Advanced Asymmetric Synthesis of Trifluoromethyl Bisoxazoles for High-Purity Anticancer Intermediates
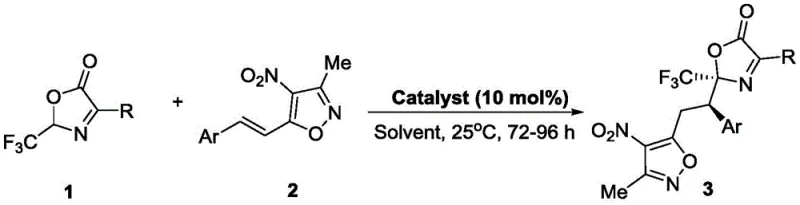
The pharmaceutical industry continuously seeks robust methodologies for introducing fluorine motifs into complex molecular scaffolds, driven by the profound impact of trifluoromethyl groups on drug pharmacokinetics and metabolic stability. Patent CN112898285A discloses a groundbreaking approach to synthesizing trifluoromethyl-containing bisoxazole compounds, which serve as potent intermediates in the development of next-generation anticancer therapeutics. This technology addresses a long-standing challenge in organic synthesis: the direct construction of carbon-carbon bonds using alpha-trifluoromethyl nucleophiles without compromising stereochemical integrity. By leveraging a novel thiourea organocatalytic system, the invention enables the coupling of 4-substituted-2-trifluoromethyl oxazolones with arylalkenyl oxazoles under exceptionally mild conditions. For R&D directors and process chemists, this represents a significant leap forward, offering a pathway to high-value chiral building blocks that were previously difficult to access with high optical purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of chiral centers adjacent to trifluoromethyl groups has been plagued by significant synthetic hurdles that hinder commercial viability. Conventional strategies often rely on direct asymmetric trifluoromethylation or the use of transition metal catalysts, which introduce severe complications for pharmaceutical manufacturing. A primary issue is the instability of alpha-CF3 carbanions, which are prone to beta-fluorine elimination, leading to the formation of unwanted difluoroolefin byproducts that are difficult to separate. Furthermore, methods employing strong electron-withdrawing groups often suffer from reduced nucleophilicity, requiring harsh reaction conditions that degrade sensitive functional groups. From a regulatory perspective, the reliance on transition metals poses a critical supply chain risk, as removing trace metal residues to meet ICH Q3D guidelines adds costly purification steps and extends lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology described in CN112898285A circumvents these historical bottlenecks by employing a metal-free organocatalytic strategy that operates at ambient temperatures. Instead of struggling with unstable carbanions, this approach utilizes 4-substituted-2-trifluoromethyl oxazolones as stable electrophilic building blocks that react efficiently with arylalkenyl oxazoles. The use of chiral thiourea catalysts facilitates the reaction through a dual hydrogen-bonding activation mechanism, ensuring precise stereocontrol without the need for cryogenic conditions or inert atmospheres. This shift not only simplifies the operational complexity but also drastically improves the atom economy and environmental profile of the synthesis. For procurement managers, this translates to cost reduction in API manufacturing by eliminating expensive metal scavengers and reducing energy consumption associated with heating or cooling large-scale reactors.
Mechanistic Insights into Thiourea-Catalyzed Asymmetric Coupling
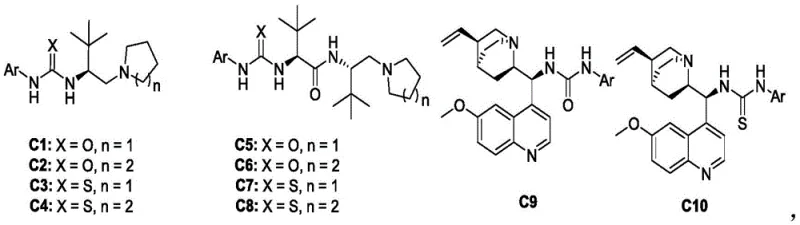
The success of this synthesis lies in the sophisticated design of the chiral thiourea catalysts, particularly the squaramide and thiourea derivatives labeled C1 through C14 in the patent data. These catalysts function by simultaneously activating both the nucleophile and the electrophile through a network of hydrogen bonds, creating a rigid chiral environment that dictates the facial selectivity of the attack. The bulky tert-butyl groups and the specific aromatic substituents on the catalyst backbone play a crucial role in shielding one face of the reactive intermediate, thereby enforcing the formation of a single enantiomer with high fidelity. This mechanistic precision is evidenced by the experimental results, where optimized catalysts like C10 consistently deliver diastereomeric ratios greater than 20:1 and enantiomeric excess values up to 96%. Such high selectivity is paramount for R&D teams, as it minimizes the formation of diastereomeric impurities that could complicate downstream crystallization or require resource-intensive chiral separations.
