Scalable One-Step Synthesis of Tetrahydrophenazine Derivatives for Advanced Material Applications
Introduction to Advanced Tetrahydrophenazine Synthesis
The landscape of organic synthesis for heterocyclic compounds is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and cost-effective manufacturing processes. A pivotal advancement in this domain is detailed in patent CN110776472B, which discloses a novel preparation method for tetrahydrophenazine derivatives. These compounds are not merely academic curiosities; they serve as critical modular molecules with extensive applications ranging from fluorescent biomarkers and dyes to organic semiconductors and solar cell materials. Historically, the synthesis of such complex heterocycles has been plagued by harsh reaction conditions and multi-step procedures that hinder commercial viability. However, this new technology leverages a sophisticated one-step reductive cyclization strategy, utilizing stable nitroaniline compounds and biomass-derived diphenol compounds as primary feedstocks. By integrating transition metal catalysis with efficient reduction protocols, this method achieves high atom economy and exceptional functional group tolerance. For R&D directors and procurement specialists alike, this represents a paradigm shift towards sustainable chemical manufacturing, offering a reliable tetrahydrophenazine supplier pathway that aligns with modern industrial standards for safety and efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for tetrahydrophenazines have long been burdened by significant operational and economic drawbacks that limit their scalability. Classical approaches often rely on the condensation of o-phenylenediamine with catechols, a process that necessitates sealing reactions in vessels at extreme temperatures of 200-210°C for prolonged periods of 35 to 40 hours. Such energy-intensive conditions impose severe stress on reactor equipment and increase the risk of thermal hazards. Furthermore, alternative pathways involving 2-nitrophenylenediamine require the use of dimethyl sulfoxide (DMSO) as a solvent and strong alkali metal alcoholates, leading to difficult purification challenges that often mandate resource-heavy column chromatography. Other methods employing halogenated ketones introduce stoichiometric amounts of strong bases and environmentally harmful halogenated reagents, generating substantial waste streams. These legacy processes are characterized by low atom economy, complex workup procedures, and the generation of large volumes of wastewater and solid residue, making them increasingly untenable in a regulatory environment focused on green chemistry principles.
The Novel Approach
In stark contrast to these archaic methodologies, the innovative process described in CN110776472B introduces a streamlined, one-pot synthesis that fundamentally redefines the production of tetrahydrophenazine derivatives. This approach utilizes readily accessible nitroamine compounds, structured as shown in general formula (1), and diphenol compounds, depicted in general formula (2), as the foundational building blocks. 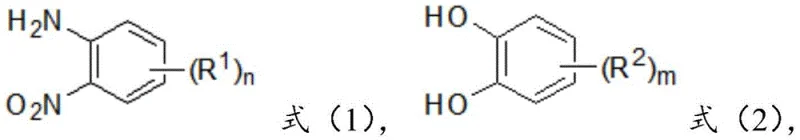
 By combining these precursors in the presence of a metal catalyst, an additive, a reducing agent, and a solvent, the reaction proceeds smoothly to yield the target heterocycle without the need for intermediate isolation. The versatility of this system is evident in the broad scope of substituents allowed; R1 groups can include methyl, methoxy, amino, piperidinyl, or trifluoromethyl moieties, while R2 groups accommodate methyl or ester functionalities. This flexibility allows for the rapid generation of diverse chemical libraries, essential for drug discovery and material science applications. The elimination of high-pressure, high-temperature steps and the avoidance of toxic halogenated reagents significantly lower the barrier to entry for commercial production, establishing a new benchmark for cost reduction in pharmaceutical intermediates manufacturing.
