Scalable Electrochemical Production of High-Purity 3-Arylselenyl-4-Aminocoumarin Derivatives
Scalable Electrochemical Production of High-Purity 3-Arylselenyl-4-Aminocoumarin Derivatives
The pharmaceutical and fine chemical industries are increasingly prioritizing sustainable manufacturing technologies that reduce environmental footprints while maintaining rigorous quality standards. A pivotal advancement in this domain is detailed in Chinese Patent CN112501642B, which discloses a novel electrochemical synthesis method for 3-arylselenyl-4-aminocoumarin compounds. These heterocyclic scaffolds are critical intermediates in the development of bioactive molecules, yet their traditional synthesis often relies on harsh chemical oxidants. This patented technology replaces stoichiometric oxidants with electricity, utilizing a catalytic amount of iodide salt in an undivided cell setup. By leveraging direct current to drive the oxidative coupling of 4-aminocoumarins with diaryl diselenides, the process achieves exceptional yields under mild conditions. For procurement and supply chain leaders, this represents a shift towards more reliable pharmaceutical intermediate supplier capabilities, where cost efficiency and environmental compliance are paramount. The elimination of heavy metal catalysts and excessive inorganic salts simplifies the impurity profile, ensuring high-purity pharmaceutical intermediates suitable for sensitive drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this electrochemical innovation, the synthesis of 3-arylselenyl-4-aminocoumarins faced significant hurdles regarding waste generation and operational complexity. As noted in the background art, previous methods, such as the visible-light promoted synthesis reported in 2018, relied heavily on stoichiometric amounts of inorganic oxidants like peroxodisulfate. Specifically, these legacy processes required two equivalents of oxidant to drive the C-H/C-Se bond radical coupling reaction. This dependency creates a dual burden: first, the sheer volume of inorganic salt waste increases disposal costs and environmental liability; second, the presence of these salts complicates the isolation and purification of the target molecule, often necessitating energy-intensive chromatographic steps. Furthermore, the use of chemical oxidants introduces safety risks associated with handling strong oxidizing agents on a large scale. For a reliable agrochemical intermediate supplier or pharma partner, these factors translate into higher production costs and longer lead times, making the conventional routes less attractive for commercial scale-up of complex polymer additives or drug precursors.
The Novel Approach
The electrochemical methodology described in CN112501642B fundamentally reengineers the reaction landscape by employing electrons as a traceless oxidant. In this streamlined process, the reaction mixture consists of the 4-aminocoumarin substrate, diaryl diselenide, and a catalytic quantity of iodide salt dissolved in dimethyl sulfoxide (DMSO). Upon applying a direct current across a reticulated vitreous carbon (RVC) anode and a platinum mesh cathode, the system generates the necessary oxidative potential in situ. This approach not only obviates the need for external chemical oxidants but also operates at room temperature, significantly reducing energy consumption. The result is a highly selective transformation that delivers yields exceeding 90% under optimized conditions. 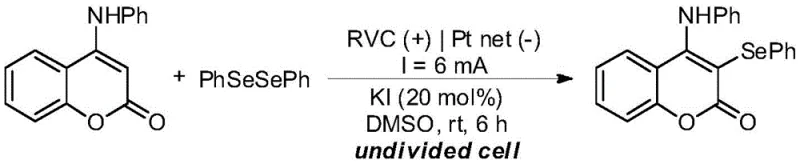 This simplicity allows for cost reduction in electronic chemical manufacturing and pharma sectors alike, as the workup procedure is drastically simplified to a water precipitation step, avoiding the need for complex extraction or column chromatography. The robustness of this method ensures consistent quality, a critical factor for maintaining supply chain continuity.
This simplicity allows for cost reduction in electronic chemical manufacturing and pharma sectors alike, as the workup procedure is drastically simplified to a water precipitation step, avoiding the need for complex extraction or column chromatography. The robustness of this method ensures consistent quality, a critical factor for maintaining supply chain continuity.
