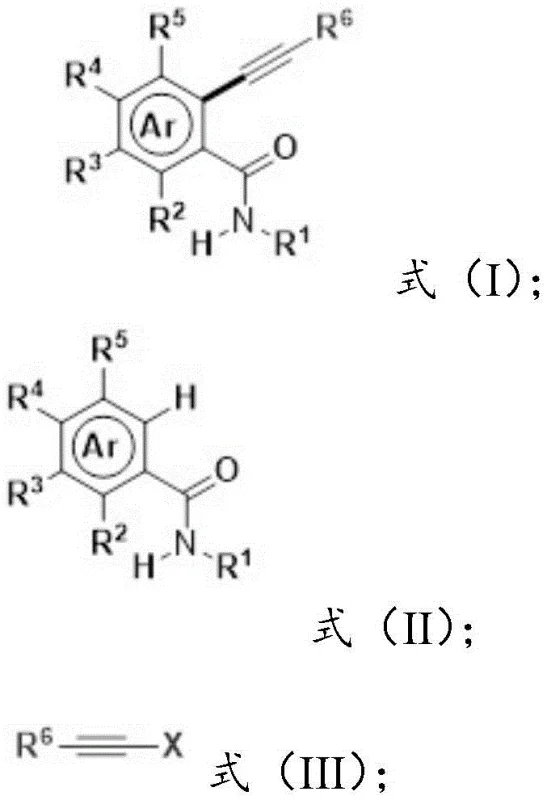 As illustrated in the general reaction scheme, the process couples an amide substrate with an alkynyl source, which can be either an alkynyl bromide or a terminal alkyne. When terminal alkynes are used, the system elegantly incorporates an oxidant to drive the reaction, maintaining high atom economy. This elimination of pre-functionalization steps significantly simplifies the supply chain for raw materials, as commodity chemicals can be used directly. The operational simplicity, combined with the broad substrate tolerance, makes this approach highly attractive for process chemistry teams looking to optimize manufacturing routes for active pharmaceutical ingredients (APIs).
As illustrated in the general reaction scheme, the process couples an amide substrate with an alkynyl source, which can be either an alkynyl bromide or a terminal alkyne. When terminal alkynes are used, the system elegantly incorporates an oxidant to drive the reaction, maintaining high atom economy. This elimination of pre-functionalization steps significantly simplifies the supply chain for raw materials, as commodity chemicals can be used directly. The operational simplicity, combined with the broad substrate tolerance, makes this approach highly attractive for process chemistry teams looking to optimize manufacturing routes for active pharmaceutical ingredients (APIs).
Mechanistic Insights into Ru/Ir Dual-Catalyzed C-H Activation
The success of this transformation hinges on a synergistic dual-catalyst system involving Ruthenium and Iridium complexes. Specifically, the patent details the use of dichloro(p-cymene)ruthenium dimer or dichloro(pentamethylcyclopentadienyl)iridium dimer as the primary catalysts, supported by silver salts such as silver bis(trifluoromethanesulfonyl)imide or silver hexafluoroantimonate. These silver species play a critical role not just as oxidants but as promoters for ligand exchange, facilitating the generation of the active cationic metal species required for C-H bond cleavage. The presence of cesium acetate as a base is also crucial, assisting in the concerted metalation-deprotonation (CMD) pathway that is characteristic of many Ru and Ir-catalyzed C-H activations. This mechanistic pathway allows for the selective activation of the ortho-C-H bond directed by the weakly coordinating amide oxygen, a feat that was previously difficult to achieve with high fidelity.
From an impurity control perspective, the mechanism offers distinct advantages. The high regioselectivity observed ensures that the alkynyl group is installed exclusively at the ortho-position relative to the amide carbonyl, avoiding the formation of meta- or para-substituted isomers that are common in electrophilic aromatic substitutions. 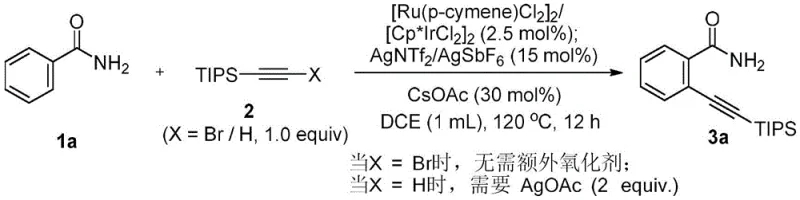 Furthermore, the catalytic system demonstrates remarkable chemoselectivity. It tolerates a wide range of functional groups that are typically sensitive to organometallic conditions, such as ketones, esters, and even free hydroxyl groups in certain contexts. For instance, the patent highlights the successful alkynylation of substrates containing acetyl groups without affecting the ketone functionality, which is a common challenge in cross-coupling chemistry. This selectivity minimizes the formation of side products, thereby simplifying the downstream purification process and ensuring a cleaner crude product profile, which is essential for meeting the stringent purity specifications required in pharmaceutical manufacturing.
Furthermore, the catalytic system demonstrates remarkable chemoselectivity. It tolerates a wide range of functional groups that are typically sensitive to organometallic conditions, such as ketones, esters, and even free hydroxyl groups in certain contexts. For instance, the patent highlights the successful alkynylation of substrates containing acetyl groups without affecting the ketone functionality, which is a common challenge in cross-coupling chemistry. This selectivity minimizes the formation of side products, thereby simplifying the downstream purification process and ensuring a cleaner crude product profile, which is essential for meeting the stringent purity specifications required in pharmaceutical manufacturing.