By combining these precursors in the presence of a metal catalyst, an additive, a reducing agent, and a solvent, the reaction proceeds smoothly to yield the target heterocycle without the need for intermediate isolation. The versatility of this system is evident in the broad scope of substituents allowed; R1 groups can include methyl, methoxy, amino, piperidinyl, or trifluoromethyl moieties, while R2 groups accommodate methyl or ester functionalities. This flexibility allows for the rapid generation of diverse chemical libraries, essential for drug discovery and material science applications. The elimination of high-pressure, high-temperature steps and the avoidance of toxic halogenated reagents significantly lower the barrier to entry for commercial production, establishing a new benchmark for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Pd-Catalyzed Reductive Cyclization
The core of this technological breakthrough lies in the elegant mechanistic pathway that facilitates the construction of the phenazine core through a concerted reductive cyclization. The reaction initiates with the activation of the nitro group on the aniline derivative by the transition metal catalyst, which may include palladium species such as palladium acetate, palladium on carbon, or copper salts like cuprous iodide. Simultaneously, the diphenol component undergoes coordination, setting the stage for nucleophilic attack and ring closure. The presence of a reducing agent, which can range from hydrogen gas at mild pressures (1-20 atm) to silane reagents or formates, is crucial for driving the reduction of the nitro functionality to an amine in situ, followed immediately by condensation with the adjacent carbonyl or phenolic centers. This cascade sequence effectively bypasses the need for isolating unstable intermediates, thereby minimizing side reactions and decomposition. The catalytic cycle is further optimized by the inclusion of additives such as potassium carbonate, cesium carbonate, or organic bases like triethylamine, which modulate the pH and facilitate proton transfer steps essential for aromatization and final product stabilization.
From an impurity control perspective, this mechanism offers distinct advantages over traditional oxidative coupling methods. The reductive nature of the process inherently suppresses the formation of over-oxidized byproducts that are common in aerobic conditions. Furthermore, the high selectivity of the catalyst system ensures that sensitive functional groups, such as esters or trifluoromethyl groups, remain intact throughout the transformation. This chemoselectivity is vital for maintaining the integrity of complex molecular architectures required in advanced material applications. The general reaction equation illustrates this transformative process, highlighting the convergence of two simple aromatic systems into a fused heterocyclic framework with the loss of only small molecule byproducts like water. 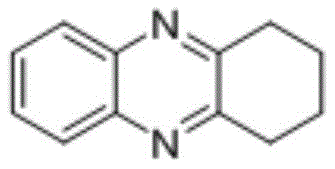 Such mechanistic efficiency translates directly to higher purity profiles in the crude reaction mixture, reducing the burden on downstream purification units and enhancing the overall yield of the commercial scale-up of complex pharmaceutical intermediates.
Such mechanistic efficiency translates directly to higher purity profiles in the crude reaction mixture, reducing the burden on downstream purification units and enhancing the overall yield of the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Tetrahydrophenazine Derivatives Efficiently
Implementing this synthesis protocol in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The process is remarkably robust, accommodating a wide range of solvents including acetonitrile, toluene, p-xylene, and even alcohols like isopropanol, allowing chemists to select media based on solubility and safety profiles. The molar ratio of the nitroamine to the diphenol is a critical variable, with experimental data indicating that a ratio of 1:1.5 often provides optimal conversion rates, although ratios ranging from 1:1 to 1:10 are feasible depending on the specific electronic properties of the substrates. Reaction temperatures can be tuned between 25°C and 130°C, offering the flexibility to run reactions at ambient conditions for sensitive substrates or elevated temperatures to accelerate kinetics for sterically hindered systems. The detailed standardized synthesis steps below outline the precise operational procedure derived from the patent examples, ensuring that technical teams can replicate the high performance observed in the initial disclosures.
- Charge a reactor with nitroamine compounds, biomass-derived diphenol compounds, a transition metal catalyst (such as Pd/C or CuI), an additive base, a reducing agent, and a suitable organic solvent.
- Maintain the reaction mixture at a temperature between 25°C and 130°C for a duration of 1 to 24 hours under stirring, optionally under hydrogen pressure or inert atmosphere depending on the reductant.
