Scalable Electrochemical Construction of Thio(Seleno)Phosphoric Acid Esters via Hydrogen Evolution Oxidative Coupling
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more sustainable synthetic methodologies. A groundbreaking development in this field is detailed in Chinese Patent CN114411180B, which discloses a novel electrochemical hydrogen evolution oxidative coupling method for constructing thio(seleno)phosphoric acid ester compounds. This technology represents a paradigm shift from traditional stoichiometric oxidation processes to a catalytic, electricity-driven approach. By utilizing simple alcohols, elemental sulfur or selenium, and diphenylphosphine in an undivided electrochemical cell, the method achieves high yields of target products while completely avoiding the use of hazardous chemical oxidants. For R&D directors and process chemists, this innovation offers a compelling route to access valuable phosphorus-containing scaffolds that serve as critical building blocks for drug prodrugs, metal ligands, and flame retardants. The ability to generate these complex structures under mild conditions with excellent functional group tolerance positions this technology as a key enabler for the next generation of high-purity pharmaceutical intermediates.
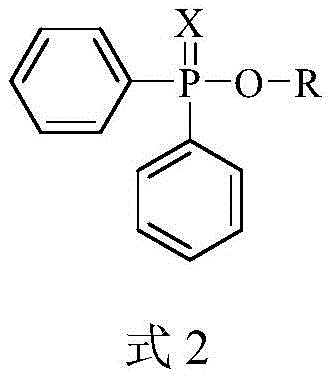
Historically, the synthesis of oxyphosphorothioates and related species has been fraught with significant operational and environmental challenges. Conventional methods typically rely on the cross-coupling of thiodisubstituted phosphine hydrides with alcohols or phenols, often necessitating the use of aggressive chlorinating agents like carbon tetrachloride or stoichiometric amounts of external oxidants. These traditional pathways suffer from poor atom economy, generate substantial toxic waste, and frequently require harsh reaction conditions, such as elevated temperatures exceeding 80°C. Furthermore, the reliance on air-sensitive phosphorus halides introduces severe handling risks and complicates the supply chain due to the need for specialized storage and transportation. From a procurement manager perspective, these legacy processes translate into higher raw material costs, expensive waste disposal fees, and increased regulatory compliance burdens. The inefficiency of these methods often results in lower overall throughput and inconsistent product quality, creating bottlenecks in the manufacturing of downstream active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The entrenched reliance on chemical oxidants and phosphorus halides in traditional synthesis creates a fragile supply chain vulnerable to raw material volatility and regulatory crackdowns on hazardous substances. Processes utilizing carbon tetrachloride are increasingly restricted globally due to their ozone-depleting potential and toxicity, forcing manufacturers to seek alternative, compliant routes. Additionally, the exothermic nature of many traditional oxidation reactions poses significant safety risks during scale-up, requiring expensive engineering controls and cooling systems. The generation of stoichiometric salt byproducts further complicates downstream purification, often necessitating energy-intensive crystallization or chromatography steps to meet the stringent purity specifications required by the pharmaceutical industry. These cumulative factors result in a high cost of goods sold (COGS) and extended lead times, undermining the competitiveness of manufacturers relying on outdated synthetic strategies.
The Novel Approach
In stark contrast, the electrochemical method described in patent CN114411180B leverages electrons as traceless reagents to drive the oxidative coupling, fundamentally altering the economic and environmental profile of the synthesis. By employing a simple undivided cell with a carbon anode and platinum cathode, the system facilitates the in situ generation of reactive radical species without the need for external oxidants. This approach not only eliminates the procurement and disposal costs associated with hazardous chemicals but also significantly simplifies the reaction workup. The use of elemental sulfur and selenium, which are abundant and inexpensive feedstocks, further enhances the cost-effectiveness of the process. As illustrated in the general reaction scheme below, the direct coupling of alcohols with diphenylphosphine and chalcogens proceeds smoothly under mild conditions, offering a robust platform for cost reduction in pharmaceutical intermediate manufacturing.
