Advanced Copper-Catalyzed Synthesis of Delta Epsilon Alkenyl Ketones for Pharmaceutical Intermediates
Introduction to Next-Generation Ketone Synthesis
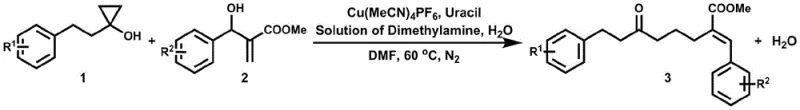
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for greener, more cost-effective methodologies that do not compromise on yield or purity. A significant breakthrough in this domain is detailed in patent CN115322100A, which discloses a novel preparation method for δ,ε-alkenyl ketone compounds. These structural motifs are ubiquitous in high-value bioactive molecules, including immunosuppressants like Sirolimus and Everolimus, as well as various natural products exhibiting cytotoxicity and hormonal activity. The traditional reliance on stoichiometric salt waste and expensive precursors has long been a bottleneck for industrial scalability. This new technology leverages a copper-catalyzed ring-opening allylation of cyclopropanols, utilizing simple allyl alcohols directly. For a reliable pharmaceutical intermediate supplier, mastering such atom-economical transformations is critical to maintaining competitiveness in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbon-carbon bonds to form δ,ε-alkenyl ketones has relied heavily on transition metal-promoted allylation reactions using pre-activated allylic reagents. These traditional pathways typically necessitate the use of allyl halides, allyl methanesulfonates, or allyl carbonates, which possess good leaving groups to facilitate the reaction. However, the preparation of these activated reagents is inherently inefficient, often requiring multiple synthetic steps and generating substantial amounts of stoichiometric salt waste as a byproduct. Furthermore, many existing protocols depend on relatively expensive catalysts that restrict their viability for large-scale industrial applications. From an environmental and economic perspective, the generation of chemical waste and the high cost of raw materials create significant barriers to entry for cost reduction in API manufacturing. The presence of difficult-to-remove impurities from these salt byproducts also complicates downstream purification, impacting the overall yield and quality of the final high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN115322100A introduces a paradigm shift by utilizing unactivated allyl alcohols directly as the olefin source. This approach capitalizes on the ring-opening of cyclopropanols to introduce the carbonyl fragment, driven by a cheap and readily available copper catalyst system. The reaction proceeds under mild conditions, typically between 50°C and 100°C, and remarkably produces water as the sole byproduct, thereby achieving high atom economy. This elimination of stoichiometric salt waste not only simplifies the workup procedure but also aligns with stringent green chemistry principles required by modern regulatory bodies. By bypassing the need for pre-functionalized allyl reagents, the process drastically shortens the synthetic route, offering a streamlined pathway for the commercial scale-up of complex organic syntheses. The versatility of this method allows for a broad substrate scope, accommodating various substituted phenyl and alkyl groups, making it an invaluable tool for diverse chemical libraries.
Mechanistic Insights into Copper-Catalyzed Ring-Opening Allylation
The core of this innovation lies in the unique catalytic cycle facilitated by copper species, such as tetraacetonitrile copper hexafluorophosphate or cuprous chloride, in conjunction with specific ligands like uracil or bipyridines. The mechanism likely involves the activation of the allyl alcohol by the copper center, overcoming the kinetic barrier associated with the poor leaving group ability of the hydroxyl moiety. Simultaneously, the strained three-membered ring of the cyclopropanol undergoes oxidative addition or coordination-induced ring opening, generating a reactive organometallic intermediate. This intermediate then engages in a transmetallation or migratory insertion step with the activated allyl species. Crucially, the catalyst system is designed to suppress competitive β-hydride elimination reactions, which are common side reactions in copper catalysis that lead to undesired byproducts. Instead, the system favors the formation of the thermodynamically stable δ,ε-alkenyl ketone framework. This precise control over the reaction pathway ensures high selectivity and minimizes the formation of regioisomers or reduction products.
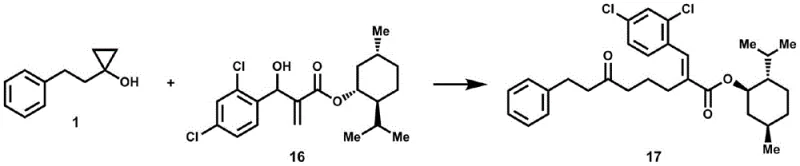
From an impurity control perspective, the use of water as the only byproduct is a game-changer for process chemistry. In traditional methods, inorganic salts can co-elute with the product or form emulsions during extraction, requiring extensive washing and drying steps that increase solvent consumption and processing time. In this novel protocol, the removal of the reaction solvent followed by standard chromatographic purification yields the target compound with exceptional purity. The compatibility of the reaction with various functional groups, including esters, ethers, and halides, further underscores its robustness. For R&D teams focused on process optimization, this means fewer unit operations and a reduced environmental footprint. The ability to modify complex natural product scaffolds, as demonstrated in the patent with menthol and cholesterol derivatives, highlights the method's tolerance to steric hindrance and sensitive functional groups, ensuring reliable production of complex intermediates.