Furthermore, the tolerance of this catalytic system towards diverse electronic and steric environments underscores its robustness for library synthesis. The reaction proceeds efficiently regardless of whether the aryl ring on the oxazole substrate bears electron-withdrawing groups like nitro or halogens, or electron-donating groups like methoxy and methyl. This broad substrate scope suggests that the transition state is stabilized primarily by the catalyst-substrate interactions rather than being overly sensitive to the electronic nature of the incoming nucleophile. For process development, this implies a versatile platform technology capable of generating a wide array of analogues for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new derivative, thereby accelerating the drug discovery timeline significantly.
How to Synthesize Trifluoromethyl Bisoxazole Efficiently
Implementing this synthesis on a pilot or commercial scale requires strict adherence to the optimized parameters identified during the patent's screening phase to ensure consistent quality and yield. The process involves dissolving the 4-substituted-2-trifluoromethyl oxazolone and the arylalkenyl oxazole in a suitable solvent such as pentafluorobenzene or chlorobenzene, followed by the addition of the chiral thiourea catalyst at a loading of 5 to 10 mol%. The reaction mixture is then stirred at a controlled temperature of 25°C for a period ranging from 72 to 96 hours, allowing the slow but highly selective bond formation to reach completion. Detailed standardized synthetic steps see the guide below.
- Combine 4-substituted-2-trifluoromethyl oxazolone and arylalkenyl oxazole in an organic solvent such as pentafluorobenzene.
- Add a chiral thiourea catalyst (e.g., C10) at a loading of 5-10 mol% to the reaction mixture.
- Stir the reaction at 25°C for 72-96 hours, then purify the crude product via column chromatography to isolate the high-purity bisoxazole.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this organocatalytic route offers distinct strategic advantages over traditional metal-mediated processes. The elimination of precious metals such as palladium, rhodium, or iridium from the catalyst system removes a major variable cost driver and mitigates the risk of supply disruptions associated with geopolitical instability in mining regions. Additionally, the mild reaction conditions (25°C) significantly reduce the energy load required for temperature control, contributing to a lower carbon footprint and reduced utility costs per kilogram of product. The simplicity of the post-reaction workup, which typically involves solvent removal and standard column chromatography, further streamlines the manufacturing workflow, enabling faster turnaround times from raw material intake to finished intermediate.
- Cost Reduction in Manufacturing: The absence of transition metals eliminates the need for expensive metal scavenging resins and rigorous testing for residual metals, which are mandatory for GMP compliance. This simplification of the purification train directly lowers the cost of goods sold (COGS) and reduces the consumption of specialized consumables. Moreover, the high stereoselectivity achieved means that less material is wasted as unwanted isomers, improving the overall yield and maximizing the value derived from each batch of starting materials.
- Enhanced Supply Chain Reliability: The starting materials, specifically the oxazolones and arylalkenyl oxazoles, are structurally simple and can be sourced from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction across different solvents, including common industrial solvents like toluene and chlorobenzene, provides flexibility in sourcing and allows manufacturers to switch solvents based on availability and price fluctuations without compromising reaction performance.
- Scalability and Environmental Compliance: Operating at ambient temperature and pressure makes this process inherently safer and easier to scale from gram to multi-ton quantities without requiring specialized high-pressure equipment. The use of organocatalysts aligns with green chemistry principles by avoiding heavy metal waste streams, simplifying wastewater treatment and ensuring compliance with increasingly stringent environmental regulations regarding hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these trifluoromethyl bisoxazole intermediates. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing drug development pipeline. The answers provided are derived directly from the experimental data and technical specifications outlined in the patent documentation.
Q: What is the stereoselectivity of this thiourea-catalyzed reaction?
A: The method achieves excellent stereoselectivity, with diastereomeric ratios (d.r.) typically exceeding 20:1 and enantiomeric excess (ee) values reaching up to 96% when using optimized catalysts like C10.
Q: Does this synthesis involve heavy metal catalysts?
A: No, this process utilizes metal-free organocatalysis based on chiral thiourea derivatives, eliminating the risk of toxic metal residue contamination in the final pharmaceutical intermediate.
Q: What are the biological applications of these compounds?
A: The synthesized trifluoromethyl-containing bisoxazole compounds demonstrate significant inhibitory activity against various cancer cell lines, including liver (HepG2), lung (PC-9), and gastric (MGC-803) cancers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Bisoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the successful development of oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from laboratory discovery to clinical supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to guarantee that every batch of trifluoromethyl bisoxazole meets the exacting standards required for pharmaceutical applications.
We invite you to contact our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into potential efficiency gains specific to your project needs. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process and help you secure a reliable supply of these vital anticancer intermediates.