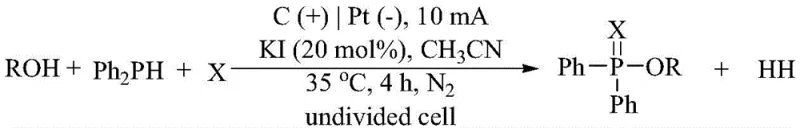
Mechanistic Insights into Electrochemical Hydrogen Evolution Oxidative Coupling
A deep understanding of the catalytic cycle is essential for R&D teams aiming to optimize this process for specific substrates. The mechanism initiates at the anode, where iodide ions from the electrolyte (preferably KI) are oxidized to generate iodine radicals. These highly reactive species abstract a hydrogen atom from diphenylphosphine, yielding a diphenylphosphino radical. Concurrently, the alcohol substrate interacts with electro-generated iodine cations to form a transient O-I intermediate, which undergoes homolytic cleavage to produce an alkoxy radical. The crucial bond-forming step involves the cross-coupling of the diphenylphosphino radical and the alkoxy radical to form an O-P(III) species. Finally, this intermediate reacts with elemental sulfur or selenium to furnish the stable O-P(V) thio(seleno)phosphoric acid ester product. This radical-mediated pathway avoids high-energy ionic intermediates, thereby minimizing side reactions and decomposition pathways that often plague ionic mechanisms.
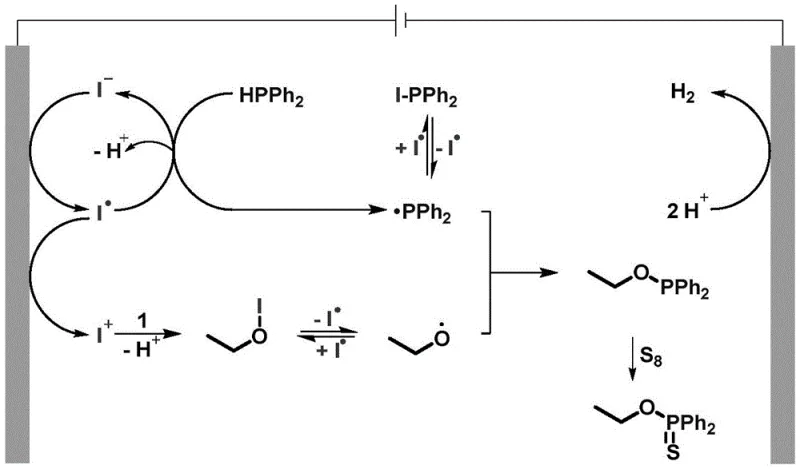
The elegance of this mechanism lies in its inherent impurity control. By avoiding strong chemical oxidants, the process prevents over-oxidation of sensitive functional groups, such as alkenes or alkynes, which might otherwise be degraded in traditional acidic or basic media. The use of a mediating electrolyte like KI ensures that the electron transfer is efficient and selective, reducing the likelihood of direct anodic oxidation of the organic substrates which could lead to polymerization or tar formation. Furthermore, the cathodic half-reaction simply evolves hydrogen gas, a benign byproduct that exits the system without requiring complex separation protocols. This clean reaction profile translates directly to higher crude purity, reducing the burden on purification units and increasing the overall yield of the isolated API intermediate. For process chemists, this means a more predictable impurity profile and a smoother path to regulatory approval.
How to Synthesize Thio(Seleno)Phosphoric Acid Esters Efficiently
Implementing this electrochemical protocol requires careful attention to reaction parameters to maximize efficiency and reproducibility. The standard procedure involves dissolving the alcohol, elemental sulfur or selenium, diphenylphosphine, and potassium iodide in acetonitrile under an inert nitrogen atmosphere. The choice of electrode materials is critical; a carbon rod anode paired with a platinum sheet cathode has been identified as the optimal configuration to ensure stable current density and minimize electrode degradation. The reaction is typically conducted at a constant current of 10 mA and a temperature of 35°C for approximately 4 hours. Following electrolysis, the solvent is removed under reduced pressure, and the crude product is purified via silica gel column chromatography. This straightforward workflow eliminates the need for specialized high-pressure equipment or cryogenic conditions, making it accessible for both laboratory discovery and pilot-scale production. The detailed standardized synthesis steps are provided in the guide below.
