Scalable Metal-Free Synthesis of Functionalized Pyridine Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and agrochemical industries are constantly seeking more efficient and environmentally benign pathways to construct heterocyclic scaffolds, particularly the ubiquitous pyridine ring. Patent CN107286085B, published in mid-2020, introduces a groundbreaking synthetic methodology for producing a diverse array of pyridine derivatives, specifically functionalized nicotinate esters. This technology represents a significant paradigm shift from traditional transition-metal catalysis to a more sustainable base-promoted cyclization strategy. The core innovation lies in the ability to construct the pyridine core through an intramolecular cyclization and simultaneous desulfurization of thioamide-substituted enyne esters. This approach not only circumvents the need for costly noble metals but also streamlines the purification process, making it highly attractive for the manufacturing of high-purity pharmaceutical intermediates. The structural versatility of the resulting compounds, as defined by Formula (I), allows for extensive downstream functionalization, catering to the complex needs of modern drug discovery pipelines.
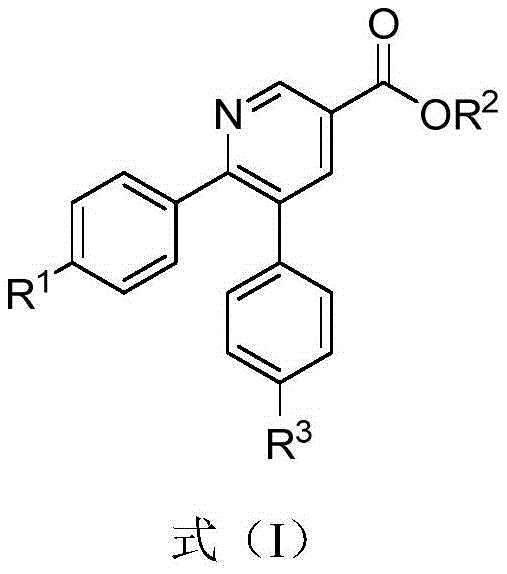
As a reliable pyridine derivative supplier, understanding the nuances of such synthetic breakthroughs is critical for maintaining a competitive edge in the global supply chain. The compounds generated via this method possess substituents such as halogens, nitro groups, and alkoxy chains, which serve as essential handles for further chemical elaboration. These structural features are indispensable in the synthesis of bioactive molecules, including potential antiviral agents and insecticides like chlorantraniliprole analogues. By leveraging this patent technology, manufacturers can access a broader chemical space with reduced regulatory hurdles associated with heavy metal residues, thereby accelerating the time-to-market for new therapeutic candidates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of substituted pyridine rings has heavily relied on transition-metal catalysis, utilizing precious metals such as palladium, rhodium, or ruthenium to facilitate C-H activation and cyclization reactions. While effective, these conventional methods suffer from significant drawbacks that impact both cost and environmental sustainability. The primary concern is the toxicity and high cost of the catalysts themselves, which necessitate rigorous purification steps to ensure the final active pharmaceutical ingredient (API) meets stringent residual metal limits. Furthermore, many metal-catalyzed processes require inert atmospheres and anhydrous conditions, adding complexity and operational costs to the manufacturing process. The generation of heavy metal waste also poses a substantial environmental burden, complicating waste disposal and increasing the overall carbon footprint of the synthesis. These factors collectively hinder the scalability and economic viability of traditional routes, especially for commodity-level production of fine chemical intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN107286085B utilizes a catalytic amount of an inexpensive inorganic base, specifically potassium tert-butoxide (t-BuOK), to drive the cyclization. This metal-free approach fundamentally alters the economic and operational landscape of pyridine synthesis. By replacing noble metals with earth-abundant alkali bases, the process eliminates the risk of heavy metal contamination entirely, thereby simplifying the downstream purification workflow. The reaction proceeds efficiently in polar aprotic solvents like NMP, often with the addition of water, which is unusual for many organometallic transformations but beneficial here for the desulfurization step. This tolerance to water and operation under ambient air conditions significantly reduces the engineering controls required for production. Consequently, this novel approach offers a robust, cost-effective, and environmentally friendly alternative that aligns perfectly with the principles of green chemistry, providing a distinct advantage for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Catalyzed Cyclization and Desulfurization
The mechanistic pathway of this transformation involves a sophisticated sequence of base-mediated deprotonation, nucleophilic attack, and elimination steps that ultimately forge the pyridine ring. The reaction initiates with the deprotonation of the thioamide nitrogen or the alpha-position of the ester by the strong base, generating a reactive nucleophilic species. This species then undergoes an intramolecular cyclization onto the alkyne moiety of the enyne ester precursor. Unlike metal-catalyzed versions that might proceed through oxidative addition and reductive elimination cycles, this base-catalyzed route relies on the inherent electronic properties of the substrate and the basicity of the medium to drive ring closure. The presence of water plays a crucial role in the subsequent desulfurization step, facilitating the removal of the sulfur atom from the thioamide group and aromatization of the ring to yield the stable pyridine system. This tandem cyclization-desulfurization process is highly efficient and avoids the formation of stable metal-sulfur complexes that often poison catalysts in traditional methods.