- Upon completion, cool the mixture to room temperature, remove the solvent via rotary evaporation, and purify the crude tetrahydrophenazine derivative using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route presents a compelling value proposition centered on cost efficiency and supply security. The shift from multi-step, high-energy processes to a single-pot catalytic reaction drastically simplifies the manufacturing workflow, eliminating the need for expensive high-pressure reactors and the associated safety infrastructure. This simplification directly correlates to a reduction in capital expenditure (CAPEX) and operational expenditure (OPEX), as the process can be executed in standard glass-lined or stainless steel reactors commonly found in fine chemical facilities. Moreover, the reliance on biomass-derived diphenols and commodity nitroanilines ensures a stable and diversified raw material supply chain, mitigating the risks associated with sourcing specialized or scarce reagents. The ability to produce high-purity tetrahydrophenazine derivatives with minimal waste generation also aligns with increasingly stringent environmental regulations, potentially lowering waste disposal costs and enhancing the company's sustainability profile.
- Cost Reduction in Manufacturing: The elimination of stoichiometric strong bases and hazardous halogenated reagents removes significant cost drivers associated with reagent procurement and hazardous waste treatment. By utilizing catalytic amounts of metal species and benign reducing agents, the process achieves a leaner material balance. The simplified workup procedure, which often involves mere solvent removal followed by standard purification, reduces labor hours and solvent consumption compared to the tedious column chromatography often required by older methods. These cumulative efficiencies result in substantial cost savings per kilogram of produced API intermediate, enhancing the overall competitiveness of the final product in the global market.
- Enhanced Supply Chain Reliability: The use of stable, commercially available starting materials such as nitroanilines and catechols ensures that production schedules are not held hostage by the lead times of exotic custom synthons. The robustness of the reaction conditions, which tolerate a variety of solvents and additives, provides manufacturing flexibility; if a specific solvent faces supply constraints, alternatives like toluene or alcohols can be substituted without compromising reaction efficacy. This adaptability is crucial for maintaining continuous production runs and meeting tight delivery windows for downstream clients in the pharmaceutical and agrochemical sectors, effectively reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The one-pot nature of the reaction minimizes the number of unit operations required, facilitating a smoother transition from gram-scale laboratory synthesis to ton-scale commercial production. The high atom economy means that a greater proportion of raw material mass is incorporated into the final product, reducing the volume of effluent generated. This inherent green chemistry characteristic simplifies compliance with environmental discharge standards and reduces the load on wastewater treatment facilities. Consequently, the process supports sustainable growth strategies, allowing manufacturers to scale up capacity without proportionally increasing their environmental footprint or regulatory burden.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetrahydrophenazine synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the practical realities of scaling this chemistry, from catalyst selection to purification strategies, ensuring that stakeholders have a comprehensive view of the operational requirements.
Q: What are the key advantages of this new tetrahydrophenazine synthesis method over traditional routes?
A: Unlike traditional methods requiring high temperatures (200-210°C), high pressure, and multi-step sequences involving hazardous halogenated reagents, this novel process operates under milder conditions (25-130°C) in a single step. It utilizes stable, inexpensive nitroanilines and biomass-derived diphenols, significantly simplifying the workflow and reducing environmental impact.
Q: Which catalysts and reducing agents are compatible with this synthetic pathway?
A: The method demonstrates broad compatibility with various transition metal catalysts including palladium acetate, palladium on carbon, palladium hydroxide, and copper salts. Reducing agents can range from hydrogen gas (1-20 atm) to silane reagents, formates, or alcohols, offering flexibility for different scale-up requirements and safety protocols.
Q: Is this process suitable for large-scale commercial manufacturing of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It features high atom economy, uses readily available raw materials, and avoids complex purification steps often associated with traditional phenazine synthesis. The simple workup involving solvent removal and standard chromatography makes it highly viable for cost reduction in pharmaceutical intermediates manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrophenazine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced catalytic methodology in meeting the evolving demands of the global fine chemical market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of transition metal catalysis, including hydrogenation capabilities and rigorous QC labs dedicated to maintaining stringent purity specifications. We understand that consistency and quality are paramount for our clients, which is why we implement comprehensive analytical protocols to verify the structural integrity and purity of every batch of tetrahydrophenazine derivatives we produce.
We invite forward-thinking pharmaceutical and material science companies to collaborate with us to leverage this cutting-edge synthesis technology. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Together, we can drive innovation and efficiency in the supply of critical chemical intermediates, securing a competitive advantage in your respective markets.