- Prepare the electrolyte solution by dissolving the alcohol compound, elemental sulfur or selenium, diphenylphosphine, and potassium iodide (KI) electrolyte in acetonitrile solvent under a nitrogen atmosphere.
- Set up an undivided electrochemical cell using a carbon rod as the anode and a platinum sheet as the cathode, ensuring proper electrode placement within the electrolyte.
- Apply a constant direct current (typically 10 mA) at a controlled temperature of 35°C for 4 hours, then purify the resulting crude mixture via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this electrochemical technology offers transformative benefits that extend far beyond the laboratory bench. The elimination of stoichiometric chemical oxidants and hazardous chlorinating agents drastically reduces the complexity of raw material sourcing and inventory management. By replacing expensive, regulated reagents with electricity and common salts, manufacturers can achieve substantial cost savings in direct material expenses. Moreover, the simplified waste stream, consisting primarily of benign salts and hydrogen gas, significantly lowers environmental compliance costs and waste treatment fees. This aligns perfectly with global sustainability goals and reduces the risk of supply disruptions caused by environmental regulations on hazardous chemical transport.
- Cost Reduction in Manufacturing: The replacement of costly chemical oxidants and air-sensitive phosphorus halides with electricity and elemental sulfur leads to a dramatic decrease in raw material costs. The process operates at ambient pressure and moderate temperatures, reducing energy consumption associated with heating and cooling. Additionally, the high atom economy minimizes waste generation, lowering the total cost of ownership for the manufacturing process. The simplified workup procedure reduces solvent usage and labor hours required for purification, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: Utilizing commodity chemicals like alcohols, diphenylphosphine, and potassium iodide ensures a stable and diversified supply base, mitigating the risk of single-source dependency. The robustness of the electrochemical method allows for flexible production scheduling, as the reaction can be easily started or stopped by controlling the power supply. This flexibility is crucial for responding to fluctuating market demands without the long lead times associated with procuring specialized reagents. The inherent safety of the process also reduces insurance premiums and facility downtime related to safety incidents.
- Scalability and Environmental Compliance: The undivided cell design is inherently scalable, allowing for seamless transition from gram-scale optimization to multi-ton commercial production without significant process redesign. The absence of toxic byproducts simplifies environmental permitting and ensures compliance with increasingly stringent global regulations on volatile organic compounds (VOCs) and hazardous waste. This green chemistry profile enhances the brand reputation of the manufacturer and facilitates easier market entry into regions with strict environmental standards, securing long-term business continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a reliable foundation for decision-making. Understanding these nuances is critical for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: What are the primary advantages of this electrochemical method over traditional phosphorus halide routes?
A: Unlike traditional methods that rely on air-sensitive phosphorus halides and stoichiometric chemical oxidants like carbon tetrachloride, this electrochemical approach utilizes electricity as a clean reagent. This eliminates hazardous waste streams, improves atom economy, and operates under significantly milder conditions, enhancing overall process safety.
Q: Does this synthesis method tolerate diverse functional groups on the alcohol substrate?
A: Yes, the method demonstrates excellent functional group compatibility. It successfully accommodates saturated and unsaturated aliphatic chains, halogen substituents (chloro, bromo), and aromatic systems including benzyl and phenoxy groups, making it highly versatile for synthesizing complex pharmaceutical intermediates.
Q: Is the electrochemical setup suitable for large-scale industrial production?
A: The process utilizes a simple undivided cell configuration with commercially available electrode materials like carbon rods and platinum sheets. The mild reaction temperatures and lack of explosive oxidants make the technology inherently safer and easier to scale up compared to exothermic traditional oxidation processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thio(Seleno)Phosphoric Acid Ester Supplier
As the pharmaceutical and agrochemical industries continue to demand more sustainable and efficient synthetic routes, NINGBO INNO PHARMCHEM stands at the forefront of adopting these cutting-edge technologies. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this electrochemical method are fully realized at an industrial level. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced electrochemical reactors capable of maintaining stringent purity specifications required for GMP manufacturing. We are committed to delivering high-quality intermediates that meet the evolving needs of our global partners.
We invite you to collaborate with us to leverage this innovative synthesis platform for your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this green chemistry approach can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the market through superior chemical manufacturing.