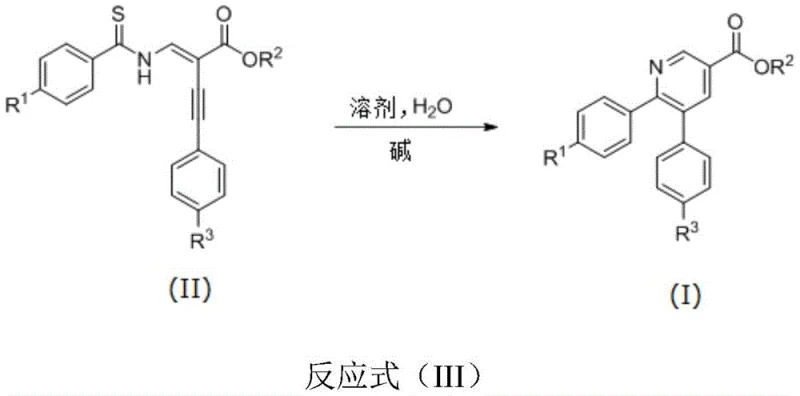
From an impurity control perspective, this mechanism offers distinct advantages. The absence of metal catalysts means there are no metal-ligand complexes or metal-induced side reactions to monitor, simplifying the impurity profile. The primary byproducts are likely inorganic salts and sulfur-containing species that are easily removed during aqueous workup. The reaction conditions, typically ranging from 50°C to 110°C, are mild enough to prevent thermal degradation of sensitive functional groups while being energetic enough to overcome the activation barrier for cyclization. The broad substrate scope, accommodating electron-withdrawing groups like nitro and halogens as well as electron-donating alkoxy groups, suggests a robust mechanistic tolerance. This reliability ensures consistent quality and high purity specifications, which are paramount for clients seeking high-purity OLED material precursors or specialized pharmaceutical building blocks.
How to Synthesize Ethyl 6-(4-chlorophenyl)-5-(4-methoxyphenyl)nicotinate Efficiently
To implement this synthesis effectively, precise control over reaction parameters such as temperature, stoichiometry, and solvent choice is essential. The patent outlines a generalized procedure that can be adapted for various substrates within the defined scope. Typically, the reaction is conducted in NMP solvent with a specific molar ratio of substrate to base to water, ensuring optimal conversion rates. The process is designed to be operationally simple, requiring standard laboratory equipment without the need for specialized gloveboxes or high-pressure reactors. For detailed operational protocols and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining the thioamide-substituted enyne ester starting material, potassium tert-butoxide (t-BuOK) catalyst, and NMP solvent in a reaction vessel under air atmosphere.
- Add water to the reaction mixture to facilitate the cyclization and desulfurization process, ensuring the molar ratio of substrate to catalyst to water is approximately 1.0: 1.5:15.0.
- Heat the reaction mixture to 90°C and maintain stirring for 24 to 26 hours until TLC analysis confirms complete consumption of the starting material, followed by purification to isolate the target pyridine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this base-catalyzed synthesis route translates into tangible strategic benefits beyond mere chemical efficiency. The shift away from precious metal catalysts directly impacts the bill of materials, removing a volatile cost component subject to global market fluctuations. Moreover, the simplified workup procedure reduces the consumption of scavenging resins and specialized filtration media, leading to substantial cost savings in consumables. The ability to run reactions under air rather than inert gas lowers utility costs and increases reactor throughput by reducing turnaround times associated with purging and leak testing. These factors collectively enhance the overall economic viability of the production process, making it a superior choice for large-scale manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts such as palladium or rhodium removes a significant cost driver from the production budget. Additionally, the simplified purification process reduces the need for costly metal scavengers and extensive chromatographic separations. This streamlined workflow lowers labor and material costs, resulting in a more competitive pricing structure for the final pyridine derivatives. The use of common industrial solvents and bases further ensures that raw material procurement remains stable and affordable.
- Enhanced Supply Chain Reliability: Relying on earth-abundant reagents like potassium tert-butoxide mitigates the risk of supply disruptions often associated with rare earth metals or specialized ligands. The robustness of the reaction conditions, including tolerance to water and air, reduces the likelihood of batch failures due to minor environmental deviations. This reliability ensures consistent delivery schedules and strengthens the resilience of the supply chain against external shocks. Suppliers can maintain higher inventory levels of key starting materials without the fear of rapid degradation or sensitivity issues.
- Scalability and Environmental Compliance: The metal-free nature of this synthesis significantly simplifies waste management and regulatory compliance. Without heavy metal waste streams, the environmental footprint of the manufacturing process is drastically reduced, easing the burden on wastewater treatment facilities. This alignment with green chemistry principles facilitates smoother regulatory approvals and enhances the corporate sustainability profile. The process is inherently scalable, allowing for seamless transition from pilot plant to commercial production volumes without significant re-engineering of the process infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. They are derived from the specific experimental data and beneficial effects outlined in the patent documentation. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production workflows.
Q: What are the primary advantages of this pyridine synthesis method over traditional metal-catalyzed routes?
A: The primary advantage is the elimination of expensive and toxic noble metal catalysts. Traditional methods often rely on palladium or rhodium, which require complex removal steps to meet pharmaceutical purity standards. This base-catalyzed method uses inexpensive potassium tert-butoxide, significantly simplifying post-treatment and reducing heavy metal contamination risks.
Q: What is the typical yield range for this synthesis protocol?
A: According to the patent data, the isolated yields for this synthetic method generally range from 10% to 60%, depending on the specific substituents on the aromatic rings. For example, the synthesis of ethyl 6-(4-chlorophenyl)-5-(4-methoxyphenyl)nicotinate achieved a 60% yield under optimized conditions at 90°C.
Q: Can this method be scaled for industrial production of agrochemical intermediates?
A: Yes, the method is highly suitable for scale-up. It utilizes common industrial solvents like NMP and operates under air atmosphere rather than requiring inert gas protection. The use of robust inorganic bases and the absence of sensitive metal catalysts make the process more forgiving and easier to manage in large-scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthetic methodologies like the one described in CN107286085B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into industrial reality. Our state-of-the-art facilities are equipped to handle base-catalyzed reactions with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. We are committed to delivering high-purity pyridine derivatives that meet the exacting standards of the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can optimize your supply chain and accelerate the development of your next-generation therapeutic or agricultural products.