How to Synthesize Delta Epsilon Alkenyl Ketones Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible roadmap for laboratory and pilot-scale production. The process begins with the preparation of the catalytic mixture under an inert atmosphere, ensuring that oxygen-sensitive copper species remain active throughout the reaction. The careful selection of solvents, such as N,N-dimethylformamide or tetrahydrofuran, plays a pivotal role in solubilizing both the polar catalyst system and the organic substrates. Following the reaction period, which can range from 12 to 48 hours depending on the substrate reactivity, the workup is remarkably straightforward. The detailed standardized synthesis steps below outline the precise molar ratios and conditions required to achieve optimal yields, serving as a foundational guide for process chemists aiming to implement this technology.
- Prepare the catalytic system by mixing copper catalyst, ligand, dimethylamine solution, and water in a solvent like DMF under inert gas.
- Add cyclopropanol and allyl alcohol substrates to the mixture and stir at 50-100°C for 12-48 hours.
- Remove the solvent via rotary evaporation and purify the crude product using column chromatography to isolate the target ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this copper-catalyzed methodology translates into tangible strategic advantages beyond mere technical feasibility. The shift away from precious metal catalysts and pre-activated reagents fundamentally alters the cost structure of producing δ,ε-alkenyl ketones. By leveraging abundant base metals and commodity chemicals like allyl alcohol, manufacturers can insulate themselves from the volatility of precious metal markets and the supply chain disruptions often associated with specialized reagents. Furthermore, the simplified purification process reduces the demand for bulk solvents and consumables, contributing to a leaner and more sustainable operation. This efficiency is crucial for reducing lead time for high-purity intermediates, allowing suppliers to respond more agilely to fluctuating market demands.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive catalysts and reagents with cost-effective alternatives. Traditional methods often require palladium or rhodium catalysts, along with activated allyl halides that necessitate separate synthesis steps. By utilizing inexpensive copper salts and direct allyl alcohols, the raw material costs are significantly lowered. Additionally, the generation of water as the sole byproduct eliminates the costs associated with the disposal of hazardous salt waste and the extensive aqueous workups required to remove them. This streamlined approach reduces the overall number of processing steps, leading to lower labor and utility costs per kilogram of product. The cumulative effect is a substantial reduction in the cost of goods sold (COGS), enabling more competitive pricing strategies in the global marketplace without sacrificing margin.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Allyl alcohols and cyclopropanols are commodity chemicals produced on a massive scale, ensuring a stable and continuous supply compared to niche activated reagents that may have limited vendors. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvents, further mitigates the risk of batch failures due to minor process deviations. This reliability is essential for maintaining consistent inventory levels and meeting just-in-time delivery schedules for downstream pharmaceutical clients. Moreover, the scalability of the reaction from gram to multi-kilogram scales has been demonstrated, providing confidence that supply can be ramped up quickly to meet surges in demand without the need for complex process re-engineering.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental impact intensify, the green credentials of this synthesis method offer a distinct competitive edge. The high atom economy and the absence of toxic heavy metal waste simplify compliance with environmental regulations such as REACH or local EPA standards. The reduction in solvent usage and waste generation lowers the burden on wastewater treatment facilities and reduces the carbon footprint of the manufacturing site. This alignment with sustainability goals not only avoids potential regulatory fines but also enhances the brand reputation of the manufacturer among eco-conscious clients. The ease of scale-up, supported by mild reaction temperatures and standard pressure conditions, ensures that the transition from pilot plant to full commercial production is smooth and predictable, minimizing capital expenditure on specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial production. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of using allyl alcohol over traditional allyl halides in this synthesis?
A: Using allyl alcohol eliminates the need for pre-activation steps required for halides or sulfonates, significantly reducing chemical waste (stoichiometric salts) and lowering raw material costs while producing only water as a byproduct.
Q: Can this copper-catalyzed method be applied to complex natural product derivatives?
A: Yes, the patent demonstrates successful application on complex scaffolds such as menthol, diacetone galactose, and cholesterol derivatives, proving its robustness for synthesizing high-value bioactive intermediates.
Q: What represents the primary cost-saving factor in this manufacturing process?
A: The primary cost drivers are reduced through the use of inexpensive copper catalysts instead of precious metals, the avoidance of expensive activated allyl reagents, and the simplified purification process due to high atom economy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Delta Epsilon Alkenyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115322100A for the production of high-value pharmaceutical intermediates. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex synthetic routes, including those involving sensitive natural product derivatives, positions us as a strategic partner for your most challenging projects.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your sourcing strategy and secure a reliable supply of high-quality δ,ε-alkenyl ketones for your next generation of therapeutic agents.