Mechanistic Insights into Iodide-Mediated Electrochemical Selenylation
Understanding the catalytic cycle is essential for R&D directors evaluating the feasibility of this technology for large-scale implementation. The mechanism initiates at the anode surface, where iodide ions (I⁻) from the catalytic salt undergo a two-electron oxidation to form electrophilic iodine species (I⁺). This activated iodine species then reacts selectively with the electron-rich 4-aminocoumarin substrate to generate a key 3-iodo-4-aminocoumarin intermediate (IM1). This electrophilic substitution is highly regioselective, ensuring that the iodine atom is installed precisely at the C3 position of the coumarin ring. Subsequently, this iodinated intermediate undergoes a nucleophilic substitution or coupling reaction with the diaryl diselenide. This step displaces the iodine moiety and forms the desired C-Se bond, releasing molecular iodine (I₂) as a byproduct. The elegance of the system lies in its closed-loop nature: the molecular iodine generated is reduced back to iodide ions at the platinum cathode, thereby regenerating the catalyst and completing the cycle. Simultaneously, protons in the solution are reduced at the cathode to evolve hydrogen gas, which safely vents from the system. 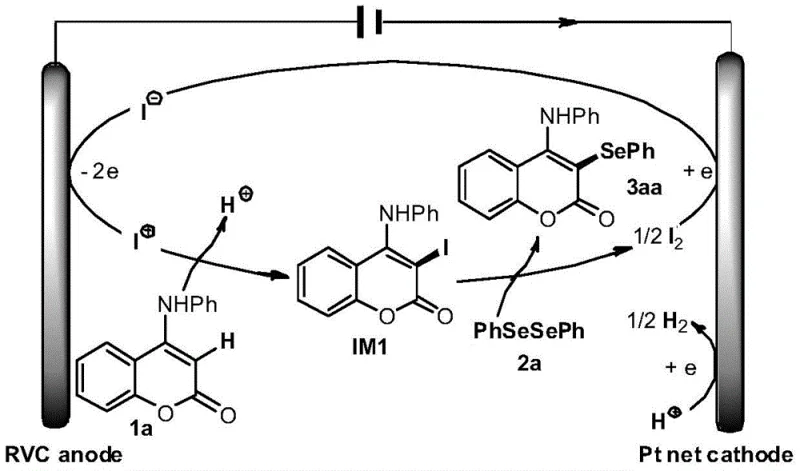 This continuous regeneration of the iodide catalyst means that only a sub-stoichiometric amount (20 mol%) is required, contrasting sharply with traditional methods that consume reagents irreversibly.
This continuous regeneration of the iodide catalyst means that only a sub-stoichiometric amount (20 mol%) is required, contrasting sharply with traditional methods that consume reagents irreversibly.
From an impurity control perspective, this mechanism offers distinct advantages. Because the oxidation potential is controlled electrically rather than chemically, over-oxidation of the sensitive selenium or amine functionalities is minimized. The patent data highlights excellent functional group tolerance, accommodating substrates with electron-withdrawing groups such as trifluoromethyl, cyano, and ester moieties without significant yield degradation. 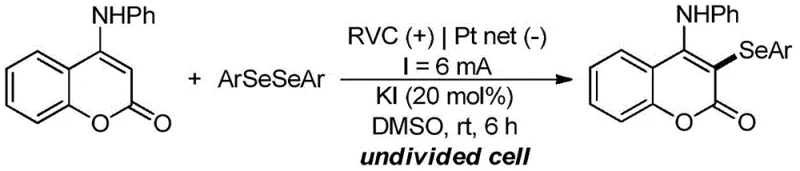 For instance, substrates containing para-fluoro, para-chloro, and para-cyano substituents on the diselenide ring all afforded products with yields ranging from 85% to 97%. This broad compatibility suggests that the electrochemical environment is mild enough to preserve delicate chemical bonds while being potent enough to drive the C-H activation. For quality assurance teams, this implies a cleaner crude reaction profile, reducing the burden on downstream purification units and ensuring that the final high-purity OLED material or API intermediate meets stringent regulatory specifications with minimal effort.
For instance, substrates containing para-fluoro, para-chloro, and para-cyano substituents on the diselenide ring all afforded products with yields ranging from 85% to 97%. This broad compatibility suggests that the electrochemical environment is mild enough to preserve delicate chemical bonds while being potent enough to drive the C-H activation. For quality assurance teams, this implies a cleaner crude reaction profile, reducing the burden on downstream purification units and ensuring that the final high-purity OLED material or API intermediate meets stringent regulatory specifications with minimal effort.
How to Synthesize 3-Arylselenyl-4-Aminocoumarin Efficiently
Implementing this electrochemical protocol requires precise control over reaction parameters to maximize efficiency and reproducibility. The standard procedure involves charging an undivided cell with the substrate, diselenide, and catalytic potassium iodide in DMSO, followed by electrolysis at a constant current of 6 mA for 6 hours. The choice of electrode materials is critical; the patent specifies that a combination of a mesh glassy carbon anode and a platinum mesh cathode provides the optimal balance of conductivity and stability. Deviating from these materials, such as using nickel or graphite rods, results in noticeably lower yields. The solvent system is equally vital, with DMSO proving superior to acetonitrile or ethanol due to its ability to dissolve both organic substrates and ionic species effectively. Detailed standardized synthesis steps for replicating this high-yield transformation are provided in the guide below, ensuring that technical teams can validate the process in their own pilot facilities.
- Prepare the electrolyte solution by dissolving 4-aminocoumarin, diaryl diselenide, and catalytic potassium iodide (20 mol%) in dimethyl sulfoxide (DMSO).
