Advanced Carbazole Compound Manufacturing: Scalable Rhodium-Catalyzed Process for Global Pharmaceutical Supply Chains
The Chinese patent CN108658841B introduces a groundbreaking method for synthesizing carbazole-based compounds through a rhodium-catalyzed reaction between polysubstituted diarylboronic acids and azides. This innovative approach represents a significant advancement in heterocyclic compound synthesis, offering unprecedented substrate versatility and operational simplicity compared to conventional techniques. The method enables the efficient production of diverse carbazole derivatives with various substituents and heterocyclic moieties, addressing critical limitations in traditional synthesis pathways that required harsh reaction conditions and extensive pre-functionalization of substrates. This patent demonstrates remarkable potential for industrial-scale implementation across multiple sectors including pharmaceuticals, agrochemicals, and electronic materials manufacturing, where carbazole compounds serve as essential building blocks for advanced functional materials. The technology's broad applicability and operational efficiency position it as a transformative solution for manufacturers seeking reliable production of complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing carbazole compounds have been plagued by significant limitations that hinder their industrial applicability and economic viability. The conventional approaches typically require high-temperature conditions coupled with strong oxidizing agents, creating substantial safety concerns and increasing energy consumption during manufacturing processes. Furthermore, these methods often necessitate extensive pre-functionalization of substrates, adding multiple synthetic steps that reduce overall yield and increase production costs. The narrow substrate scope of existing techniques restricts the diversity of accessible carbazole derivatives, limiting their application in developing novel pharmaceutical agents and advanced materials. Additionally, many conventional processes demand strictly anhydrous and oxygen-free environments, requiring specialized equipment and increasing operational complexity. These constraints collectively result in lower production efficiency, higher manufacturing costs, and limited scalability for commercial applications in pharmaceutical and specialty chemical industries.
The Novel Approach
The patented rhodium-catalyzed methodology overcomes these longstanding challenges through an elegant one-pot reaction sequence that operates under remarkably mild conditions (-10°C to 120°C). This innovative approach eliminates the need for substrate pre-functionalization by directly utilizing readily available diarylboronic acids and azide precursors, significantly streamlining the synthetic pathway. The reaction demonstrates exceptional functional group tolerance, accommodating a wide range of substituents including alkyl, alkoxy, halogen, nitro, cyano, trifluoromethyl, and various heterocyclic moieties without requiring specialized protection-deprotection strategies. Crucially, the process does not demand anhydrous or oxygen-free conditions, substantially reducing equipment requirements and operational complexity while maintaining high yields (up to 90%). This methodology represents a paradigm shift in carbazole synthesis, offering manufacturers unprecedented flexibility to produce diverse compound libraries for drug discovery and materials science applications with significantly improved process economics.
Mechanistic Insights into Rhodium-Catalyzed Carbazole Synthesis
The rhodium-catalyzed synthesis proceeds through a sophisticated cascade mechanism that begins with oxidative addition of the rhodium catalyst to the azide group, followed by migratory insertion of the boronic acid moiety. This sequence generates a key rhodacycle intermediate that undergoes reductive elimination to form the crucial C-N bond that defines the carbazole core structure. The silver oxidant plays a critical role in regenerating the active rhodium species while facilitating the elimination of nitrogen gas as a benign byproduct. This mechanistic pathway operates with exceptional efficiency due to the synergistic interaction between the rhodium catalyst and silver co-catalyst, which maintains optimal oxidation states throughout the reaction cycle. The mild temperature range (75-85°C) prevents unwanted side reactions while ensuring sufficient kinetic energy for the transformation to proceed at a practical rate.
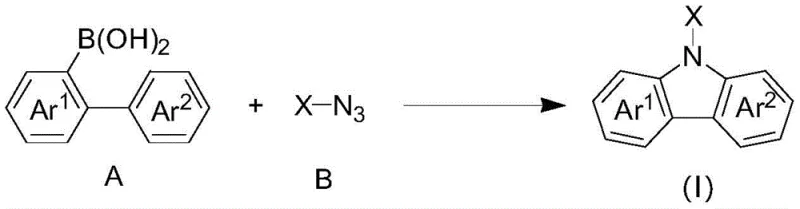
The remarkable substrate versatility stems from the mechanism's tolerance to electronic and steric variations across both coupling partners. The rhodium catalyst accommodates electron-donating and electron-withdrawing substituents on both the boronic acid and azide components without significant rate depression or selectivity issues. This broad functional group compatibility enables the synthesis of highly diverse carbazole derivatives that would be challenging or impossible to access through conventional methods. The mechanism also inherently minimizes impurity formation through its well-defined reaction pathway that avoids common side reactions such as protodeboronation or homocoupling that plague other transition metal-catalyzed processes.