How to Synthesize Ortho-Alkynylated Amides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The patent outlines a standardized protocol that balances reaction efficiency with safety and ease of handling. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically nitrogen, to prevent catalyst deactivation by oxygen. The choice of solvent is critical, with 1,2-dichloroethane (DCE) identified as the optimal medium for solubilizing both the organic substrates and the metal catalysts while maintaining thermal stability at the required reaction temperatures. The detailed标准化 synthesis steps provided in the guide below reflect the optimized conditions found in the patent examples, ensuring that technical teams can replicate the high performance reported.
- Combine the amide substrate (Formula II) and alkynyl source (Formula III, either bromide or terminal alkyne) in an inert solvent like 1,2-dichloroethane.
- Add the dual catalyst system comprising [Ru(p-cymene)Cl2]2 or [Cp*IrCl2]2 along with silver salts (AgNTf2 or AgSbF6) and cesium acetate as the base.
- Heat the reaction mixture to 80-120°C under nitrogen atmosphere for 12-16 hours, adding silver acetate oxidant if using terminal alkynes, then purify via chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the raw material portfolio. By eliminating the need for custom-synthesized, directing-group-modified starting materials, manufacturers can source cheaper, commodity-grade amides and alkynes. This reduction in material complexity directly correlates to a reduction in procurement lead times and a decrease in the risk of supply chain disruptions associated with specialized intermediates. Moreover, the shortened synthetic route—removing the steps for directing group installation and removal—means fewer unit operations, less solvent consumption, and reduced energy usage per kilogram of product. These factors collectively contribute to a significantly lower cost of goods sold (COGS), enhancing the overall competitiveness of the final pharmaceutical product in the market.
- Cost Reduction in Manufacturing: The economic impact of this method is profound due to the elimination of auxiliary reagents and steps. Traditional routes often incur high costs associated with the synthesis and subsequent cleavage of directing groups, which adds no value to the final molecule. By bypassing these steps, the process inherently reduces labor, equipment usage, and waste disposal costs. Additionally, the catalyst loading is kept relatively low (in the range of 1-5 mol%), which helps mitigate the cost impact of precious metals like Iridium and Ruthenium. The use of terminal alkynes, which are generally less expensive than alkynyl bromides, further drives down material costs when the oxidative variant of the reaction is employed. This lean manufacturing approach aligns perfectly with initiatives to reduce the environmental footprint and financial overhead of API production.
- Enhanced Supply Chain Reliability: Reliability in the supply of key intermediates is critical for maintaining continuous pharmaceutical production. This method enhances reliability by relying on robust, commercially available starting materials rather than fragile, multi-step intermediates. The broad substrate scope means that a single manufacturing platform can be adapted to produce a wide variety of analogues without significant retooling or process redevelopment. This flexibility allows supply chain managers to respond quickly to changes in demand or to switch between different product candidates during clinical development phases. The stability of the reaction conditions also reduces the likelihood of batch failures, ensuring a consistent and predictable output of high-quality intermediates that can be relied upon for downstream processing.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant often reveals hidden challenges, but this methodology is designed with scalability in mind. The reaction conditions (temperatures around 100-120°C and standard pressures) are compatible with existing stainless steel reactor infrastructure, avoiding the need for specialized high-pressure or cryogenic equipment. From an environmental compliance standpoint, the high atom economy and reduced waste generation simplify the handling of effluent streams. The avoidance of toxic tin or palladium residues often associated with traditional coupling methods eases the burden on purification units and helps meet strict regulatory limits on heavy metal impurities in drug substances. This makes the technology not only economically viable but also sustainable and compliant with increasingly rigorous global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amide synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows. The clarity provided here aims to bridge the gap between academic innovation and industrial application, ensuring that stakeholders have a comprehensive understanding of the process capabilities and limitations.
Q: Does this method require pre-installation of directing groups on the amide?
A: No, a key advantage of patent CN108640945B is that it utilizes ordinary primary and secondary amides directly without the need for installing and subsequently removing extra directing groups like pyridine or quinoline.
Q: What types of alkynyl sources are compatible with this synthesis?
A: The method is versatile and accepts both alkynyl bromides and terminal alkynes. When using terminal alkynes, an additional oxidant such as silver acetate is required to facilitate the oxidative coupling.
Q: Is the reaction regioselective for complex substrates?
A: Yes, the process demonstrates excellent regioselectivity, specifically targeting the ortho-position of the aryl amide ring even in the presence of other functional groups like ketones or heterocyclic nitrogens.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modern drug synthesis. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate complex laboratory protocols like those in CN108640945B into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle sensitive organometallic chemistry safely and efficiently makes us the ideal partner for leveraging this novel amide functionalization technology.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your supply chain and reduce your overall development costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Whether you require specific COA data for quality assurance or detailed route feasibility assessments to validate the process for your target molecule, we are here to support your goals. Contact us today to discuss how we can accelerate your timeline and enhance the economic viability of your pharmaceutical projects through our advanced manufacturing capabilities.