- Insert a reticulated vitreous carbon (RVC) anode and a platinum mesh cathode into the undivided cell and apply a constant direct current of 6 mA at room temperature.
- After 6 hours of electrolysis, precipitate the product by adding excess water, filter the crystals, and recycle the filtrate containing the iodide catalyst and solvent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this electrochemical methodology offers transformative benefits that extend beyond mere chemical curiosity. The primary value proposition lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for stoichiometric oxidants like peroxodisulfate, the process removes a major cost center and a source of hazardous waste. Furthermore, the ability to recycle the solvent and catalyst system creates a circular economy within the manufacturing plant, significantly lowering the total cost of ownership. This aligns perfectly with global trends towards green chemistry and sustainable manufacturing, enhancing the corporate social responsibility profile of the end product. The operational simplicity also translates to reduced downtime between batches, as the cleaning and setup procedures are less burdensome compared to traditional multi-step syntheses involving filtration of inorganic salts.
- Cost Reduction in Manufacturing: The economic impact of replacing chemical oxidants with electricity cannot be overstated. In traditional synthesis, the cost of purchasing, handling, and disposing of large quantities of inorganic oxidants adds a substantial premium to the final product price. By utilizing electrons as the oxidant, this variable cost is virtually eliminated. Additionally, the catalytic nature of the iodide salt means that expensive reagents are not consumed in a 1:1 ratio with the product but are instead regenerated continuously. This leads to a significant reduction in raw material expenditure. Moreover, the simplified workup procedure, which involves merely adding water to precipitate the product, removes the need for expensive chromatographic silica gel and large volumes of organic extraction solvents. These cumulative savings contribute to a much more competitive pricing structure for the final intermediate.
- Enhanced Supply Chain Reliability: Dependence on specialized chemical oxidants can introduce fragility into the supply chain, especially if those reagents are subject to regulatory restrictions or transportation hazards. This electrochemical method relies on commodity chemicals—potassium iodide and DMSO—which are widely available from multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions, which proceed efficiently at room temperature without the need for inert gas protection (nitrogen or argon), further enhances operational reliability. This means that production can continue uninterrupted even if there are temporary disruptions in the supply of specialty gases. For supply chain planners, this resilience ensures consistent delivery schedules and reduces the need for large safety stocks of hazardous reagents.
- Scalability and Environmental Compliance: Scaling electrochemical processes is inherently safer than scaling exothermic chemical oxidations, as the reaction rate is controlled by the applied current rather than by mixing or heat transfer limitations. This makes the technology highly amenable to commercial scale-up of complex pharmaceutical intermediates. From an environmental standpoint, the absence of heavy metal catalysts and stoichiometric salt waste simplifies wastewater treatment. The patent explicitly notes that the filtrate containing the iodide catalyst and DMSO can be recycled after simple water removal, drastically reducing the volume of liquid waste generated. This ease of waste management facilitates compliance with increasingly strict environmental regulations, avoiding potential fines and shutdowns. The overall process footprint is smaller, cleaner, and more sustainable, positioning the manufacturer as a leader in green chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation. They are intended to provide clarity for engineering teams evaluating the feasibility of adopting this technology for their specific production lines. Understanding these nuances is crucial for making informed decisions about process integration and resource allocation.
Q: Why is electrochemical synthesis preferred over visible-light methods for aminocoumarins?
A: Electrochemical methods eliminate the need for stoichiometric chemical oxidants like peroxodisulfate, which generate significant inorganic waste and complicate purification. Using electrons as a traceless oxidant simplifies downstream processing and improves atom economy.
Q: Can the catalyst and solvent be recycled in this process?
A: Yes, the patent data demonstrates that the DMSO solvent and iodide catalyst system can be directly recycled after simple water removal. This significantly reduces raw material consumption and waste disposal costs compared to single-use systems.
Q: What is the functional group tolerance of this electrochemical selenylation?
A: The method exhibits excellent compatibility with various substituents on the diaryl diselenide, including halogens (F, Cl), trifluoromethyl groups, cyano groups, and esters, yielding products with high purity and minimal side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylselenyl-4-Aminocoumarin Supplier
As the global demand for complex heterocyclic intermediates continues to rise, partnering with a technically proficient CDMO is essential for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle electrochemical synthesis and other advanced organic transformations with the highest standards of safety and quality. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of 3-arylselenyl-4-aminocoumarin meets the exacting requirements of the pharmaceutical industry. Our commitment to excellence ensures that you receive a product that is not only chemically pure but also consistent in quality, supporting your downstream drug development efforts without delay.
We invite you to engage with our technical procurement team to discuss how this innovative electrochemical route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits applicable to your volume requirements. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique molecular targets. Whether you require small quantities for preclinical studies or metric tons for commercial launch, our team is ready to provide the support and expertise needed to bring your vision to market efficiently and sustainably.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