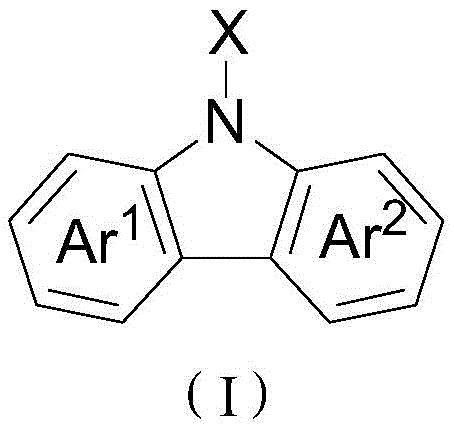
Impurity control is achieved through precise reaction parameter optimization and the inherent selectivity of the catalytic system. The use of pentamethylcyclopentadiene rhodium chloride dimer as catalyst ensures high regioselectivity during C-N bond formation, while the carefully controlled stoichiometry (1:1.0-1.5:1.0 molar ratio) prevents excess reagent accumulation that could lead to side products. The mild reaction conditions (-10°C to 120°C) minimize thermal degradation pathways that typically generate impurities in high-temperature processes. Post-reaction purification using column chromatography with petroleum ether/ethyl acetate (90-110:1) effectively separates any minor impurities from the desired product, consistently delivering high-purity carbazole compounds suitable for pharmaceutical applications where stringent quality standards apply.
How to Synthesize Carbazole Compounds Efficiently
This patented methodology represents a significant advancement in carbazole compound synthesis, offering manufacturers a streamlined approach that eliminates multiple processing steps required by conventional techniques while delivering superior product quality and yield consistency. The rhodium-catalyzed process provides exceptional flexibility in producing diverse carbazole derivatives with various substituent patterns essential for pharmaceutical development and electronic material applications. Detailed standardized synthesis steps are provided below to ensure successful implementation across different manufacturing scales.
- Prepare the reaction mixture by combining diarylboronic acid (0.18 mmol), rhodium catalyst (pentamethylcyclopentadiene rhodium chloride dimer at 0.0075 mmol), silver oxidant (silver acetate at 0.18 mmol), base (sodium carbonate at 0.225 mmol), azide precursor (0.15 mmol), and solvent (1,4-dioxane at 2 ml) under nitrogen atmosphere.
- Heat the reaction mixture to optimal temperature range (75°C to 85°C) and maintain for appropriate duration (typically 2-24 hours), monitoring reaction progress by TLC until completion is confirmed.
- Purify the product through solvent removal under reduced pressure followed by column chromatography using petroleum ether/ethyl acetate (90-110: 1 ratio), with optional recrystallization to achieve high-purity final product.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology directly addresses critical pain points in specialty chemical procurement by delivering a robust manufacturing solution that enhances supply chain resilience while optimizing production economics. The process eliminates dependency on complex multi-step syntheses that traditionally created bottlenecks in intermediate supply chains, providing procurement teams with greater confidence in material availability and quality consistency.
- Cost Reduction in Manufacturing: The elimination of pre-functionalization steps significantly reduces raw material costs while minimizing solvent usage and waste generation through its streamlined one-pot process. The ability to operate without specialized anhydrous or oxygen-free equipment substantially lowers capital investment requirements and maintenance costs associated with traditional synthesis methods.
- Enhanced Supply Chain Reliability: By utilizing readily available starting materials from multiple global suppliers, this methodology mitigates single-source dependency risks while ensuring consistent production capacity across varying demand scenarios. The process's robustness across different scales provides procurement teams with confidence in securing uninterrupted supply of critical intermediates.
- Scalability and Environmental Compliance: The methodology demonstrates seamless scalability from laboratory to commercial production volumes while maintaining consistent product quality parameters. Its reduced environmental footprint through minimized solvent consumption and waste generation aligns with increasingly stringent regulatory requirements while supporting corporate sustainability initiatives.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding the implementation and benefits of this patented carbazole synthesis methodology based on extensive technical evaluation of the process parameters and performance characteristics.
Q: How does this rhodium-catalyzed method improve upon conventional carbazole synthesis techniques?
A: This method eliminates the need for pre-functionalization of substrates and operates under milder conditions (-10°C to 120°C) compared to traditional high-temperature processes requiring harsh oxidants, while maintaining excellent functional group tolerance across diverse substituent patterns including alkyl, alkoxy, halogen, nitro, cyano, trifluoromethyl groups.
Q: What are the key advantages of this synthesis method for pharmaceutical manufacturing?
A: The process delivers high-purity carbazole compounds with exceptional versatility across diverse substituents without requiring anhydrous or oxygen-free conditions, enabling reliable production of complex intermediates essential for drug development while significantly reducing processing steps compared to conventional methods.
Q: How does this technology address supply chain challenges for specialty chemical procurement?
A: By utilizing readily available starting materials from multiple global sources and eliminating complex pre-treatment steps, this methodology ensures consistent production capacity from laboratory scale to commercial volumes (up to 100 MT annually), providing reliable supply chain continuity for critical pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbazole Compound Supplier
Our company leverages this patented technology to deliver exceptional value through our comprehensive CDMO capabilities, combining deep expertise in heterocyclic chemistry with state-of-the-art manufacturing infrastructure capable of scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications through our advanced analytical capabilities housed within rigorous QC labs, ensuring consistent product quality that meets or exceeds industry standards for pharmaceutical intermediates and specialty chemicals.
To explore how our specialized expertise can enhance your supply chain efficiency, we invite you to request a Customized Cost-Saving Analysis from our technical procurement team. They will provide detailed information including specific COA data and route feasibility assessments tailored to your unique requirements, enabling you to make informed decisions about integrating this advanced synthesis methodology into your manufacturing operations.
